Chapter 13 Amines t Nomenclature Primary amines are










- Slides: 20

Chapter 13 Amines

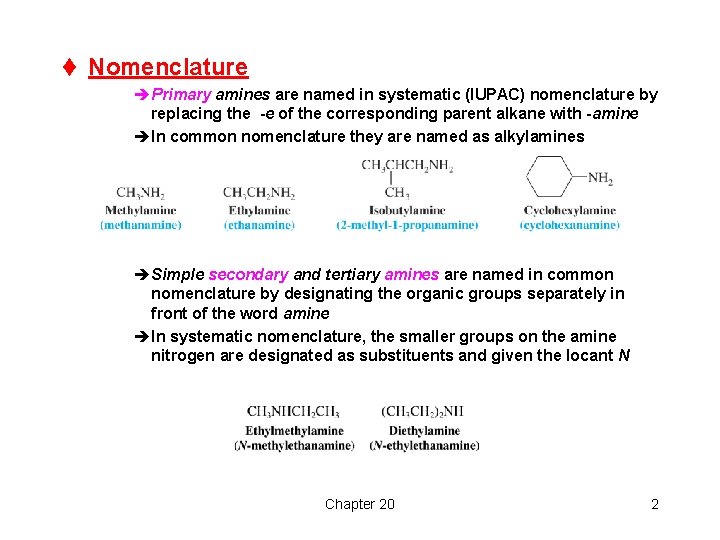
t Nomenclature èPrimary amines are named in systematic (IUPAC) nomenclature by replacing the -e of the corresponding parent alkane with -amine èIn common nomenclature they are named as alkylamines èSimple secondary and tertiary amines are named in common nomenclature by designating the organic groups separately in front of the word amine èIn systematic nomenclature, the smaller groups on the amine nitrogen are designated as substituents and given the locant N Chapter 20 2

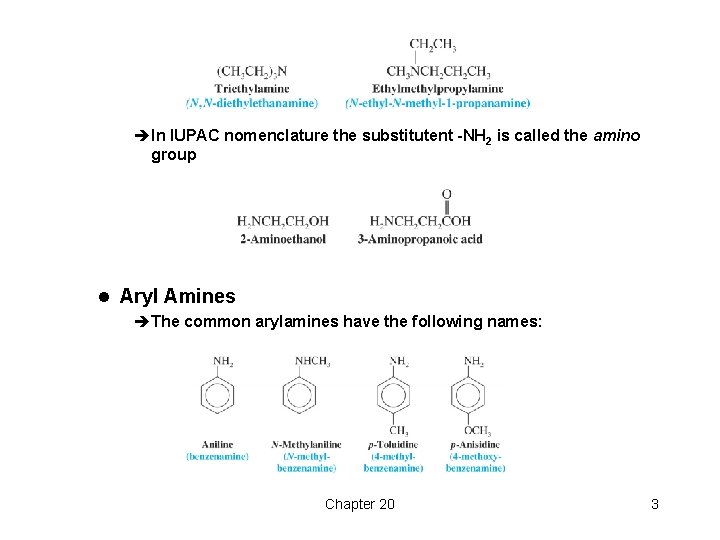
èIn IUPAC nomenclature the substitutent -NH 2 is called the amino group l Aryl Amines èThe common arylamines have the following names: Chapter 20 3

t Physical Properties and Structure of Amines èPrimary and secondary amines can form hydrogen bonds to each other and water èTertiary amines cannot form hydrogen bonds to each other but can form hydrogen bonds to hydrogen bond donors such as water èTertiary amines have lower boiling points than primary or secondary amines of comparable molecular weights èLow molecular weight amines tend to be water soluble whether they are primary, secondary or tertiary Chapter 20 4

t Basicity of Amines: Amine Salts èAmines are weak bases èPrimary alkyl amines are more basic than ammonia H An alkyl group helps to stabilize the alkylaminium ion resulting from protonation of the amine Chapter 20 5

