Ch 13 Solutions Dr Namphol Sinkaset Chem 152
























- Slides: 27

Ch. 13: Solutions Dr. Namphol Sinkaset Chem 152: Introduction to General Chemistry

I. Chapter Outline I. III. IV. V. Introduction Concentration Preparation of a Solution Dilution Solution Stoichiometry

I. Introduction • Solution chemistry is the most well studied – why? • Although solutions tend to be liquid based, the general definition allows for other types of solutions. • solution: homogeneous mixture of two or more substances

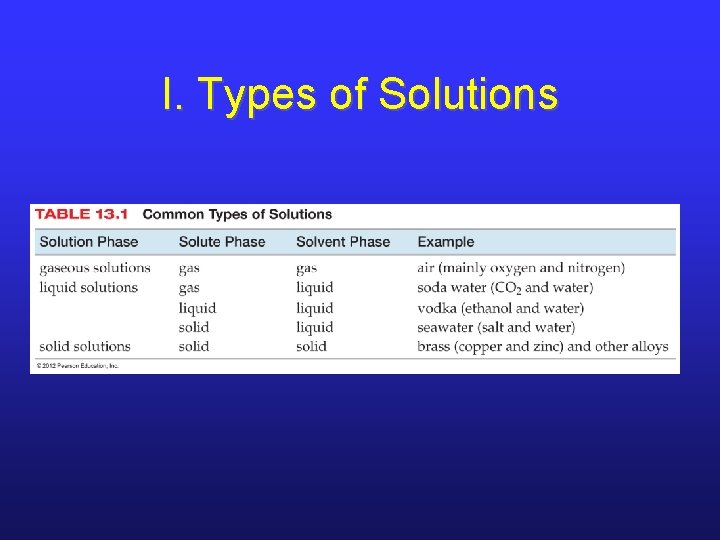
I. Types of Solutions

I. Solution Components • There are two parts of a solution. § solute: substance present in smaller amount § solvent: substance present in larger amount • For stoichiometry, the important aspect of a solution is its concentration. • concentration: amount of solute present in a certain volume of solution

II. Solution Concentration • Solutions can be either dilute or concentrated. • dilute: small amount of solute relative to amount of solvent • concentrated: large amount of solute relative to amount of solvent • There are several different ways to express solution concentration.

II. Mass Percent • Mass percent expresses the number of grams of solute per 100 g of solution.

II. Using Mass Percent • If expressed as a fraction over 100, the mass percent can be used as a conversion factor. • For a solution that is 23. 2% ethanol by mass: OR

II. Sample Problem • Calculate the mass percent of a solution containing 15. 5 g of fructose and 249. 6 g of water.

II. Sample Problem • Ocean water contains 3. 5% sodium chloride by mass. How much sodium chloride does a 2. 00 L sample of ocean water contain? Note that ocean water has a density of 1. 027 g/m. L.

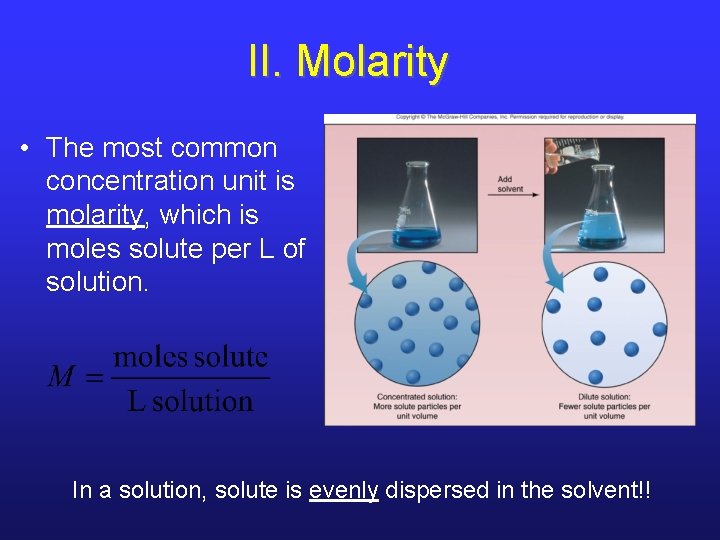
II. Molarity • The most common concentration unit is molarity, which is moles solute per L of solution. In a solution, solute is evenly dispersed in the solvent!!

