Chapter 23 The Chemistry of Amines Amines 2





- Slides: 30

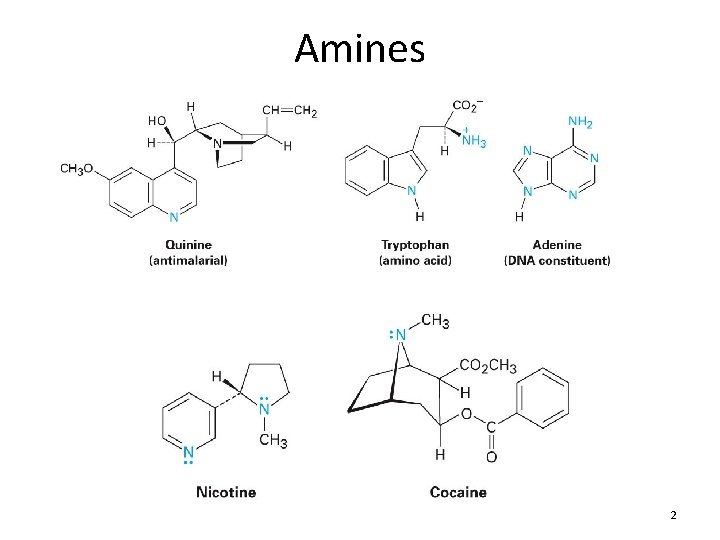
Chapter 23 The Chemistry of Amines

Amines 2

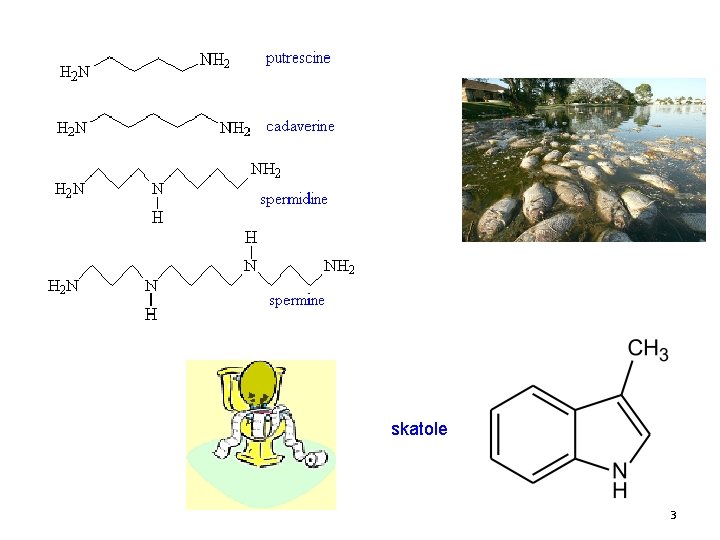
skatole 3

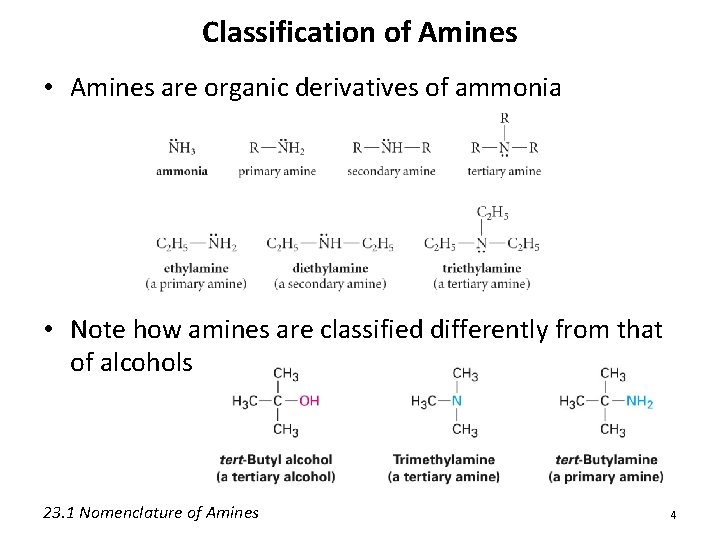
Classification of Amines • Amines are organic derivatives of ammonia • Note how amines are classified differently from that of alcohols 23. 1 Nomenclature of Amines 4

