Chapter 10 Chemical Quantities Section 10 1 The


















- Slides: 20

Chapter 10 Chemical Quantities Section 10. 1 The Mole: A measurement of Matter

What is a Mole? A mole (mol) of a substance is 6. 02 X 1023 representative particles of that substance. Usually atoms or molecules. 6. 02 X 1023 is called Avogadro’s number

Converting Number of Particles to Moles Particles Molecules or atoms Av. Moles number e. g. How many moles of magnesium in 1. 25 X 10 23 atoms of magnesium = 0. 208 mol Mg Moles - Particles Page 291: #3 & 4

Converting Moles to Number of Particles e. g. How many atoms are in 2. 12 mol of propane (C 3 H 8)? Number of atoms = 2. 12 x 6. 02 x 10 23 x 11 = 1. 4 x 10 25 atoms Page: 292 #5 & 6

The mass of a Mole of an Element The mass of a mole of an element is its molar mass. How can you calculate the molar mass of a compound? To calculate the molar mass of a compound: 1. Find the number of grams of each element. 2. Add the masses of the elements in the compound. e. g. what is the molar mass of calcium oxide? Chemical formula = Ca. O, Ca = 40, O = 16 Molar mass = 40 + 16 = 56 g Page: 296, #7 & 8

Moles – Atoms or Molecules or Particles Number of Moles If atoms Multiplies by number of atoms Avogadro’s Number

Section 10. 2 Mole – Mass & Mole – Volume Relationships

Converting Mass to Moles e. g. How many moles of iron(III) oxide are contained in 92. 2 g of pure Fe 2 O 3? Mass = 92. 2 g Molar mass = 2 x 55. 8 + 3 x 16 = 159. 6 g/mol Number of moles = 92. 2/159. 6 = 0. 578 mol Fe 2 O 3 Moles - Mass Page: 299, # 18 & 19

Converting Moles to Mass = number of Moles x molar mass e. g. What is the mass of 9. 45 mol of aluminum oxide? Chemical Formula = Al 2 O 3 Number of moles = 9. 45 mol Molar mass = 2 x 27 + 3 x 16 = 102 g Al 2 O 3 Mass = 9. 45 x 102 =964 g Al 2 O 3 Page: 298, # 16 & 17

The volume of a Mole of an Element The volume of a mole of an element is called molar volume The molar volume of a gas at STP is 22. 4 L Standard Temperature and Pressure To calculate the volume of a gas at STP: Volume of gas = Number of Moles x 22. 4 e. g. Determine the volume of 0. 6 mol sulfur dioxide gas at STP. Volume of SO 2 = 0. 6 x 22. 4 = 13. 4 L SO 2 Moles - Volume Page: 301, #20 & 21

Calculating Molar Mass from Density Molar mass = density at STP x molar volume at STP e. g. The density of a gaseous compound carbon and oxygen is found to be 1. 964 g/L at STP. What is the molar mass of the compound? Molar mass = 1. 964 x 22. 4 = 44 g/mol Page: 302, #22 & 23 Moles - Density

