BIOLOGY OF HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS G E Janka Hamburg

BIOLOGY OF HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS G. E. Janka Hamburg Iran 2018

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Biology of HLH Pathogenesis Which biological mechanisms lead to the disease? Pathophysiology Which physiological processes explain symptoms and laboratory findings?

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Definition HLH is not an independent disease but a life-threatening clinical syndrome of severe uncontrolled hyperinflammation, due to activation of lymphocytes and macrophages secreting high levels of cytokines

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis There are similarities and differences in the pathogenesis of genetic and acquired HLH In both there is an inadequate immune reaction with high levels of cytokines In genetic HLH, in most cases this is due to permanently impaired function of natural killer cells and cytotoxic T cells, leading to the inability to kill the target and to terminate the immune response In acquired HLH mechanisms there are several possible mechanisms.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Genetic defects Disease Chromosome Gene function Familial HLH FLH-1 FLH-2 FLH-3 FLH-4 FLH-5 9 q 21. 3 -22 10 q 22 17 q 25 6 q 24 19 p 13 not known PFR 1 UNC 13 D STX 11 UNC 18 B (STXBP 2) not known induction of apoptosis vesicle priming vesicle docking/fusion? RAB 27 A vesicle docking Griscelli syndr. 2 15 q 21 Chédiak-Higashi syndrome 1 1 q 42. 1 - q 42. 2 LYST vesicle maturation/ fusion XLP (Purtilo syndrome) Xq 25 SH 2 D 1 A, XIAP signal transduction and activation of lymphocytes
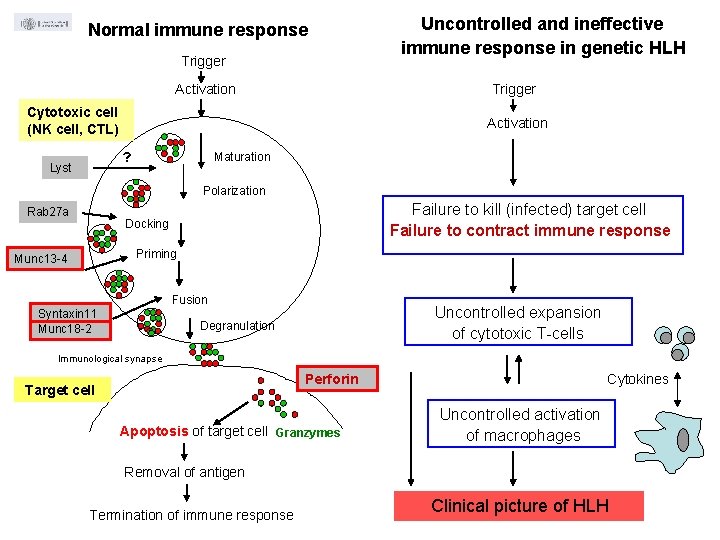
Normal immune response Trigger Activation Cytotoxic cell (NK cell, CTL) Activation Maturation ? Lyst Uncontrolled and ineffective immune response in genetic HLH Polarization Rab 27 a Failure to kill (infected) target cell Failure to contract immune response Docking Priming Munc 13 -4 Fusion Syntaxin 11 Munc 18 -2 Uncontrolled expansion of cytotoxic T-cells Degranulation Immunological synapse Perforin Target cell Apoptosis of target cell Granzymes Cytokines Uncontrolled activation of macrophages Removal of antigen Termination of immune response Clinical picture of HLH

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Perforin is a key molecule for cytotoxic T-cells (CTLs) and NK cells Perforin leads to apoptosis of (infected) cells and downregulation of the immune response (killing of i. DCs, CTLs) It is important for immune homeostasis, protecting against autoimmune diseases. It plays a role in immune surveillance against malignancies.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Besides cytotoxic defects in T cells there are other pathogenetic mechanisms to induce HLH T cell activation is not always necessary for HLH: HLH can develop in patients with SCID and T cells <100/µl (Bode S 2015)

