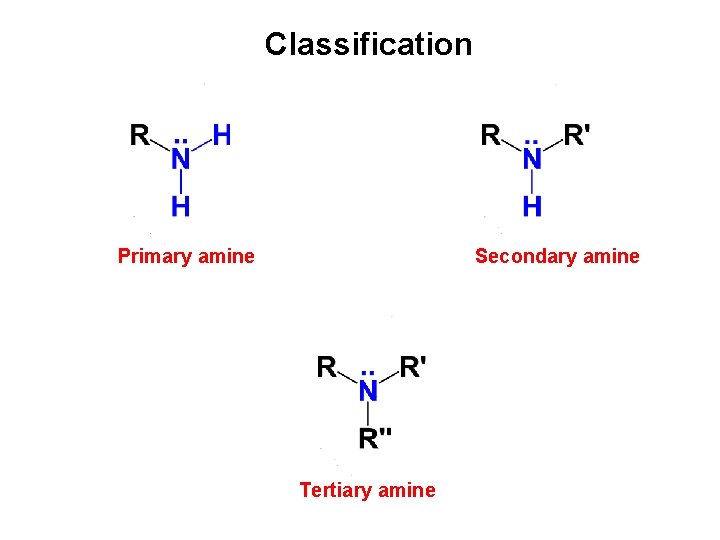
AMINES Classification Primary amine Secondary amine Tertiary amine











- Slides: 52

AMINES

Classification Primary amine Secondary amine Tertiary amine

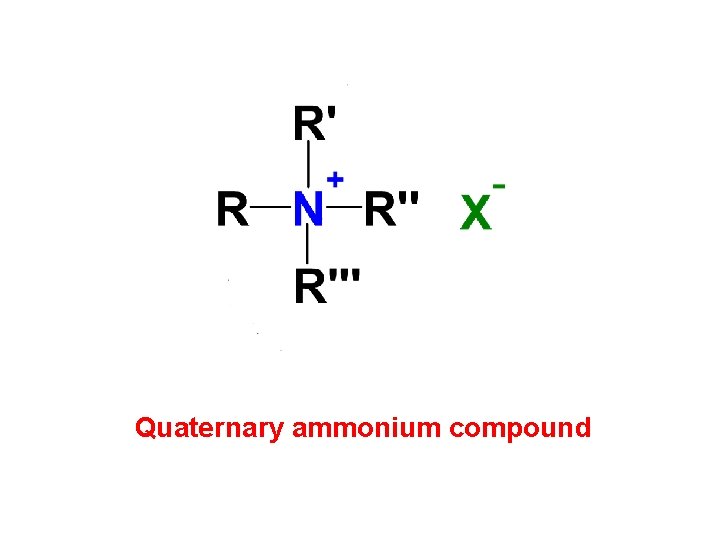
Quaternary ammonium compound

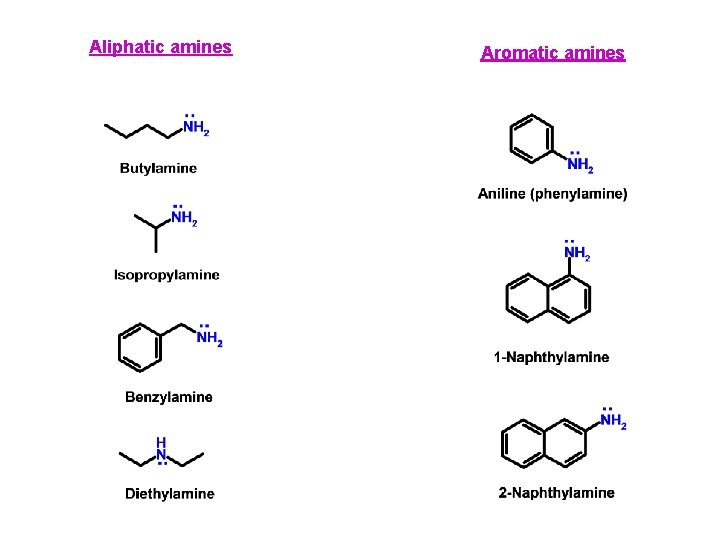
Aliphatic amines Aromatic amines

Examples of nomenclature

Cyclic amines

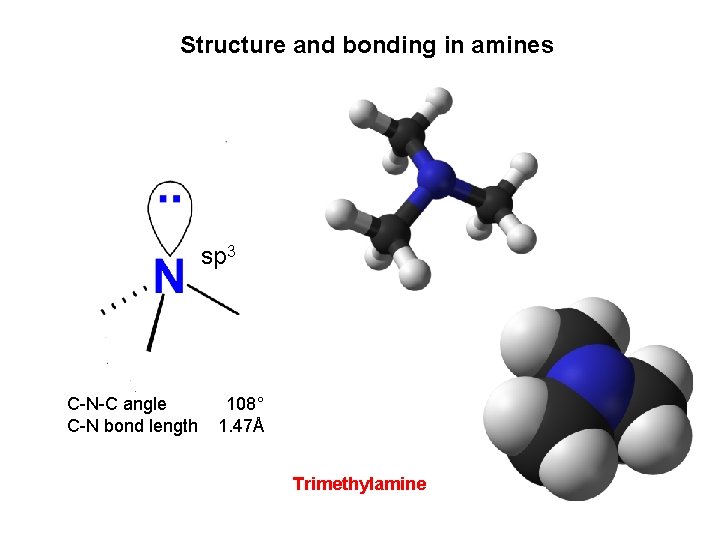
Structure and bonding in amines sp 3 C-N-C angle C-N bond length 108° 1. 47Å Trimethylamine

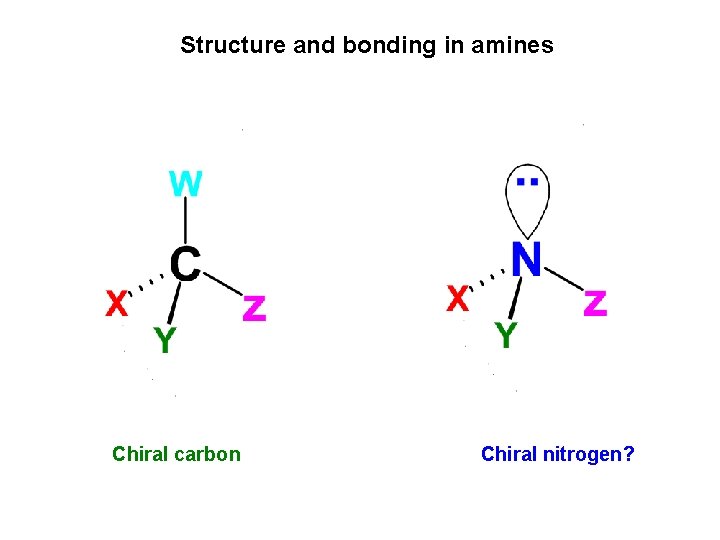
Structure and bonding in amines Chiral carbon Chiral nitrogen?

