2 The basicity of amines depends on The

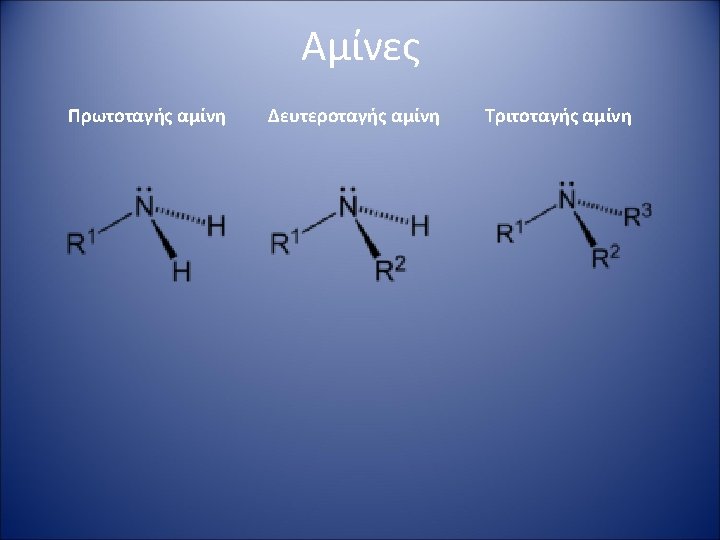
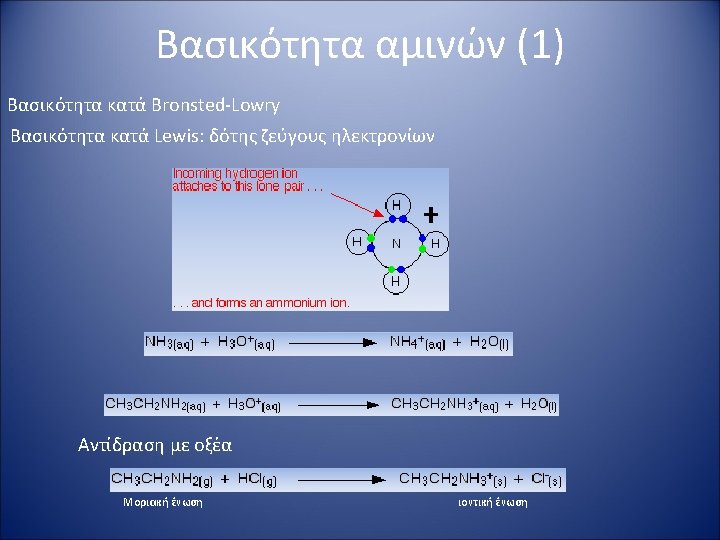

Βασικότητα αμινών (2) The basicity of amines depends on: • The electronic properties of the substituents (alkyl groups enhance the basicity, aryl groups diminish it). • Steric hindrance offered by the groups on nitrogen. • The degree of solvation of the protonated amine. Solubility in water: Primary ammonium (RNH 3+) > secondary ammonium (R 2 NH 2+) > tertiary ammonium (R 3 NH+). Quaternary ammonium salts usually exhibit the lowest solubility of the series
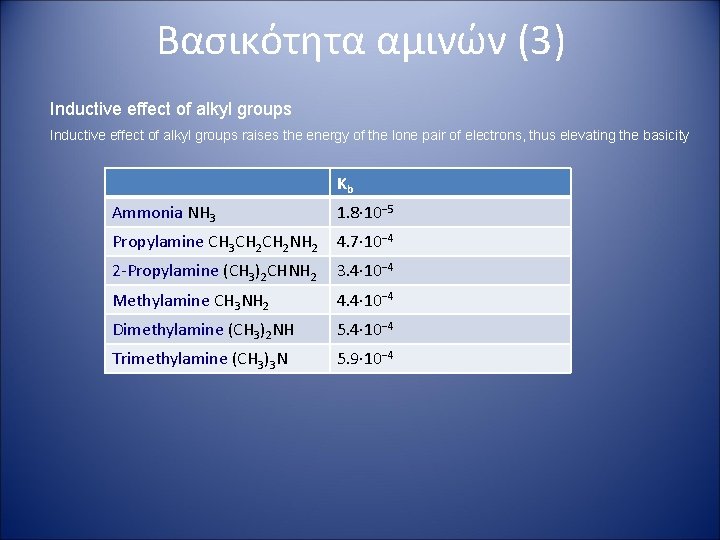
Βασικότητα αμινών (3) Inductive effect of alkyl groups raises the energy of the lone pair of electrons, thus elevating the basicity Kb Ammonia NH 3 1. 8· 10− 5 Propylamine CH 3 CH 2 NH 2 4. 7· 10− 4 2 -Propylamine (CH 3)2 CHNH 2 3. 4· 10− 4 Methylamine CH 3 NH 2 4. 4· 10− 4 Dimethylamine (CH 3)2 NH 5. 4· 10− 4 Trimethylamine (CH 3)3 N 5. 9· 10− 4