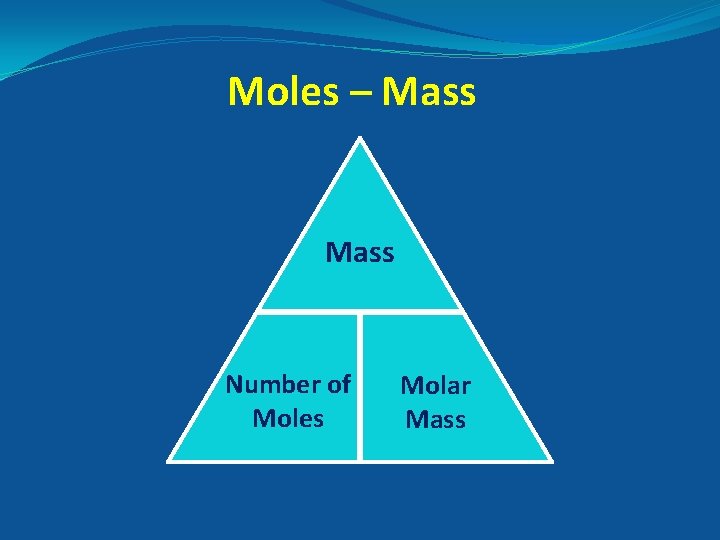
Moles – Mass Number of Moles Molar Mass

Moles – Volume Number of Moles 22. 4

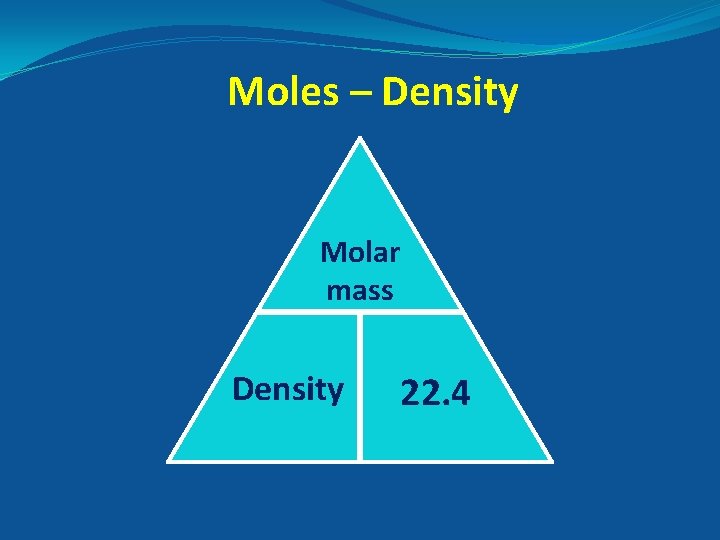
Moles – Density Molar mass Density 22. 4

Section 10. 3 Percent Composition and Chemical Formulas

The percent composition is the relative amounts of the elements in a compound. The percent by mass of an element in a compound is the number of grams of the element divided by the mass in grams of the compound, multiplied by 100 Calculating Percent Composition from Mass Data e. g. When a 13. 6 g sample of a compound containing only magnesium and oxygen. 5. 4 g of oxygen is obtained. What is the percent composition of this compound? Mass of compound = 13. 6 g Mass of oxygen = 5. 4 g Mass of magnesium = 13. 6 – 5. 4 = 8. 2 g Page 306, # 32 & 33

Calculating Percent Composition from the Chemical Formula e. g. Propane (C 3 H 8) is one of the compounds obtained from petroleum. Calculate the percent composition of propane. Mass of C in C 3 H 8 = 36. 0 g Mass of H in C 3 H 8 = 8. 08 g Molar mass of C 3 H 8 = 44. 08 g/mol Page: 307, # 34 & 35

Empirical Formula The empirical formula of a compound shows the smallest whole-number ratio of the atoms in the compound. It shows the kinds and lowest relative count of atoms or moles of atoms in molecules or a compound. An empirical formula may or may not be the same as a molecular formula. For example, the lowest ratio of hydrogen to oxygen in hydrogen peroxide is 1: 1 So the empirical formula of hydrogen peroxide is HO The actual molecular formula of hydrogen peroxide has twice the number of atoms as the empirical formula. The molecular formula is (HO) x 2 or H 2 O 2 Notice that the ratio of hydrogen to oxygen is still the same. The molecular formula tells the actual number of each kind of atom present in a molecule of the compound. For carbon dioxide, the empirical and molecular formulas are the same – CO 2

e. g. A compound is analyzed and found to contain 25. 9% nitrogen and 74. 1% oxygen. What is the empirical formula of the compound? 25. 9% N 74. 1% O Step 1. Assume that you have 100 g of the compound, 25. 9 g 74. 1 g Step 2. Divide each by its atomic mass. 25. 9 74. 1 14 16 1. 85 mol 4. 63 mol Step 3. Divide each by the smallest number of moles. 1. 85 4. 63 1. 85 1 2. 5 • Multiply all by 2 The empirical formula is N 2 O 5 Page: 310, # 36 & 37

Molecular Formula is the same as the empirical formula or it is a simple whole-number multiple of its empirical Once you determined the empirical formula of a compound, you can determine its molecular formula, but you must know the compound’s molar mass and empirical formula mass. e. g. Calculate the molecular formula of a compound whose molar mass is 60 g/mol and empirical formula is CH 4 N Empirical formula = CH 4 N Molar mass = 60 g/mol Molecular formula = C? H? N? Step 1. Calculate the empirical formula mass Empirical formula mass = 1 x 12 + 4 x 1. 01 + 1 x 14 = 30. 04 g/mol Step 2. Divide the molar mass by the empirical formula mass 60 = 2 30. 04 Step 3. Multiply the formula subscripts by this value (factor) Molecular formula = 2 x CH 4 N = C 2 H 8 N 2 Page: 312, # 38 & 39