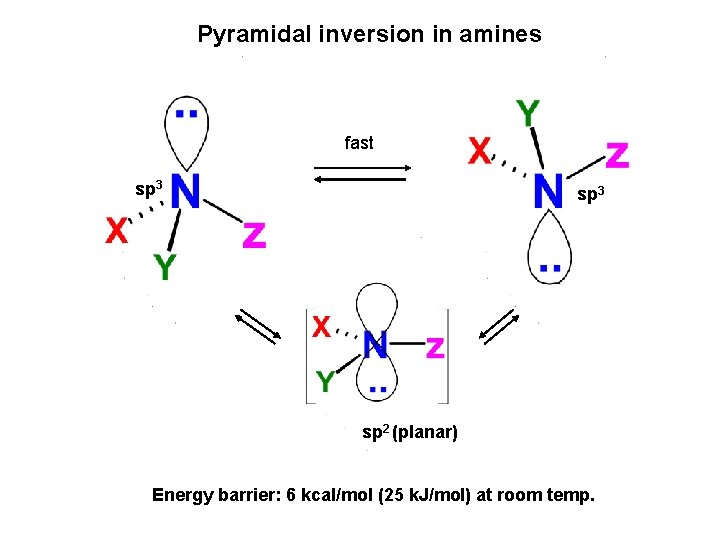
Pyramidal inversion in amines fast sp 3 sp 2 (planar) Energy barrier: 6 kcal/mol (25 k. J/mol) at room temp.

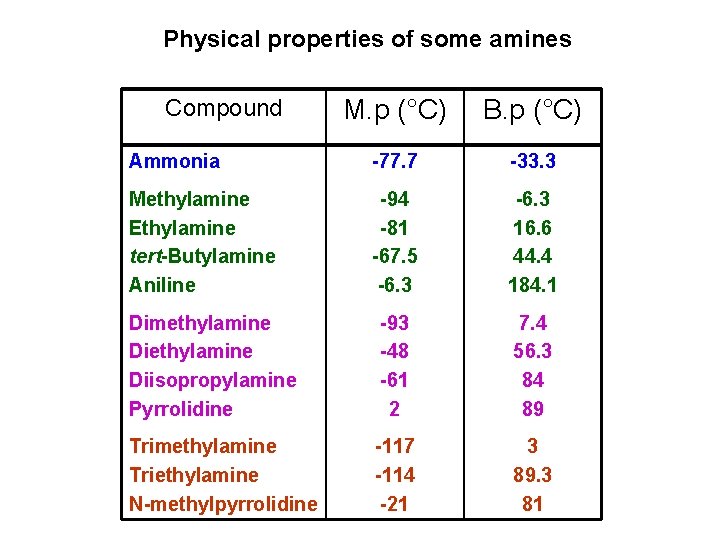
Physical properties of some amines Compound M. p (°C) B. p (°C) Ammonia -77. 7 -33. 3 Methylamine Ethylamine tert-Butylamine Aniline -94 -81 -67. 5 -6. 3 16. 6 44. 4 184. 1 Dimethylamine Diisopropylamine Pyrrolidine -93 -48 -61 2 7. 4 56. 3 84 89 Trimethylamine Triethylamine N-methylpyrrolidine -117 -114 -21 3 89. 3 81

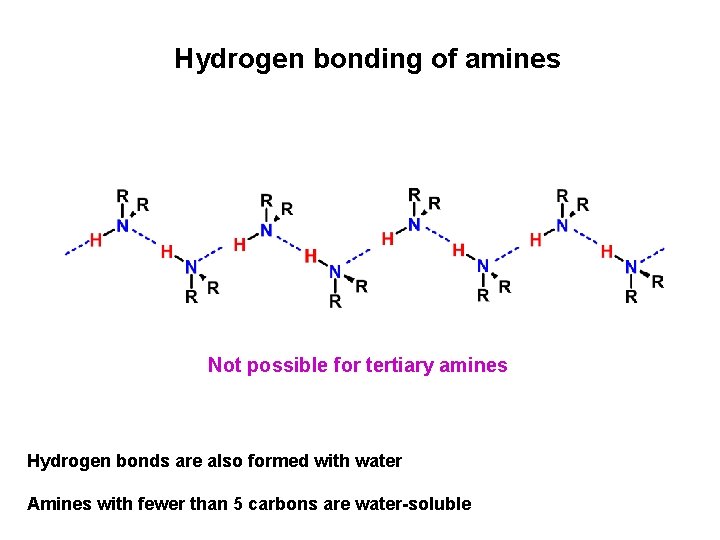
Hydrogen bonding of amines Not possible for tertiary amines Hydrogen bonds are also formed with water Amines with fewer than 5 carbons are water-soluble

Basicity of amines Because of nitrogen lone pair amines are both basic and nucleophilic An amine (a Lewis base) An acid A salt Amines are much more basic than alcohols, ethers, or water.

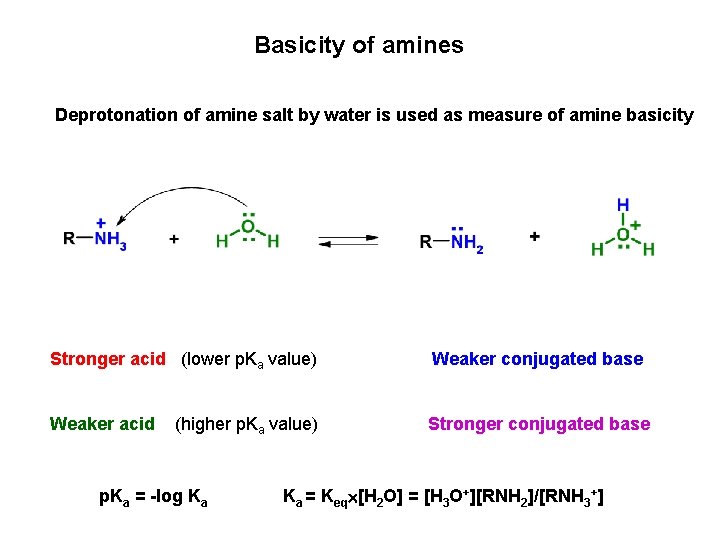
Basicity of amines Deprotonation of amine salt by water is used as measure of amine basicity Stronger acid (lower p. Ka value) Weaker conjugated base Weaker acid Stronger conjugated base (higher p. Ka value) p. Ka = -log Ka Ka = Keq [H 2 O] = [H 3 O+][RNH 2]/[RNH 3+]

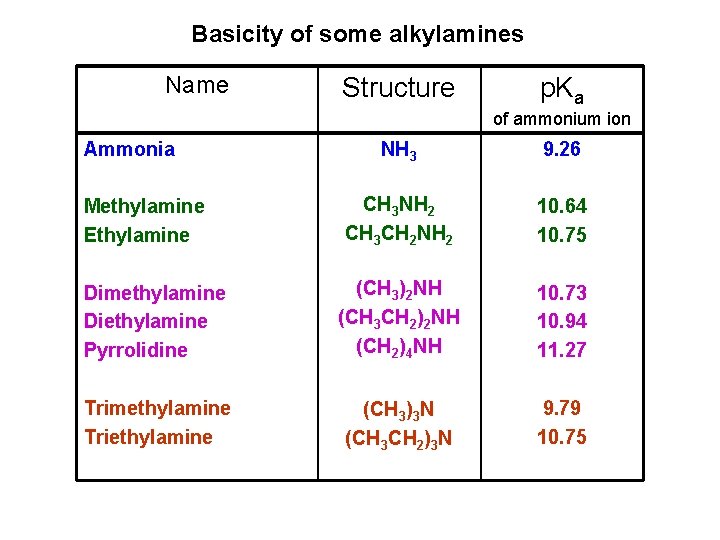
Basicity of some alkylamines Name Structure p. Ka of ammonium ion Ammonia NH 3 9. 26 CH 3 NH 2 CH 3 CH 2 NH 2 10. 64 10. 75 Dimethylamine Diethylamine Pyrrolidine (CH 3)2 NH (CH 3 CH 2)2 NH (CH 2)4 NH 10. 73 10. 94 11. 27 Trimethylamine Triethylamine (CH 3)3 N (CH 3 CH 2)3 N 9. 79 10. 75 Methylamine Ethylamine