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis: Role of the innate immune system Activation of toll-like receptor (TLR)-9 induces HLH in mice (Behrens E 2011) Abrogating the function of the TLR-adaptor myd 88 prevents HLH in UNC 13 D mice (Krebs P 2011) Myd 88 is also an adaptor for signaling by IL-1 family cytokines In LCMV- infected Prf 1 -/- mice blockade of ST 2, the receptor for the alarmin IL-33 reduced severity and mortality of HLH (Rood JE 2016) Mutations in human NLRC 4, a component of the inflammasome, lead to recurrent HLH (Cannae SW 2014) In mice protection from excessive inflammasome activation is mediated by XIAP (Yabal M 2014)

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Other possible pathogenetic mechanisms in HLH

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis: possible factors Inhibition of cytotoxic function by viruses and cytokines Interference with apoptosis by viruses and tumor cells Secretion of cytokines from tumor cells Acquired immune dysfunction by drugs; HIV Genetic factors Imbalance between viral load and immune effector cells

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Inhibition of cytotoxic function Viral evasion strategies Avian influenza (H 5 N 1) virus downregulates expression of perforin in CD 8+ T cells (Hsieh SM 2006). EBV inhibits expression of SAP, impairing the cytolytic response of CTLs to B-cell mediated Ag presentation (Chuang HC 2005). Viruses can interfere with NK cell function by inhibiting activating receptors, stimulating inhibitory receptors, and by blocking chemokines and cytokines necessary for NK cell function (Orange JS 2002). Role of cytokines High levels of IL-6 (as present in s. JIA) inhibit NK cell cytotoxicity (Cifaldi L 2015). High levels of pro-and anti-inflammatory cytokines in sepsis lead to decreased NK-cell activity and T-cell exhaustion (Hotchkiss RS 2013).

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Inhibiton of apoptosis Interference with apoptosis by viruses or tumor cells Viruses interfere both with the intrinsic and extrinsic pathway of apoptosis (Alcami A 2000, Tortorella D 2000). : - Expression of antiapoptotic molecules Blocking of apoptotic signals via the extrinsic pathway Inactivation of p 53 Inhibition of caspases Cancer cells interfere with all pathways of apoptosis (Hassan M 2014).

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Secretion of cytokines by tumor cells (Nishiwaki U 2016) Acquired immune dysfunction Treatment with immunosuppressive agents HIV-infection

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Genetic factors Single nucleotide polymorphisms (SNPs) Severe malaria is associated with several SNPs (Sinha S 2008; Seaby E. 2016). Infectious mononucleosis or silent infection with EBV is associated with SNPs in HLA class I antigens (Mc. Aulay KA 2007). Severity of enterovirus-71 infection is associated with SNP in OAS 3 encoding the IFN-induced oligoadenylate synthetase (Tan Y 2017). Risk of fatal outcome of avian or pandemic influenza is associated with SNPs in genes of innate immunity (Lee N 2017). Heterozygous mutations in FHL-related genes Mutations in other genes regulating the immune system

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Genetic factors Heterozygous mutations in HLH-related genes (Cheng X, Clin Genet 2018) Allele frequencies >1% excluded) 265 patients (243 children) Bi-allelic (19) or hemizygous (16) mutations (SAP, XIAP) 35/265 (13%) patients At least 1 mutation of uncertain significance 10/35 (29%) Heterozygous mutations 44/265 (17%) Of uncertain significance 24/44 (54%) Digenic mutations 7/265 (3%) At least one mutation of uncertain significance 4/7