II. Using Molarity • Molarity can be used as a conversion factor between moles of solute and liters of solution. • For a 0. 500 M Na. Cl solution: OR

II. Sample Problem • Calculate the molarity of a solution formed when 24. 2 g Na. Cl is dissolved in 124. 1 m. L of water.

II. Sample Problem • How many grams of Na 2 HPO 4 are needed to make 1. 50 L of a 0. 500 M Na 2 HPO 4 solution?

II. Concentration of Ions • When soluble ionic compounds are added to water, they dissociate. • Soluble ionics dissociate because the solventsolute attraction is greater than the solute attraction.

II. The Dissolving Process

II. Na. Cl in Solution

II. Ion Concentrations • Sometimes, we need to know the concentration of an individual ion. • When calculating, we must account for the ratio seen in the formula of the ionic compound. • e. g. Mg. Cl 2 has two anions for every one cation; anion will be twice as concentrated.

II. Sample Problem • Calculate the concentration of the ions when 19. 6 g of iron(III) sulfate is dissolved in enough water to make 200. 0 m. L of solution.

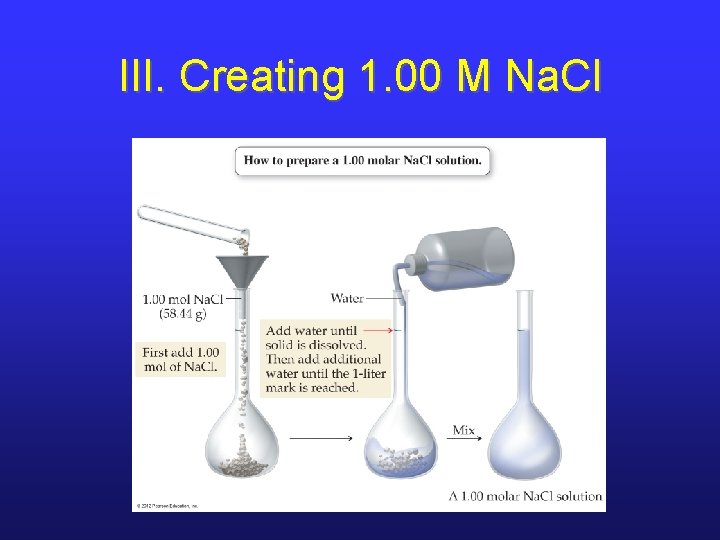
III. Solution Creation • The last sample problem is an example of a calculation needed in order to create a certain volume of solution of a certain concentration. • This type of calculation is very common in any research lab. • To make the solution, special glassware and a specific procedure must be used.

III. Creating 1. 00 M Na. Cl

IV. Dilution • Less concentrated solutions can be made from more concentrated solutions in a process called dilution. • The more concentrated solution is known as a stock solution. • To perform a dilution, you need to know how much of the stock solution to use.

IV. Dilution Equation • The following dilution equation makes it easy to calculate how much stock solution is needed.

IV. Sample Problem • How many m. L of a 2. 0 M Na. Cl solution are needed to make 250. 0 m. L of a 0. 50 M Na. Cl solution?

V. Solution Stoichiometry • Since molarity is a ratio between moles of solute and volume of solution, it can be used in stoichiometric calculations. • The key is to remember that molarity breaks down into units of mole/L!

V. Sample Problem • How many m. L of 0. 10 M HCl reacts with 0. 10 g Al(OH)3 according to the reaction below? Al(OH)3(s) + 3 HCl(aq) Al. Cl 3(aq) + 3 H 2 O(l)

V. Sample Problem • How much Pb. Cl 2 forms when 267 m. L 1. 50 M lead(II) acetate reacts with 125 m. L 3. 40 M sodium chloride according to the reaction below? Pb(CH 3 COO)2(aq) + 2 Na. Cl(aq) Pb. Cl 2(s) + 2 Na. CH 3 COO(aq)