Note the diference in basicity of amine and amide nitrogen Amides are completely nonbasic Resonance stabilization of amide

After amide protonation No resonance stabilization Inductive destabilization of the positive charge

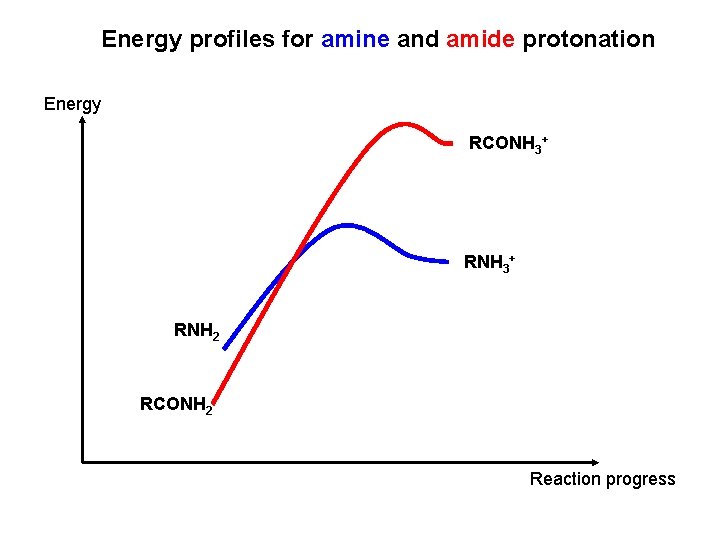
Energy profiles for amine and amide protonation Energy RCONH 3+ RNH 2 RCONH 2 Reaction progress

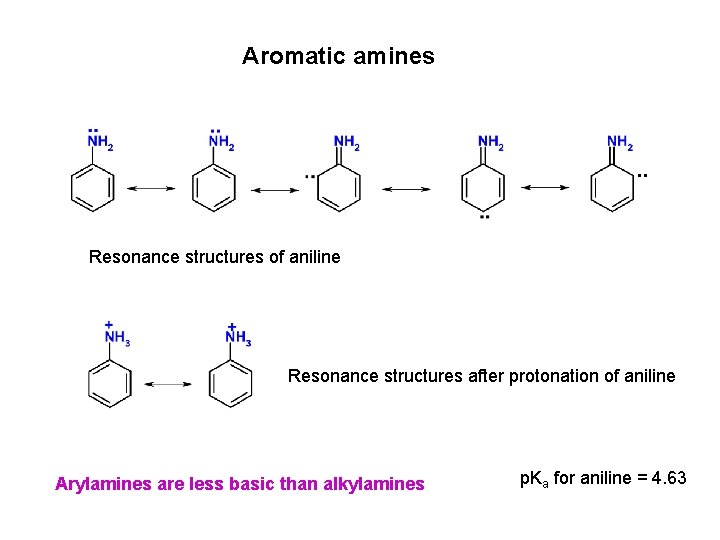
Aromatic amines Resonance structures of aniline Resonance structures after protonation of aniline Arylamines are less basic than alkylamines p. Ka for aniline = 4. 63

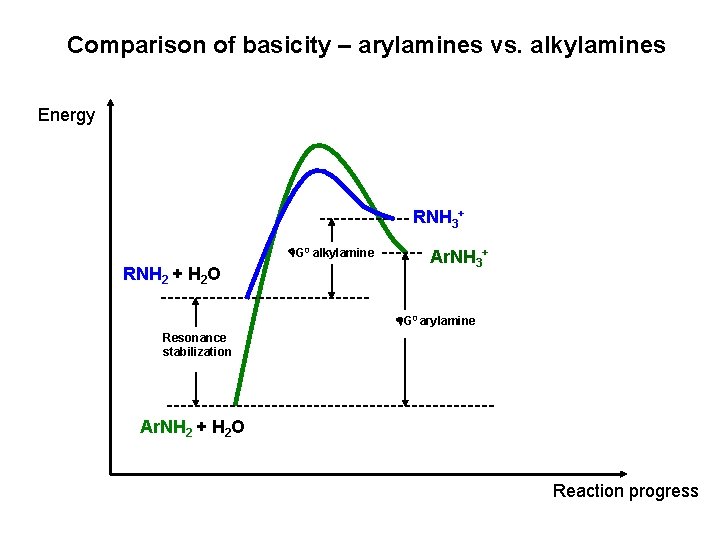
Comparison of basicity – arylamines vs. alkylamines Energy RNH 3+ G 0 alkylamine RNH 2 + H 2 O Ar. NH 3+ G 0 arylamine Resonance stabilization Ar. NH 2 + H 2 O Reaction progress

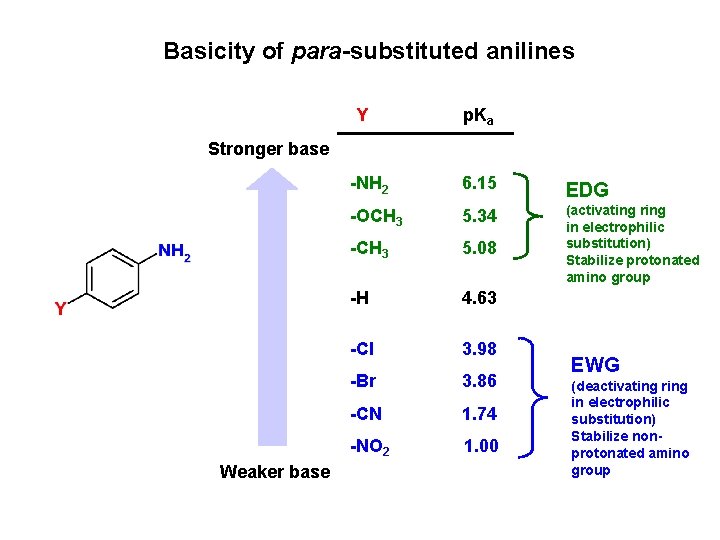
Basicity of para-substituted anilines Y p. Ka Stronger base Weaker base -NH 2 6. 15 EDG -OCH 3 5. 34 -CH 3 5. 08 (activating ring in electrophilic substitution) Stabilize protonated amino group -H 4. 63 -Cl 3. 98 -Br 3. 86 -CN 1. 74 -NO 2 1. 00 EWG (deactivating ring in electrophilic substitution) Stabilize nonprotonated amino group

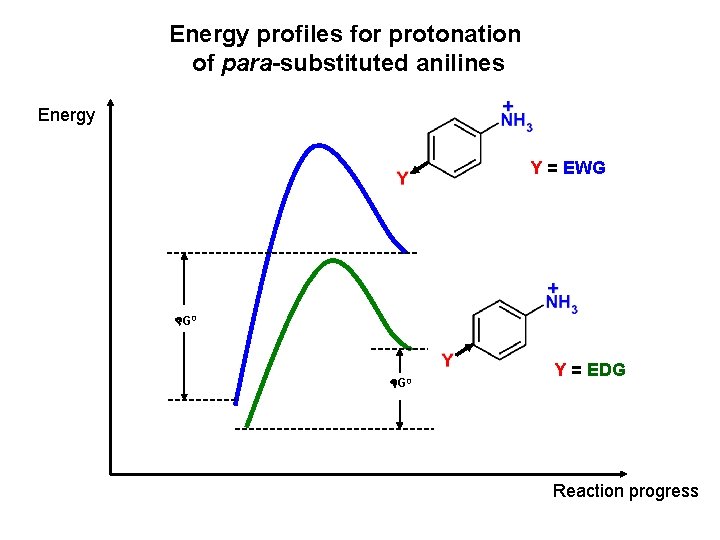
Energy profiles for protonation of para-substituted anilines Energy Y = EWG G 0 Y = EDG Reaction progress