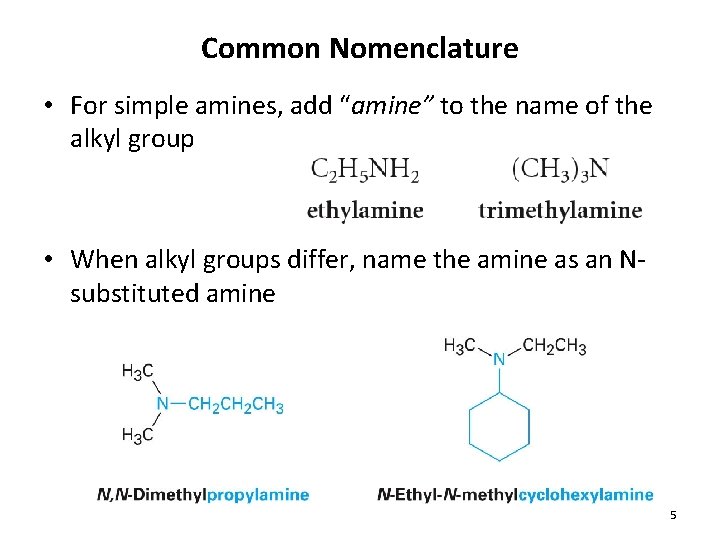
Common Nomenclature • For simple amines, add “amine” to the name of the alkyl group • When alkyl groups differ, name the amine as an Nsubstituted amine 5

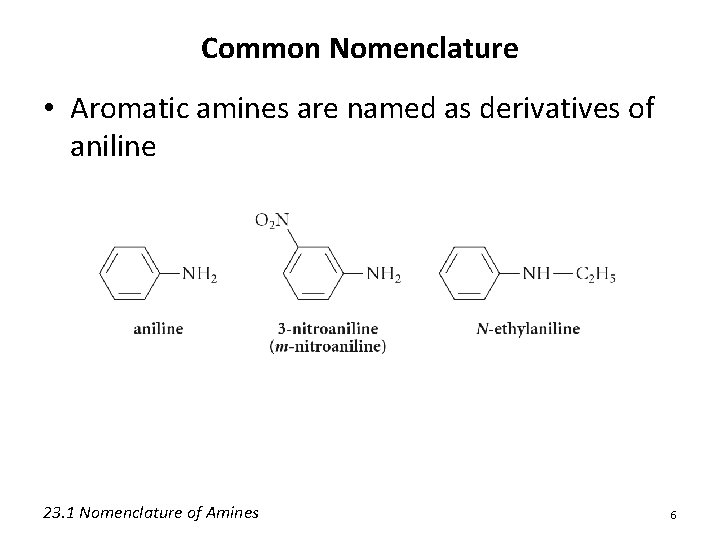
Common Nomenclature • Aromatic amines are named as derivatives of aniline 23. 1 Nomenclature of Amines 6

Substitutive Nomenclature • Amines are named in a similar fashion to their analogous alcohols – Remove the “-e” and replace it with “-amine” • If there is more than one amino group, use Greek prefixes to indicate how many and numbers to indicate location on the alkyl chain 23. 1 Nomenclature of Amines 7

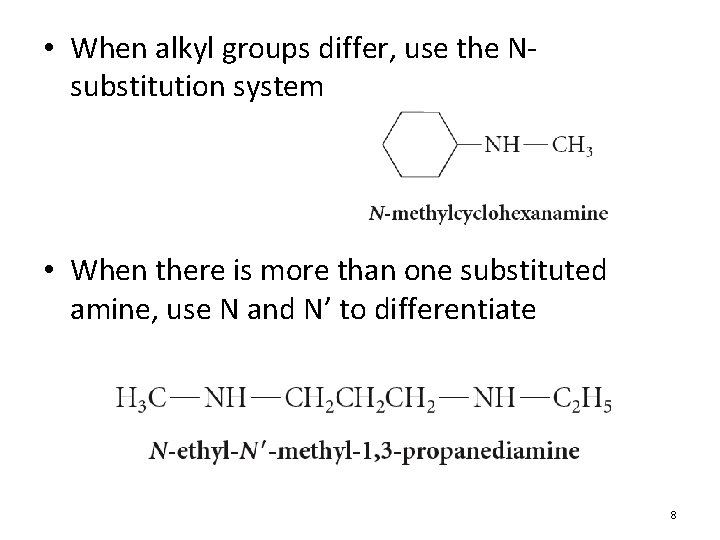
• When alkyl groups differ, use the Nsubstitution system • When there is more than one substituted amine, use N and N’ to differentiate 8