èIn the gas phase, basicity in the family of methylamines increases with increasing methyl substitution H More alkyl substitution results in more stabilization of the alkylaminium ion èIn aqueous solution, trimethylamine is less basic than dimethyl- or methylamine An alkylaminium ion in water is solvated and stabilized by hydrogen bonding of its hydrogens with water H The trimethylaminium ion has only one hydrogen with which to hydrogen bond to water H The trimethylaminium ion is solvated less well (and therefore stabilized less) than the dimethylaminium ion, which has two hydrogen atoms for hydrogen bonding H Chapter 20 6

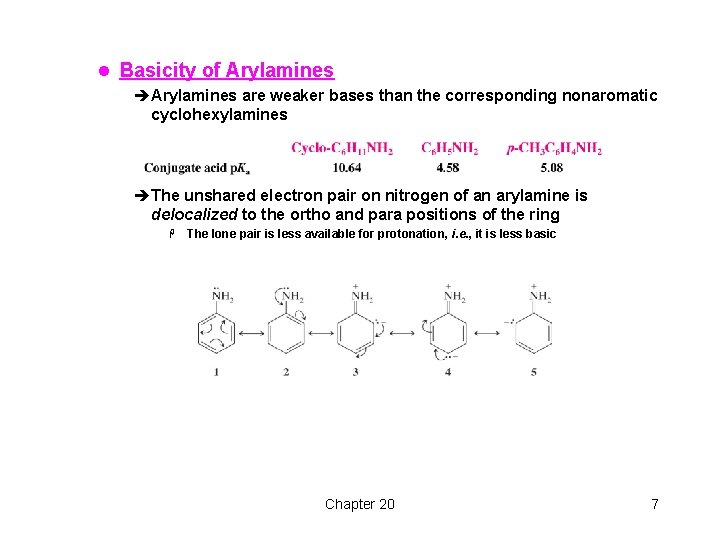
l Basicity of Arylamines èArylamines are weaker bases than the corresponding nonaromatic cyclohexylamines èThe unshared electron pair on nitrogen of an arylamine is delocalized to the ortho and para positions of the ring H The lone pair is less available for protonation, i. e. , it is less basic Chapter 20 7

l Solubility of Amines in Aqueous Acid èMany aminium chlorides, bromides, iodides and sulfates are water soluble H Amines which are not soluble in water will often dissolve in dilute aqueous acid èSolubility of amines in dilute acid can be used as a chemical test to distinguish amines from compounds that are not basic èWater-insoluble amines can be separated from water-insoluble neutral or acidic compounds by virtue of the amine’s solubility in aqueous acid The amine is extracted into aqueous acid H The amine is recovered by making the solution basic and extracting the amine into an organic solvent H èAmides are not basic and are not soluble in aqueous acids Chapter 20 8

t Preparation of Amines 1 - By Nucleophilic Substitution Reactions èAlkylation of Ammonia Reaction of ammonia with an alkyl halide leads to an aminium salt H The salt is treated with base to give the primary amine H The method is limited because multiple alkylations usually occur H H Using an excess of ammonia helps to minimize multiple alkylations Chapter 20 9

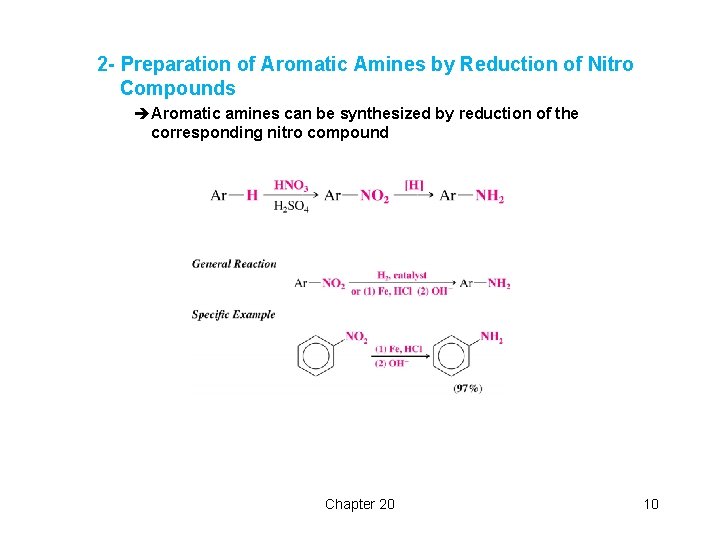
2 - Preparation of Aromatic Amines by Reduction of Nitro Compounds èAromatic amines can be synthesized by reduction of the corresponding nitro compound Chapter 20 10