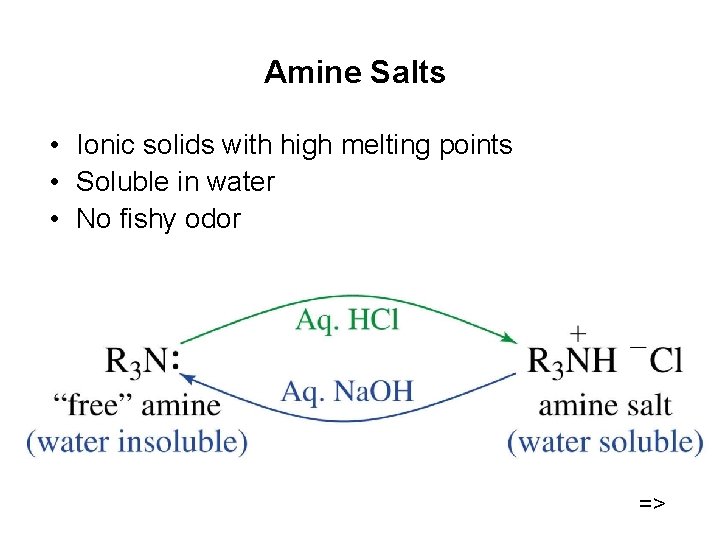
Amine Salts • Ionic solids with high melting points • Soluble in water • No fishy odor =>

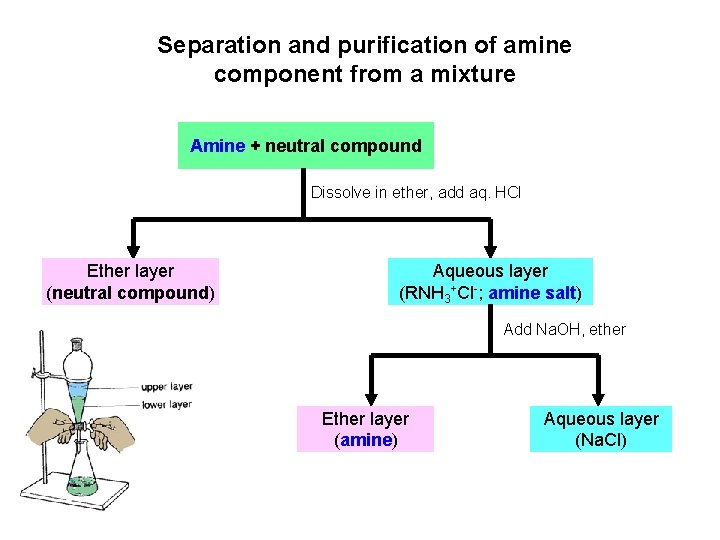
Separation and purification of amine component from a mixture Amine + neutral compound Dissolve in ether, add aq. HCl Ether layer (neutral compound) Aqueous layer (RNH 3+Cl-; amine salt) Add Na. OH, ether Ether layer (amine) Aqueous layer (Na. Cl)

Preparation of amines SN 2 reaction of alkyl halides

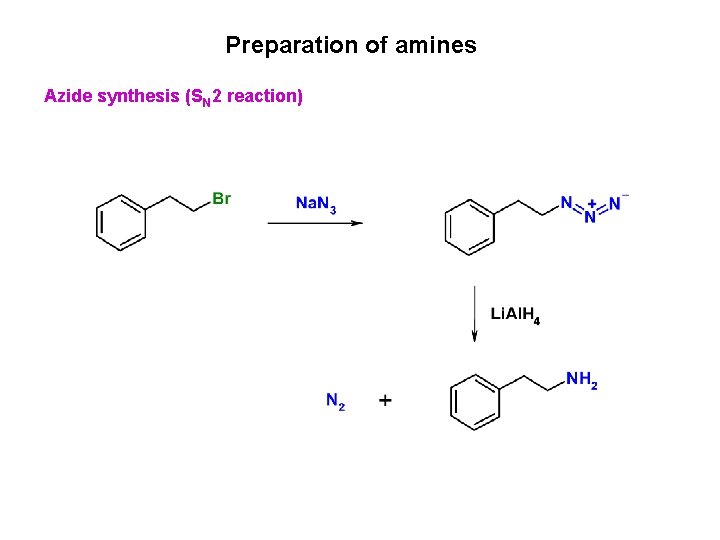
Preparation of amines Azide synthesis (SN 2 reaction)

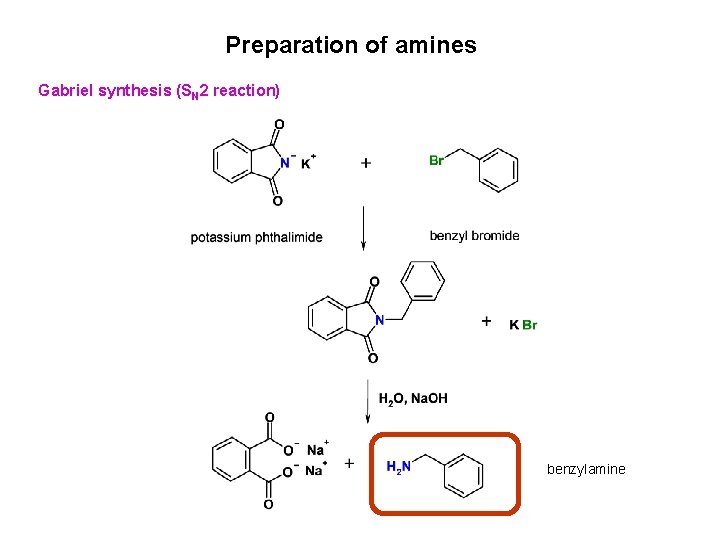
Preparation of amines Gabriel synthesis (SN 2 reaction) benzylamine

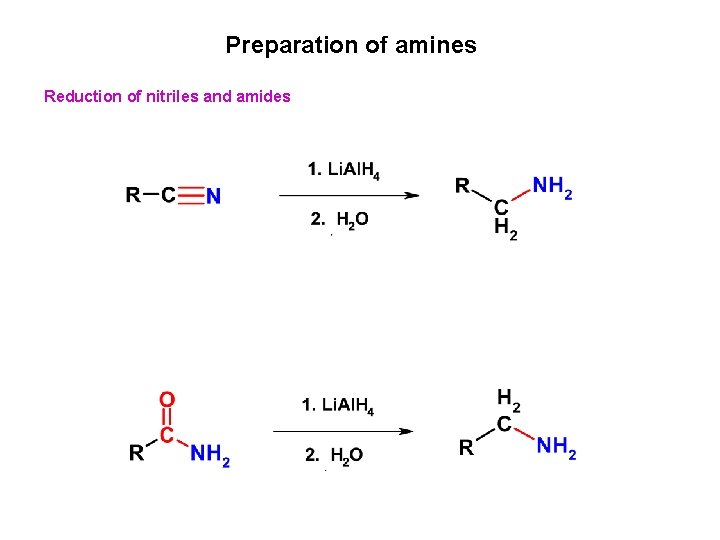
Preparation of amines Reduction of nitriles and amides