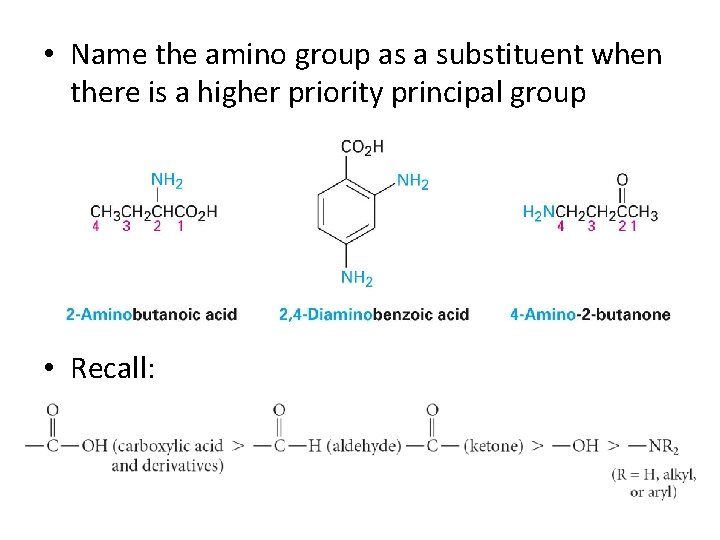
• Name the amino group as a substituent when there is a higher priority principal group • Recall:

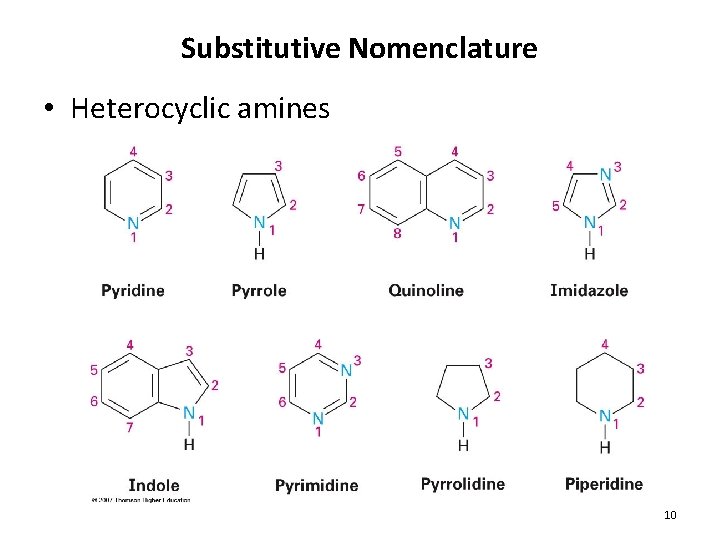
Substitutive Nomenclature • Heterocyclic amines 10

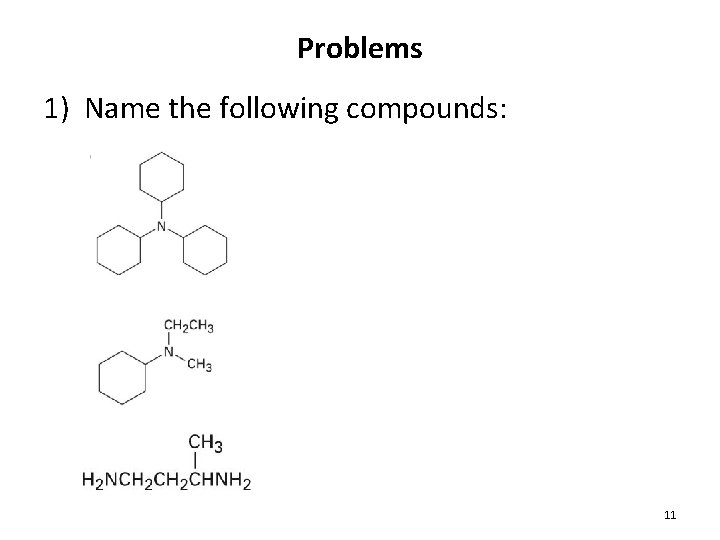
Problems 1) Name the following compounds: 11

2) Draw the following molecules • 2 -propen-1 -amine • N-methyl-N’-propyl-1, 4 -butanediamine • p-aminophenol 12