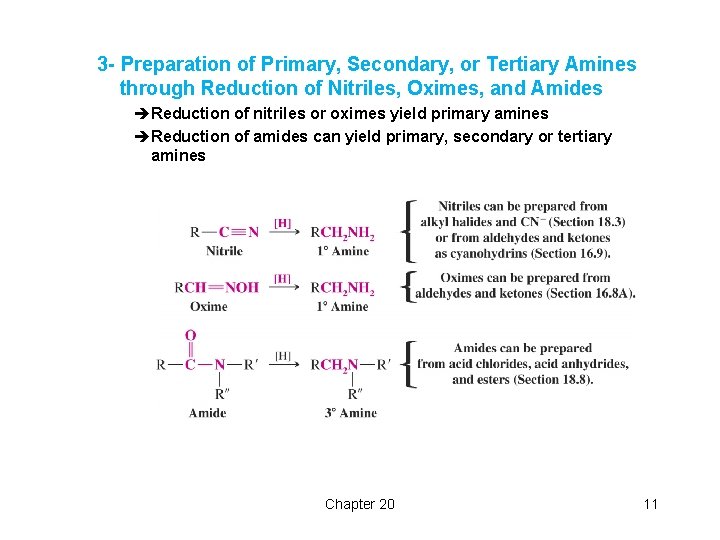
3 - Preparation of Primary, Secondary, or Tertiary Amines through Reduction of Nitriles, Oximes, and Amides èReduction of nitriles or oximes yield primary amines èReduction of amides can yield primary, secondary or tertiary amines Chapter 20 11

4 -Reactions of Amines èThe lone pair of the amine nitrogen atom accounts for most chemistry of amines H The unshared electron pair can act as a base or as a nucleophile Chapter 20 12

5 - Reactions of Amines with Nitrous Acid èNitrous acid (HONO) is prepared in situ by reaction of sodium nitrite with a strong aqueous acid 6 - Reaction of Primary Aliphatic Amines with Nitrous Acid èPrimary amines undergo diazotization with nitrous acid The unstable diazonium salts decompose to form carbocations H The carbocations react further to give alkenes, alcohols and alkyl halides H Chapter 20 13

7 - Reaction of Primary Arylamines with Nitrous Acid èReaction of primary arylamines with nitrous acid results in the formation of relatively stable arenediazonium salts This reaction occurs through the intermediacy of an N-nitrosoamine H The N-nitrosoamine is converted to a diazonium ion in a series of steps H Chapter 20 14