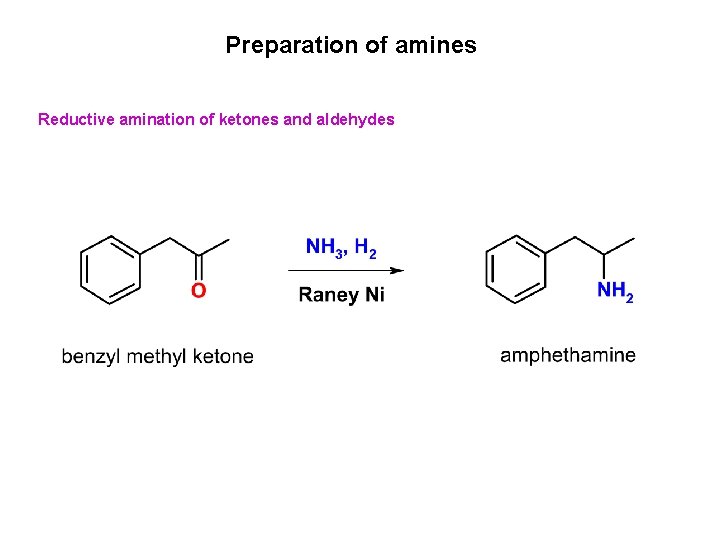
Preparation of amines Reductive amination of ketones and aldehydes

Preparation of amines Reductive amination of ketones and aldehydes Primary amine Secondary amine Tertiary amine

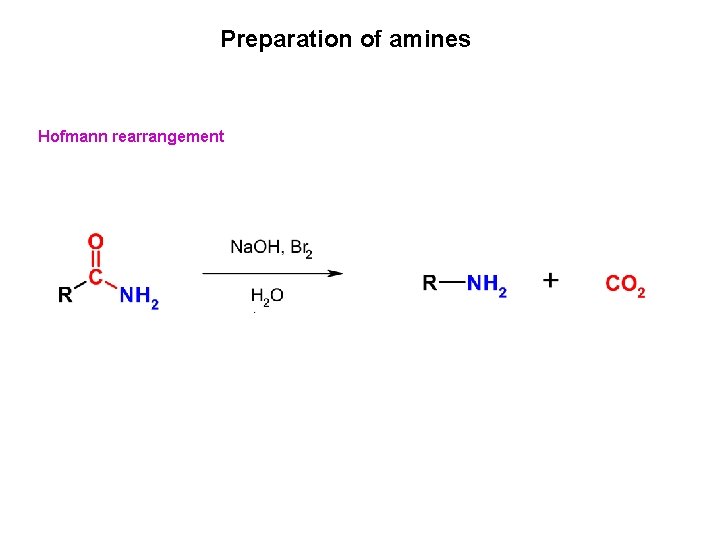
Preparation of amines Hofmann rearrangement

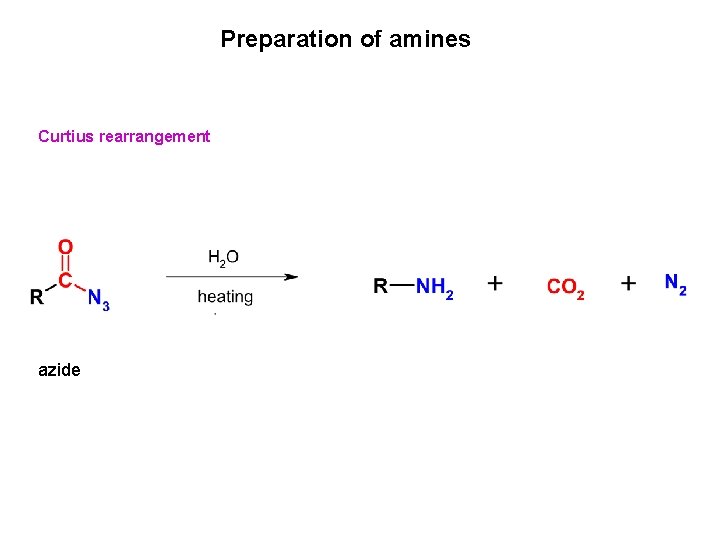
Preparation of amines Curtius rearrangement azide

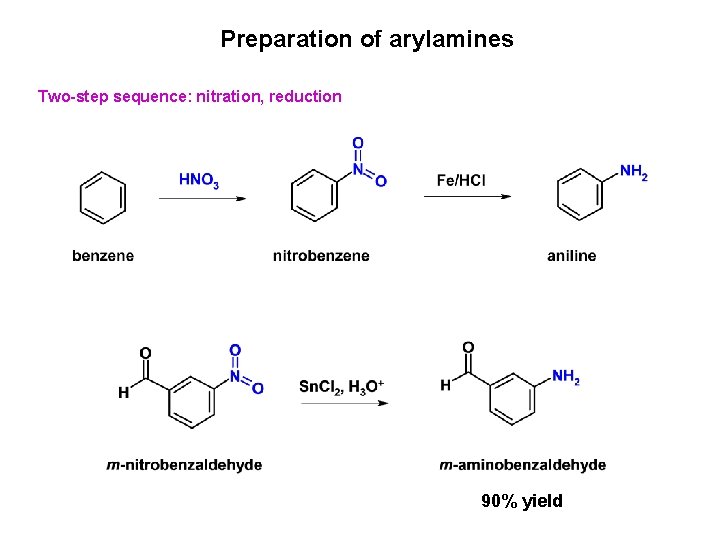
Preparation of arylamines Two-step sequence: nitration, reduction 90% yield

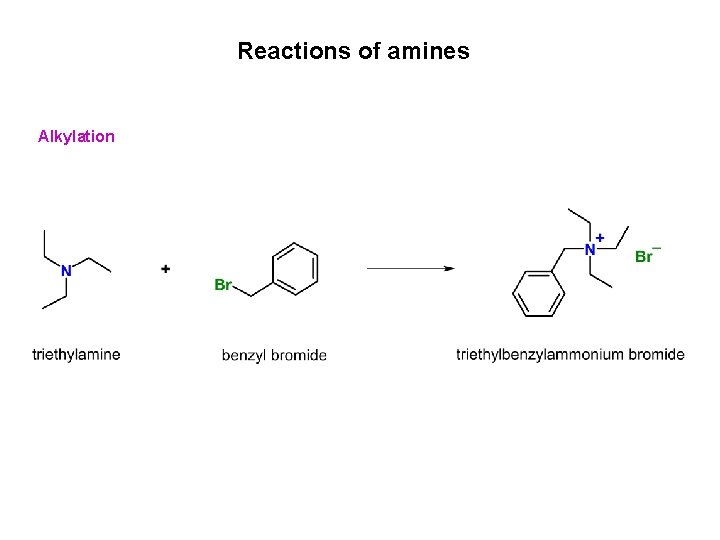
Reactions of amines Alkylation

Reactions of amines Acylation (with acid chlorides, anhydrides or esters) Tertiary amines do not react with carboxylic acids derivatives

Reactions of amines Hofmann elimination

Reactions of amines Reaction of alkylamines with nitrous acid Unstable, decomposes immediately after formation with evolution of nitrogen gas