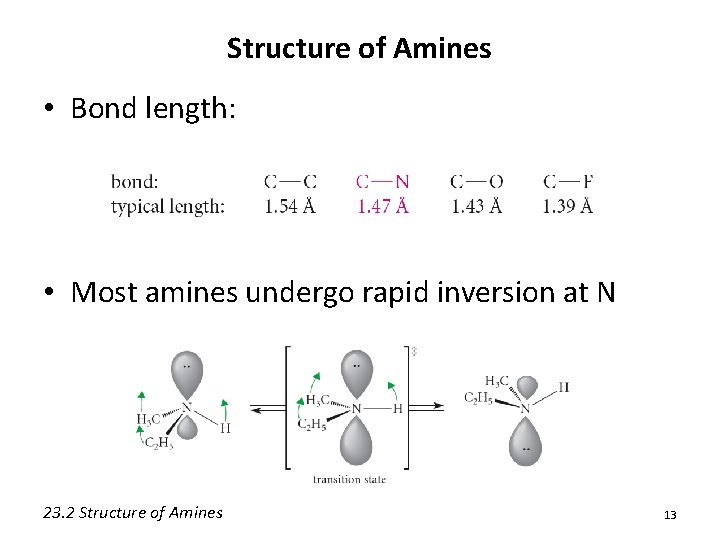
Structure of Amines • Bond length: • Most amines undergo rapid inversion at N 23. 2 Structure of Amines 13

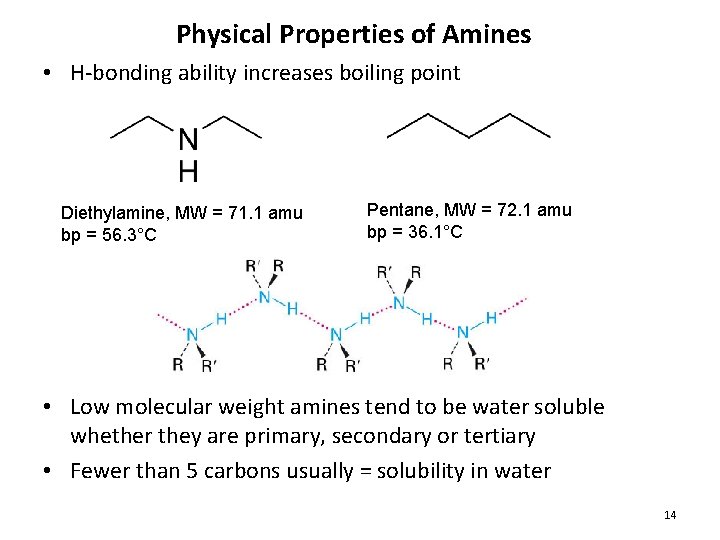
Physical Properties of Amines • H-bonding ability increases boiling point Diethylamine, MW = 71. 1 amu bp = 56. 3°C Pentane, MW = 72. 1 amu bp = 36. 1°C • Low molecular weight amines tend to be water soluble whether they are primary, secondary or tertiary • Fewer than 5 carbons usually = solubility in water 14