Βασικότητα αμινών (4) Mesomeric effect of aromatic systems Mesomeric effect of aromatic ring delocalises the lone pair of electrons on nitrogen into the ring, resulting in decreased basicity. Substituents on the aromatic ring, and their positions relative to the amine group may also considerably alter basicity. Kb Ammonia NH 3 1. 8· 10− 5 Aniline C 6 H 5 NH 2 3. 8· 10− 10 4 -Methylaniline 4 -CH 3 C 6 H 4 NH 2 1. 2· 10− 9 2 -Nitroaniline 1. 5· 10− 15 3 -Nitroaniline 2. 8· 10− 13 4 -Nitroaniline 9. 5· 10− 14



Ποιοτική ανάλυση αμινών Aliphatic amines (primary, where R = alkyl) RNH 2 e. g. CH 3 CH 2 -NH 2 (i) Lower members soluble in water but a very fishy smell! test with red litmus and conc. HCl(aq) fumes. (ii) If a suspected salt of an amine, then add sodium hydroxide solution to free the amine. Aromatic amines(where R = aryl with the amine or amino group directly attached to an aromatic ring) RNH 2 e. g. C 6 H 5 -NH 2 (i) Dissolve the primary aromatic amine in dilute hydrochloric acid at 5 o. C and mix with sodium nitrite solution. (ii) Add a phenol dissolved in dilute sodium hydroxide. (i) A fishy odour, litmus turns blue, white clouds with HCl. (i) Unless its a liquid or solid, only the more fishy odour distinguishes it from ammonia. (i) It should be a clear solution with few, if any, brown fumes. (i) If a primary aromatic amine, a 'stable' diazonium salt is formed. Diazonium salts from aliphatic amines decompose rapidly evolving colourless nitrogen. (ii) The reaction is e. g. (ii) The above is not R-NH 3+ + OH- ==> R-NH 2 + H 2 O observed until after adding the alkali. (ii) A coloured (ii) An azo dyestuff molecule is formed in a precipitate [red - coupling reaction e. g. brown - yellow etc. ] C 6 H 5 -N=N-C 6 H 4 -OH
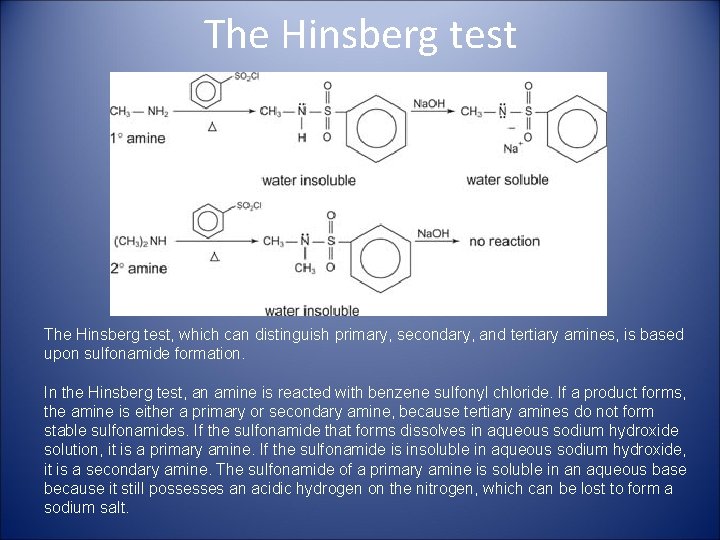
The Hinsberg test, which can distinguish primary, secondary, and tertiary amines, is based upon sulfonamide formation. In the Hinsberg test, an amine is reacted with benzene sulfonyl chloride. If a product forms, the amine is either a primary or secondary amine, because tertiary amines do not form stable sulfonamides. If the sulfonamide that forms dissolves in aqueous sodium hydroxide solution, it is a primary amine. If the sulfonamide is insoluble in aqueous sodium hydroxide, it is a secondary amine. The sulfonamide of a primary amine is soluble in an aqueous base because it still possesses an acidic hydrogen on the nitrogen, which can be lost to form a sodium salt.
- Slides: 26