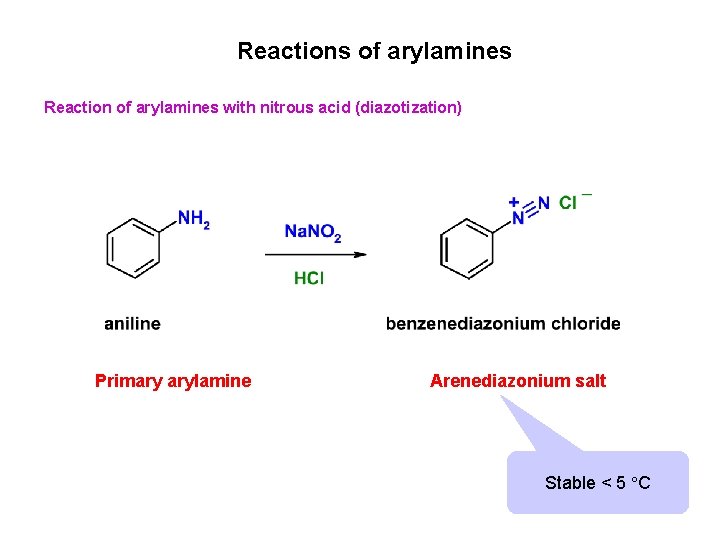
Reactions of arylamines Reaction of arylamines with nitrous acid (diazotization) Primary arylamine Arenediazonium salt Stable < 5 C

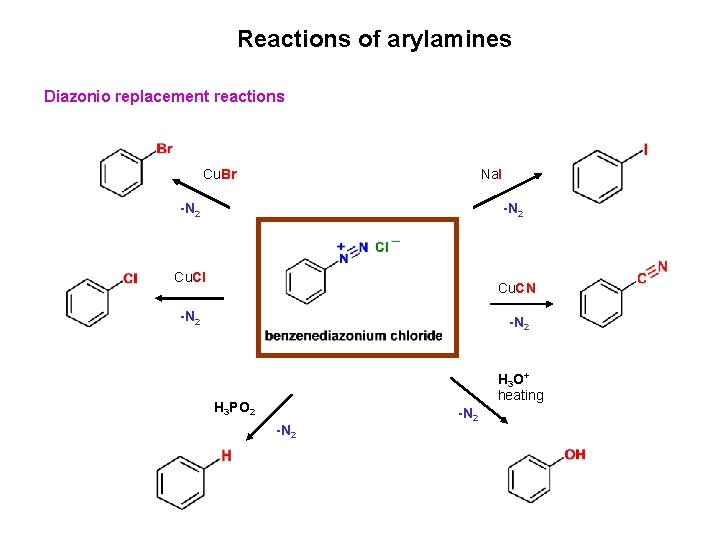
Reactions of arylamines Diazonio replacement reactions Cu. Br Na. I -N 2 Cu. Cl Cu. CN -N 2 H 3 O+ heating H 3 PO 2 -N 2

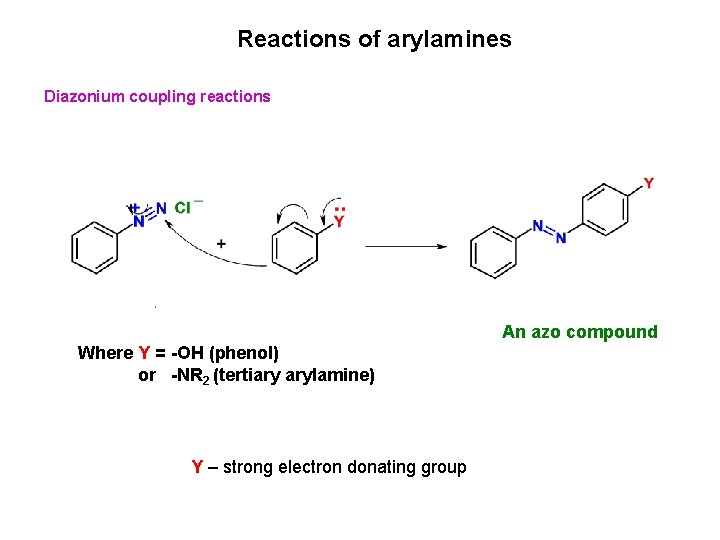
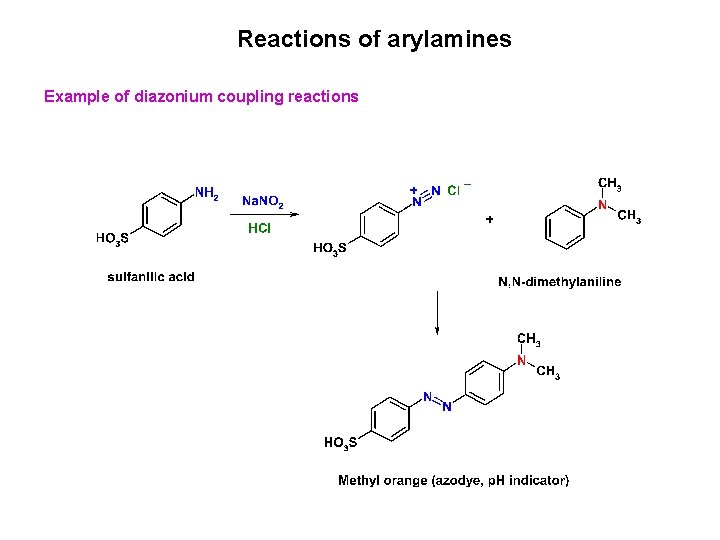
Reactions of arylamines Diazonium coupling reactions An azo compound Where Y = -OH (phenol) or -NR 2 (tertiary arylamine) Y – strong electron donating group

Reactions of arylamines Example of diazonium coupling reactions

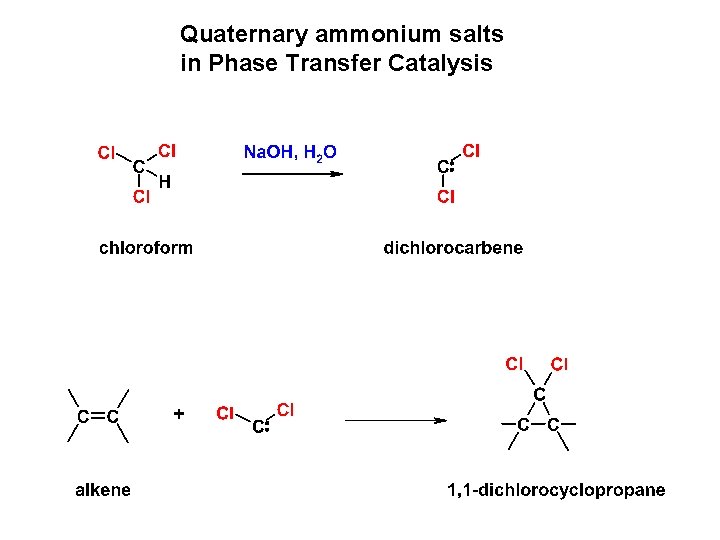
Quaternary ammonium salts in Phase Transfer Catalysis