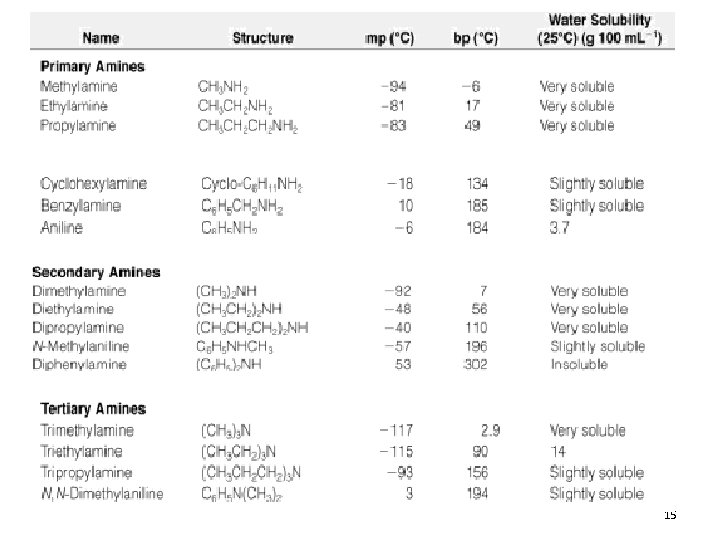
15

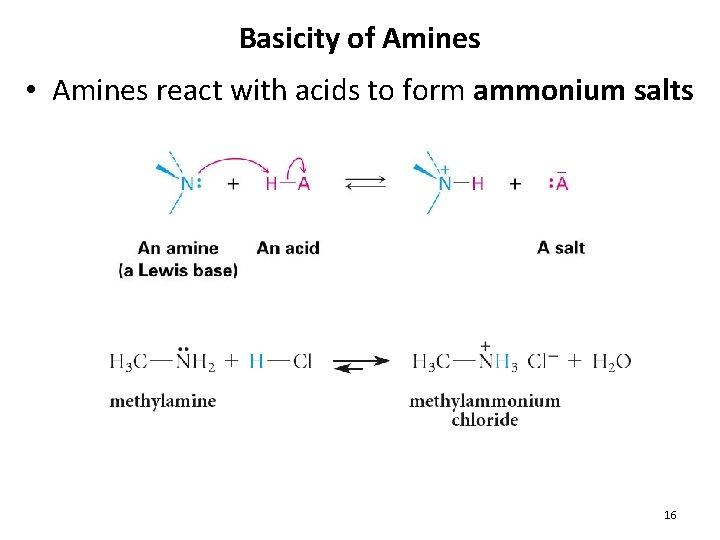
Basicity of Amines • Amines react with acids to form ammonium salts 16

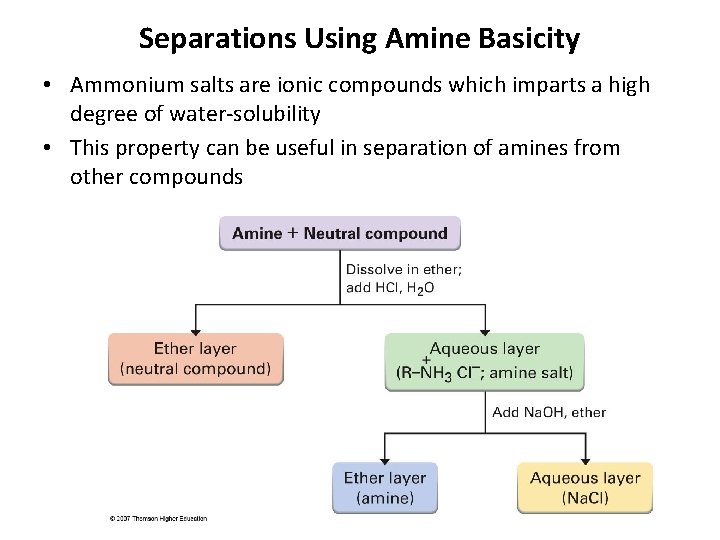
Separations Using Amine Basicity • Ammonium salts are ionic compounds which imparts a high degree of water-solubility • This property can be useful in separation of amines from other compounds

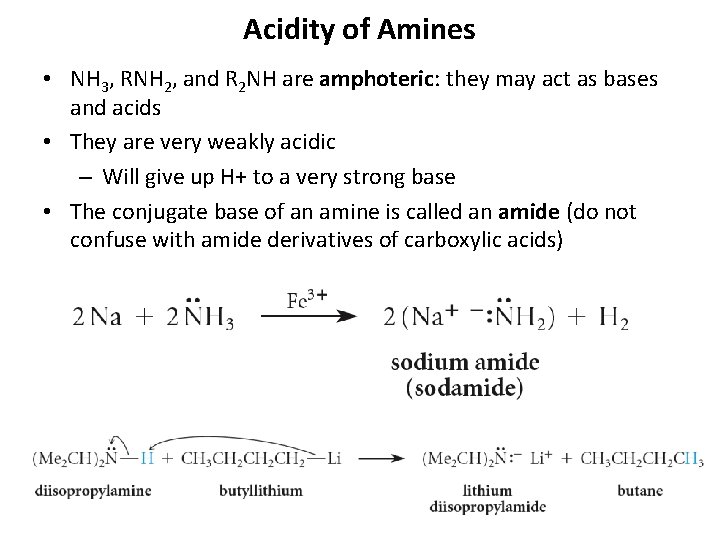
Acidity of Amines • NH 3, RNH 2, and R 2 NH are amphoteric: they may act as bases and acids • They are very weakly acidic – Will give up H+ to a very strong base • The conjugate base of an amine is called an amide (do not confuse with amide derivatives of carboxylic acids) 18

Quaternary Salts • Quaternary ammonium and phosphonium salts are compounds in which all four groups around the N and P are alkyl or aryl 23. 6 Quaternary Ammonium and Phosphonium Salts 19