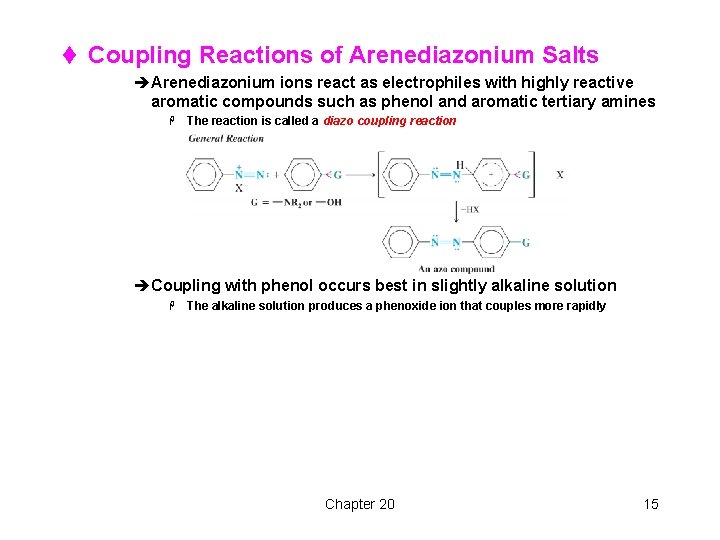
t Coupling Reactions of Arenediazonium Salts èArenediazonium ions react as electrophiles with highly reactive aromatic compounds such as phenol and aromatic tertiary amines H The reaction is called a diazo coupling reaction èCoupling with phenol occurs best in slightly alkaline solution H The alkaline solution produces a phenoxide ion that couples more rapidly Chapter 20 15

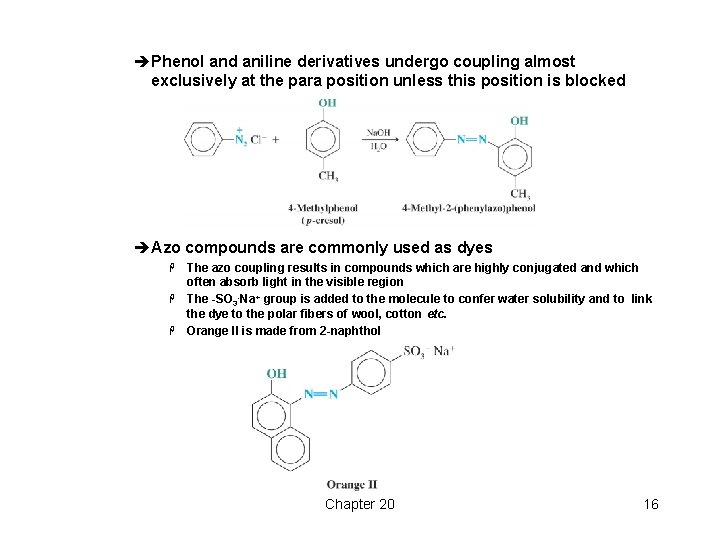
èPhenol and aniline derivatives undergo coupling almost exclusively at the para position unless this position is blocked èAzo compounds are commonly used as dyes The azo coupling results in compounds which are highly conjugated and which often absorb light in the visible region H The -SO 3 -Na+ group is added to the molecule to confer water solubility and to link the dye to the polar fibers of wool, cotton etc. H Orange II is made from 2 -naphthol H Chapter 20 16

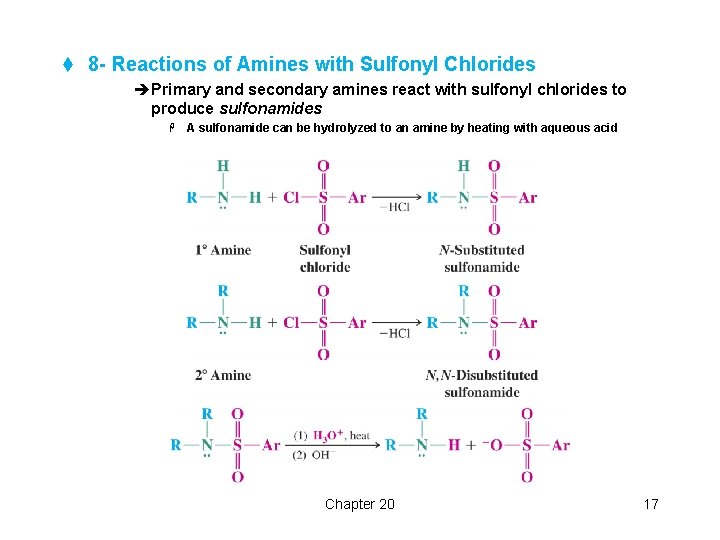
t 8 - Reactions of Amines with Sulfonyl Chlorides èPrimary and secondary amines react with sulfonyl chlorides to produce sulfonamides H A sulfonamide can be hydrolyzed to an amine by heating with aqueous acid Chapter 20 17