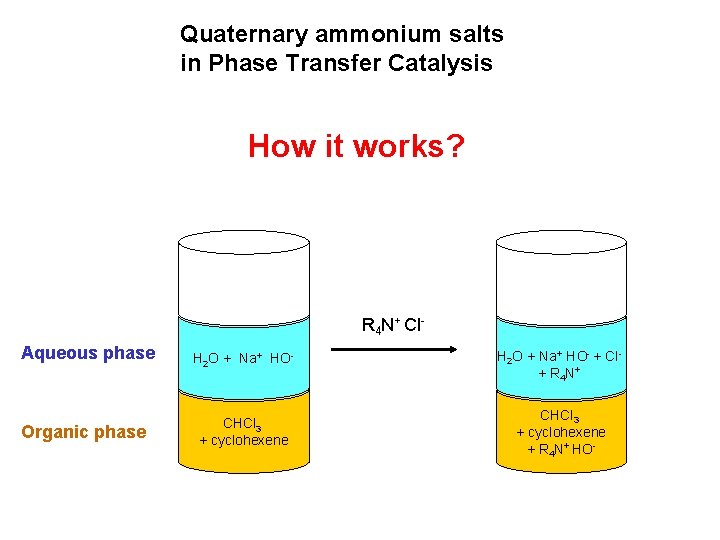
Quaternary ammonium salts in Phase Transfer Catalysis How it works? R 4 N+ Cl. Aqueous phase Organic phase H 2 O + Na+ HO- + Cl+ R 4 N+ CHCl 3 + cyclohexene + R 4 N+ HO-

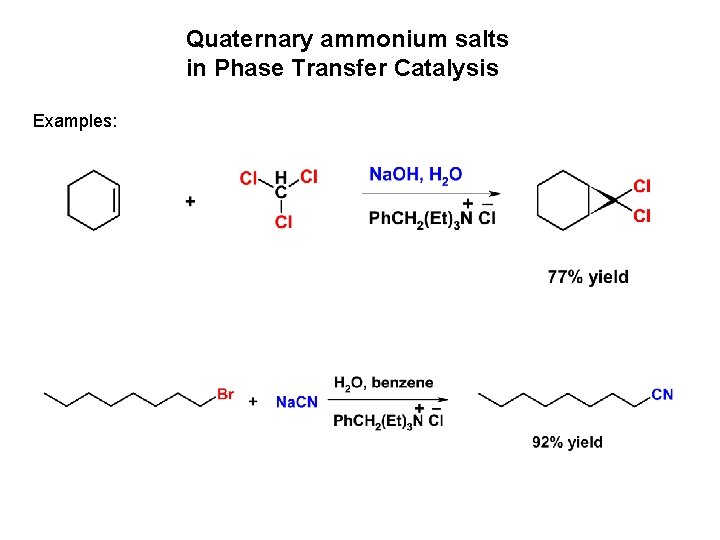
Quaternary ammonium salts in Phase Transfer Catalysis Examples:

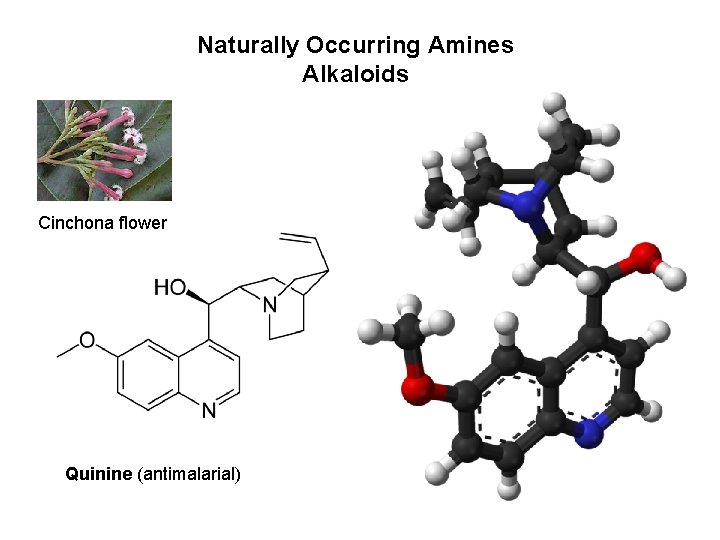
Naturally Occurring Amines Alkaloids Cinchona flower Quinine (antimalarial)

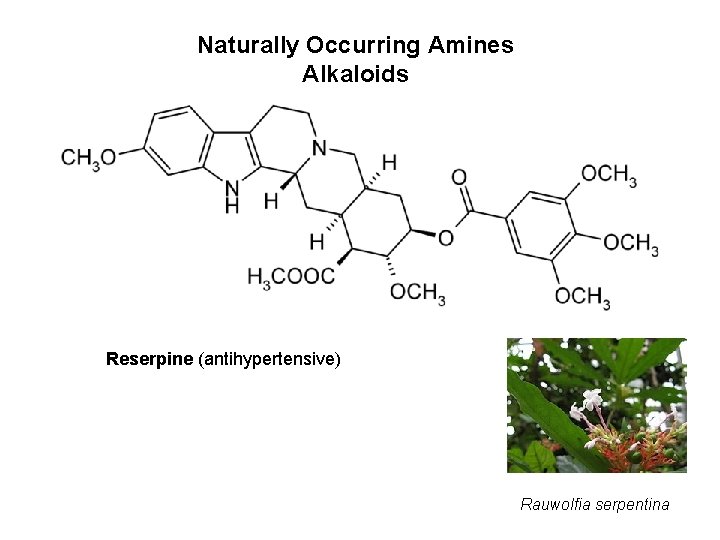
Naturally Occurring Amines Alkaloids Reserpine (antihypertensive) Rauwolfia serpentina

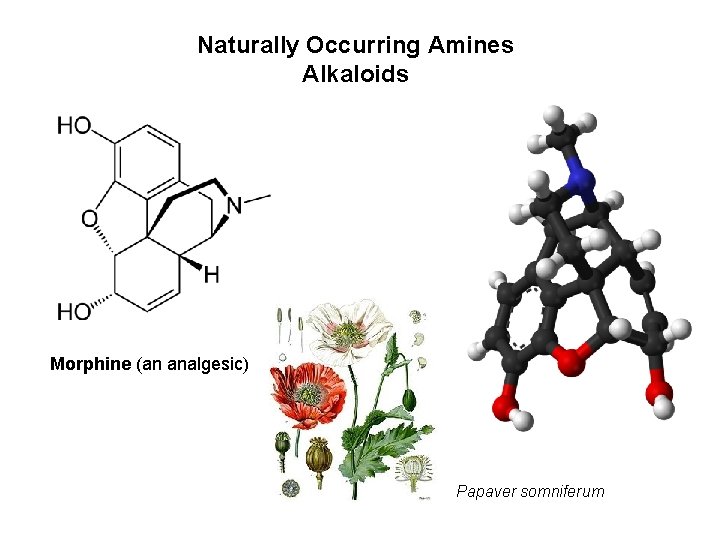
Naturally Occurring Amines Alkaloids Morphine (an analgesic) Papaver somniferum

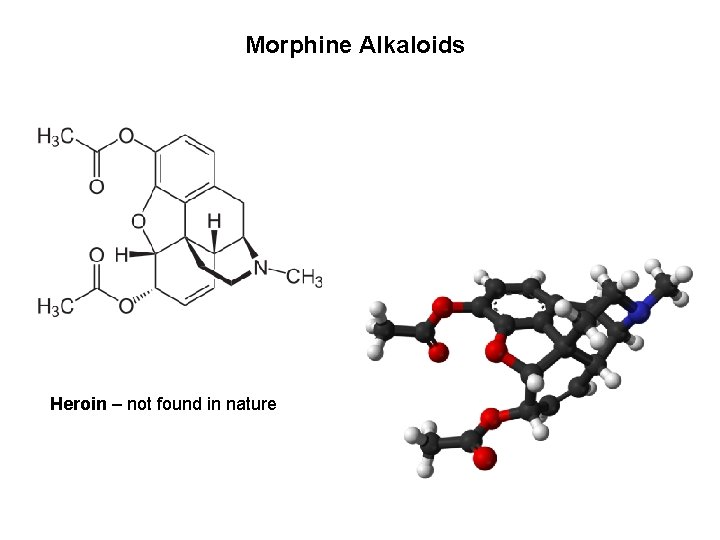
Morphine Alkaloids Heroin – not found in nature