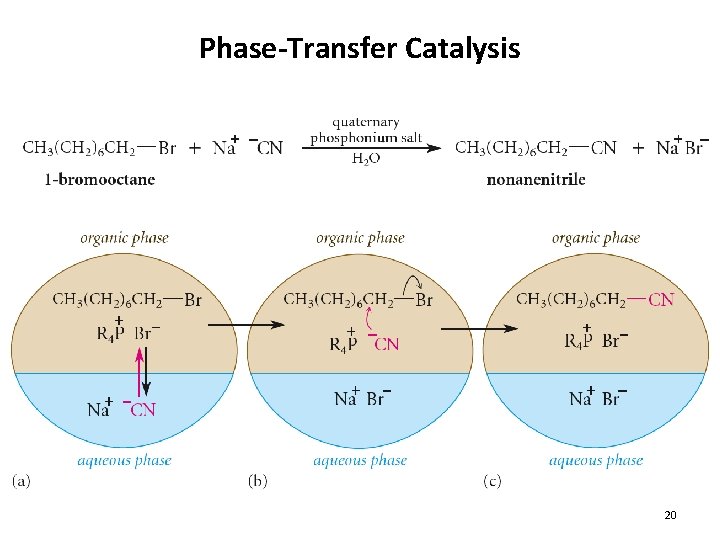
Phase-Transfer Catalysis 20

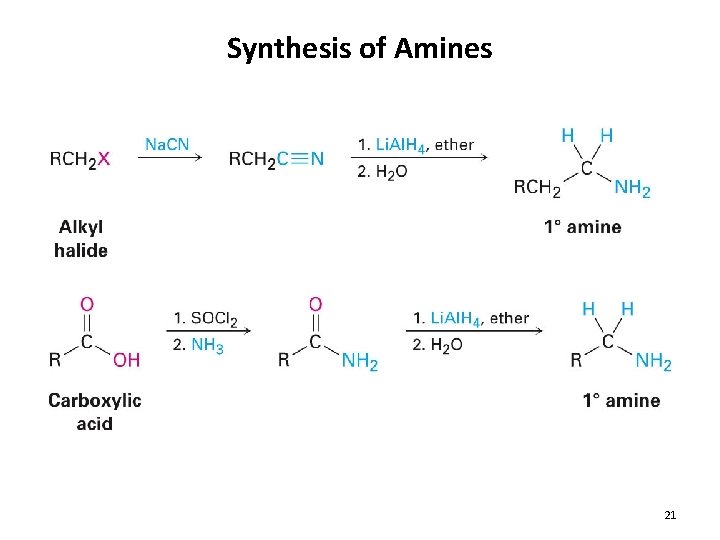
Synthesis of Amines 21

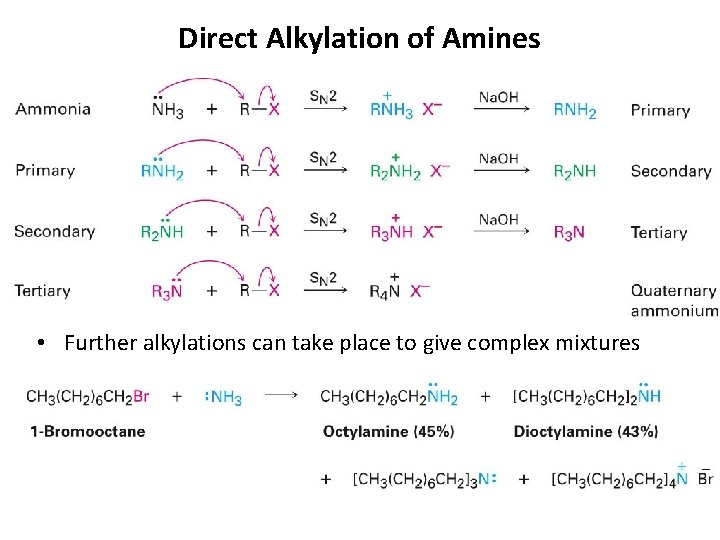
Direct Alkylation of Amines • Further alkylations can take place to give complex mixtures

Gabriel Synthesis of Primary Amines • Direct alkylation is not a good method for the preparation of primary amines • The Gabriel synthesis allows for controlled preparation 23. 11 Synthesis of Amines 23

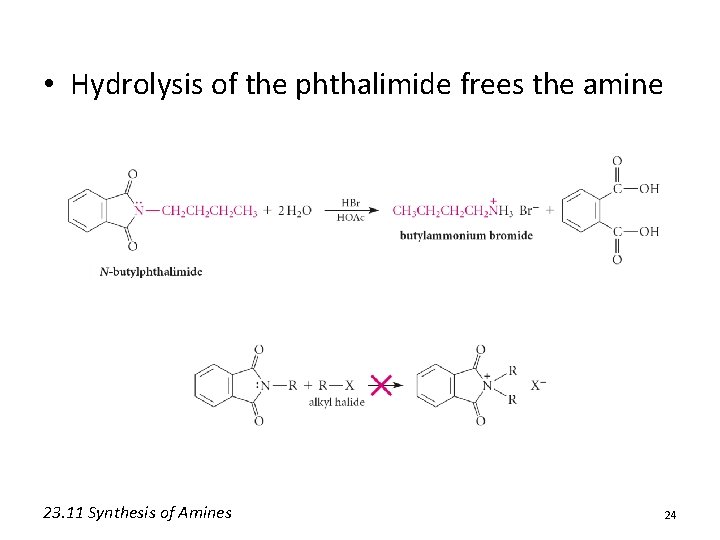
• Hydrolysis of the phthalimide frees the amine 23. 11 Synthesis of Amines 24