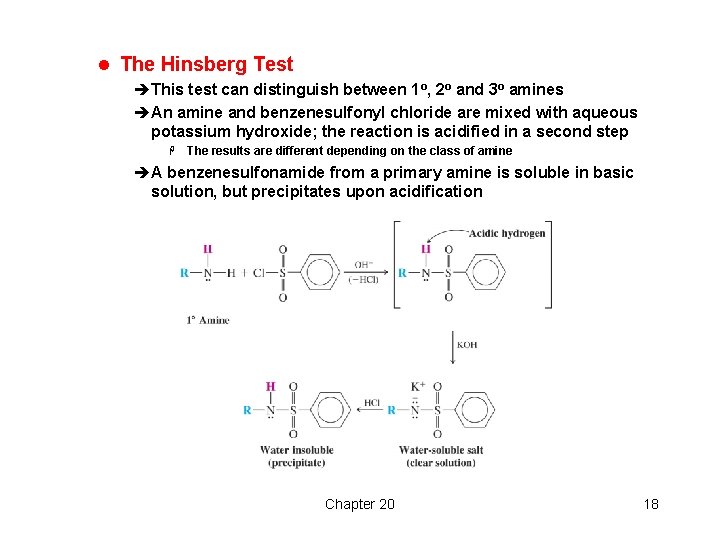
l The Hinsberg Test èThis test can distinguish between 1 o, 2 o and 3 o amines èAn amine and benzenesulfonyl chloride are mixed with aqueous potassium hydroxide; the reaction is acidified in a second step H The results are different depending on the class of amine èA benzenesulfonamide from a primary amine is soluble in basic solution, but precipitates upon acidification Chapter 20 18

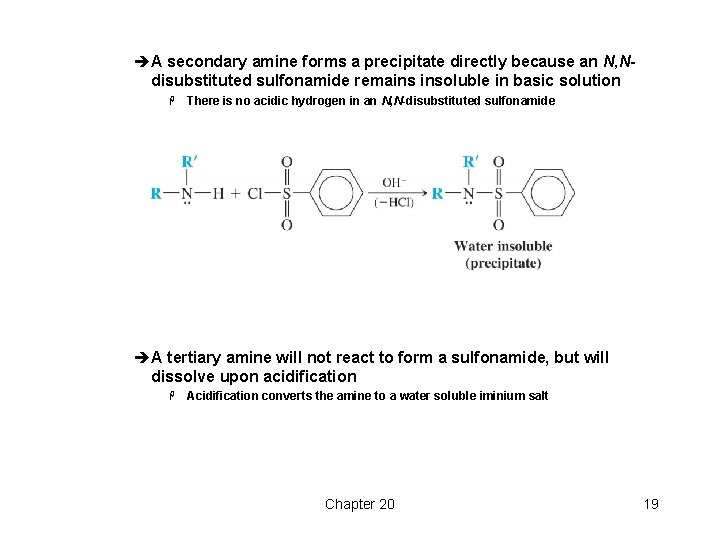
èA secondary amine forms a precipitate directly because an N, Ndisubstituted sulfonamide remains insoluble in basic solution H There is no acidic hydrogen in an N, N-disubstituted sulfonamide èA tertiary amine will not react to form a sulfonamide, but will dissolve upon acidification H Acidification converts the amine to a water soluble iminium salt Chapter 20 19

t Analysis of Amines l Chemical Analysis èAmines can generally be distinguished by their ability to dissolve in dilute aqueous acid èWet litmus paper will indicate the basicity of an amine èThe Hinsberg test can be use to distinguish among primary, secondary and tertiary amines Chapter 20 20