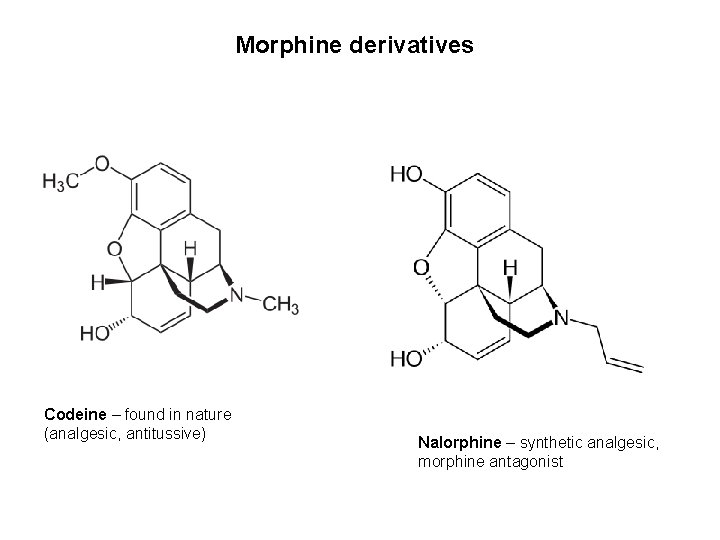
Morphine derivatives Codeine – found in nature (analgesic, antitussive) Nalorphine – synthetic analgesic, morphine antagonist

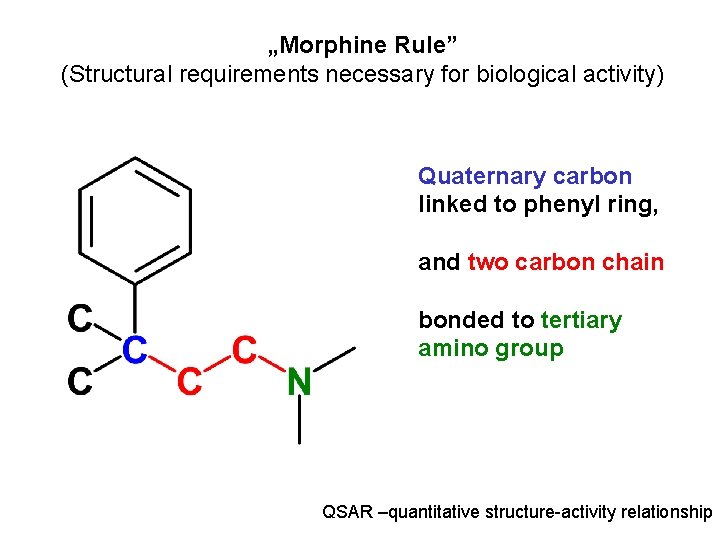
„Morphine Rule” (Structural requirements necessary for biological activity) Quaternary carbon linked to phenyl ring, and two carbon chain bonded to tertiary amino group QSAR –quantitative structure-activity relationship

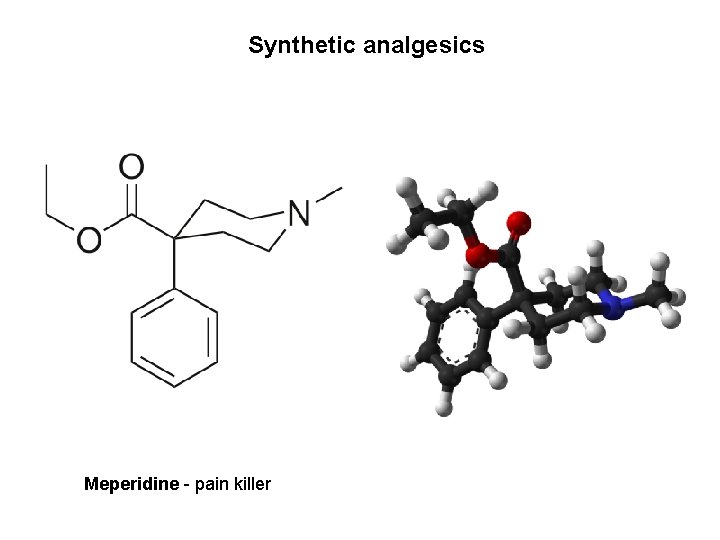
Synthetic analgesics Meperidine - pain killer

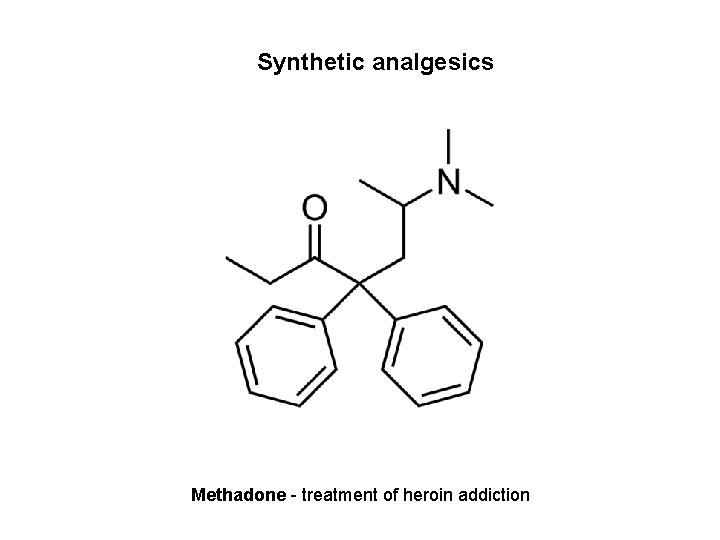
Synthetic analgesics Methadone - treatment of heroin addiction

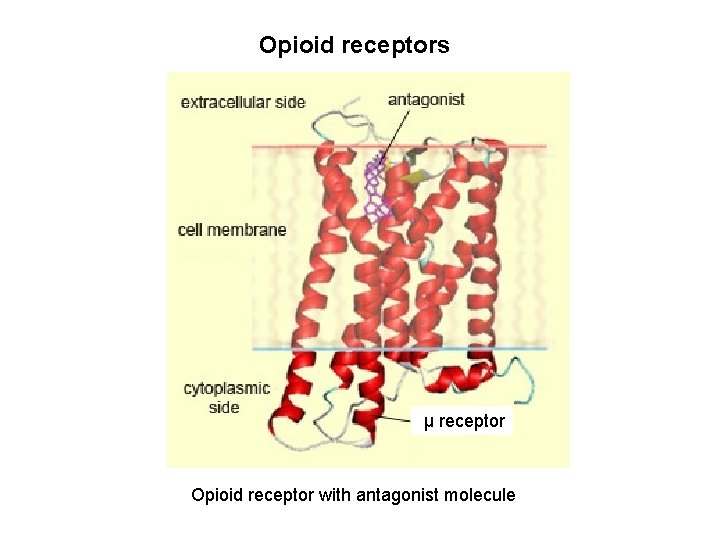
Opioid receptors μ receptor Opioid receptor with antagonist molecule