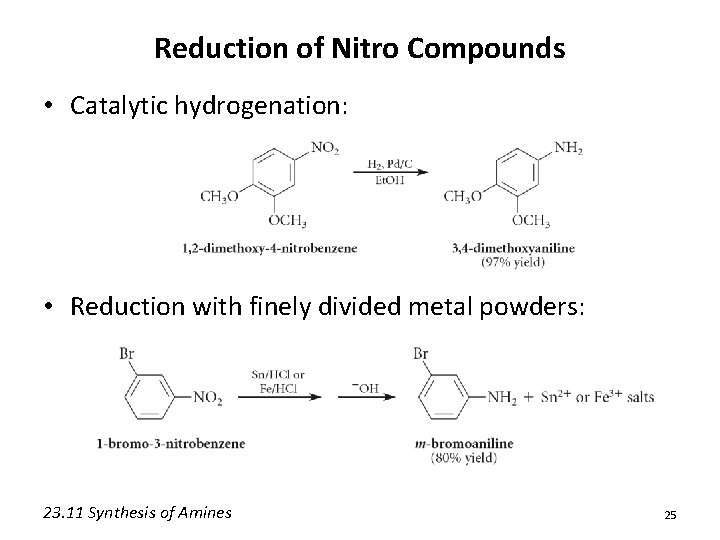
Reduction of Nitro Compounds • Catalytic hydrogenation: • Reduction with finely divided metal powders: 23. 11 Synthesis of Amines 25

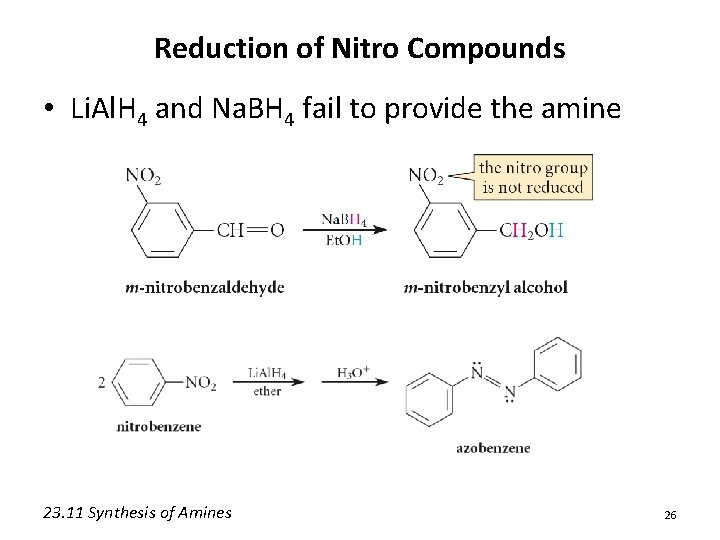
Reduction of Nitro Compounds • Li. Al. H 4 and Na. BH 4 fail to provide the amine 23. 11 Synthesis of Amines 26