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Genetic factors Mutations in other genes regulating the immune system (Chinn I Blood 2018) 101 children with genetic testing, fulfilling HLH-2004 criteria Whole-exome sequencing (WES) in 48 patients Biallelic mutations 19/101 (19%) 1 additional RAB 27 case WES 28/48 (58%) patients with mutations 14 potential primary immune deficiencies 10 inflammasome genes + 1 NRAS 5 other candidate genes in pathways of cytotoxicity, regulation of immune activation, and cotrol of hematopoiesis
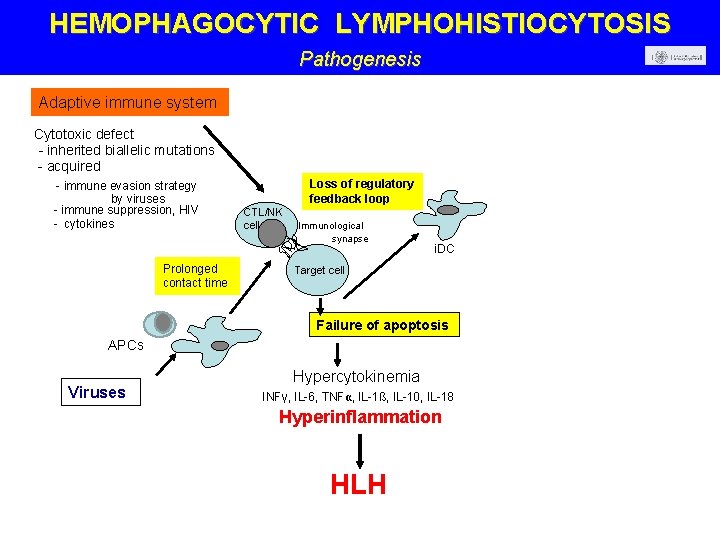
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Adaptive immune system Cytotoxic defect - inherited biallelic mutations - acquired - immune evasion strategy by viruses - immune suppression, HIV - cytokines Prolonged contact time Loss of regulatory feedback loop CTL/NK cell Immunological synapse i. DC Target cell Failure of apoptosis APCs Viruses Hypercytokinemia INFγ, IL-6, TNFα, IL-1ß, IL-10, IL-18 Hyperinflammation HLH
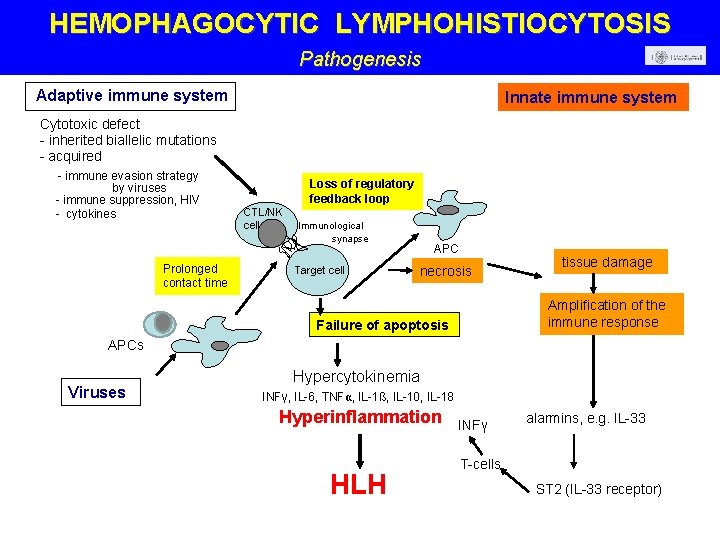
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Adaptive immune system Innate immune system Cytotoxic defect - inherited biallelic mutations - acquired - immune evasion strategy by viruses - immune suppression, HIV - cytokines Prolonged contact time Loss of regulatory feedback loop CTL/NK cell Immunological synapse Target cell APC necrosis tissue damage Amplification of the immune response Failure of apoptosis APCs Viruses Hypercytokinemia INFγ, IL-6, TNFα, IL-1ß, IL-10, IL-18 Hyperinflammation HLH INFγ alarmins, e. g. IL-33 T-cells ST 2 (IL-33 receptor)