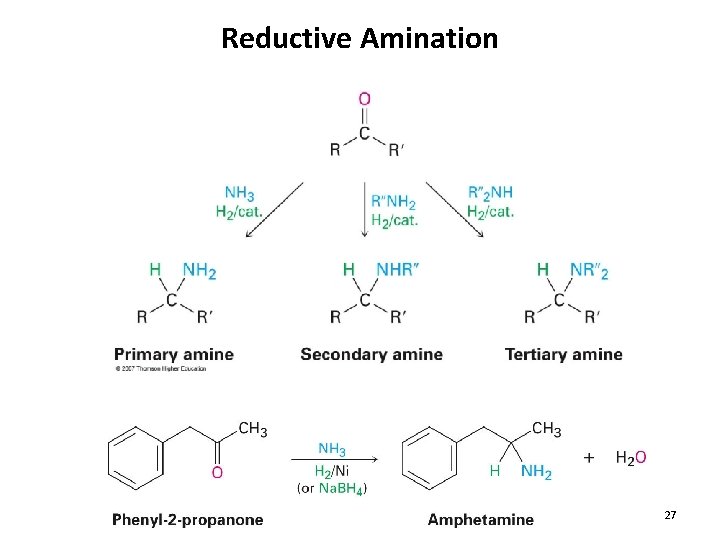
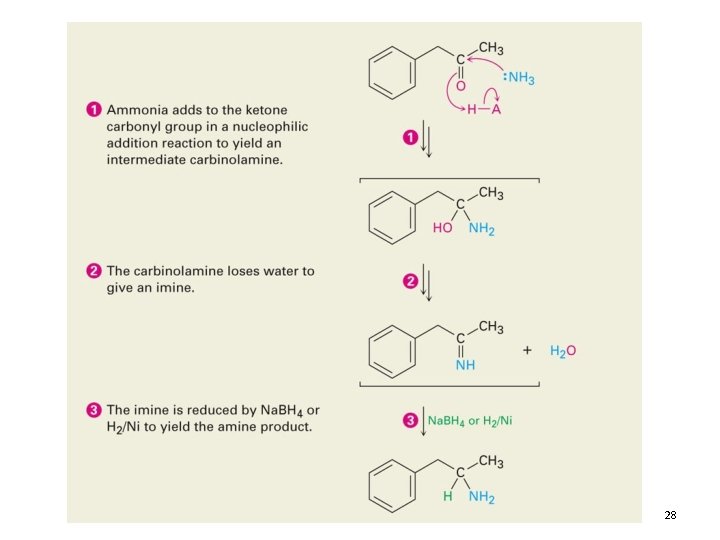
Reductive Amination 27

28

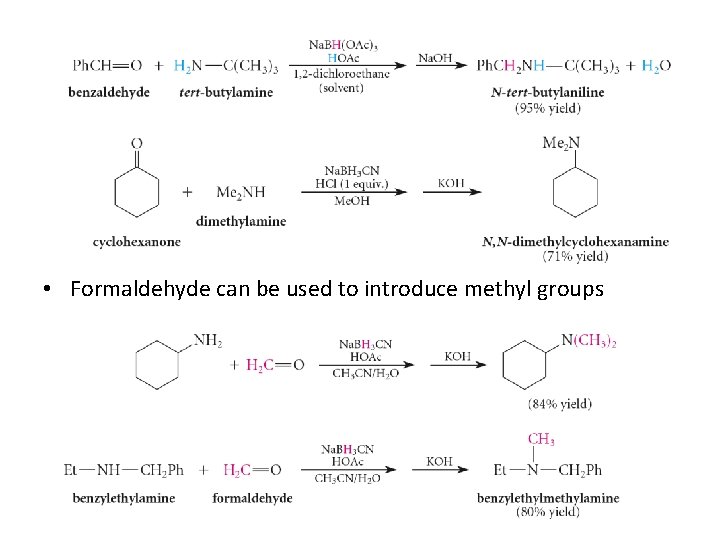
• Formaldehyde can be used to introduce methyl groups

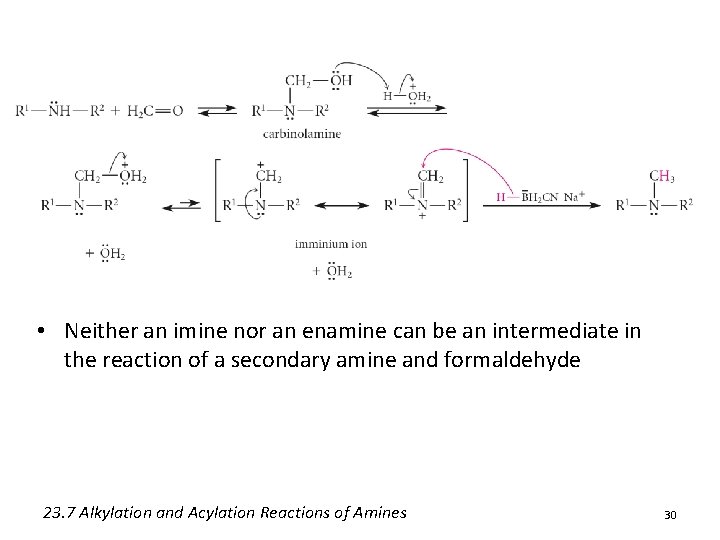
• Neither an imine nor an enamine can be an intermediate in the reaction of a secondary amine and formaldehyde 23. 7 Alkylation and Acylation Reactions of Amines 30