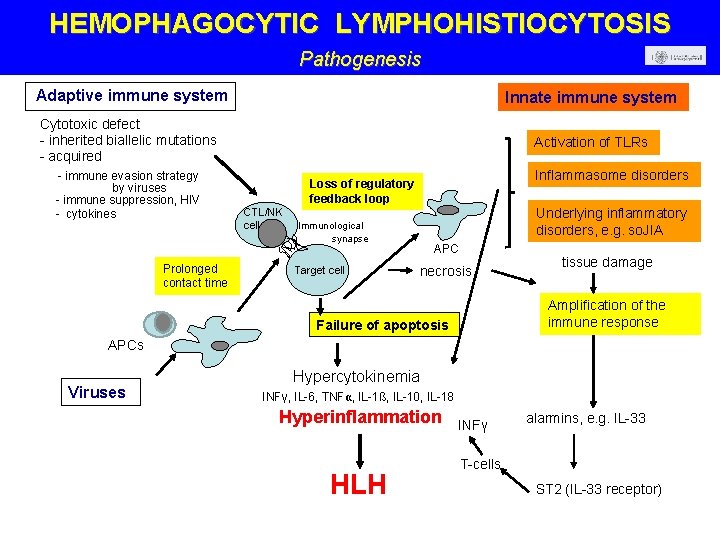
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Adaptive immune system Innate immune system Cytotoxic defect - inherited biallelic mutations - acquired Activation of TLRs - immune evasion strategy by viruses - immune suppression, HIV - cytokines Prolonged contact time Inflammasome disorders Loss of regulatory feedback loop CTL/NK cell Underlying inflammatory disorders, e. g. so. JIA Immunological synapse Target cell APC necrosis tissue damage Amplification of the immune response Failure of apoptosis APCs Viruses Hypercytokinemia INFγ, IL-6, TNFα, IL-1ß, IL-10, IL-18 Hyperinflammation HLH INFγ alarmins, e. g. IL-33 T-cells ST 2 (IL-33 receptor)
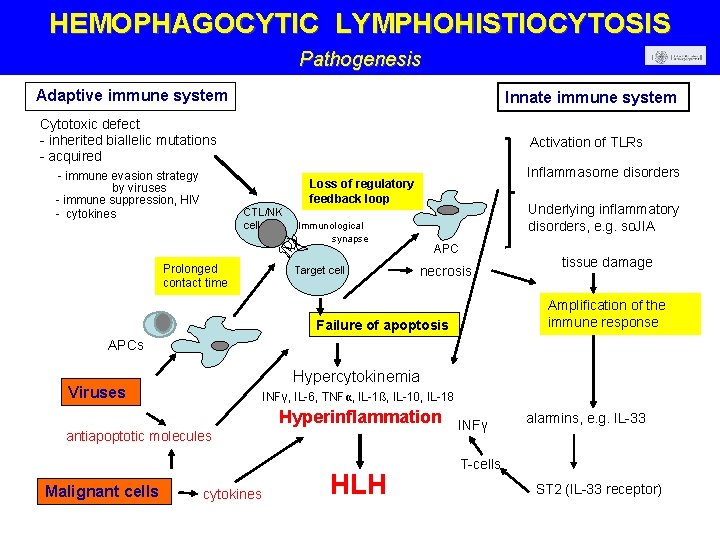
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Adaptive immune system Innate immune system Cytotoxic defect - inherited biallelic mutations - acquired - immune evasion strategy by viruses - immune suppression, HIV - cytokines Activation of TLRs Inflammasome disorders Loss of regulatory feedback loop CTL/NK cell Prolonged contact time Underlying inflammatory disorders, e. g. so. JIA Immunological synapse Target cell APC necrosis tissue damage Amplification of the immune response Failure of apoptosis APCs Hypercytokinemia Viruses INFγ, IL-6, TNFα, IL-1ß, IL-10, IL-18 Hyperinflammation antiapoptotic molecules Malignant cells cytokines HLH INFγ alarmins, e. g. IL-33 T-cells ST 2 (IL-33 receptor)
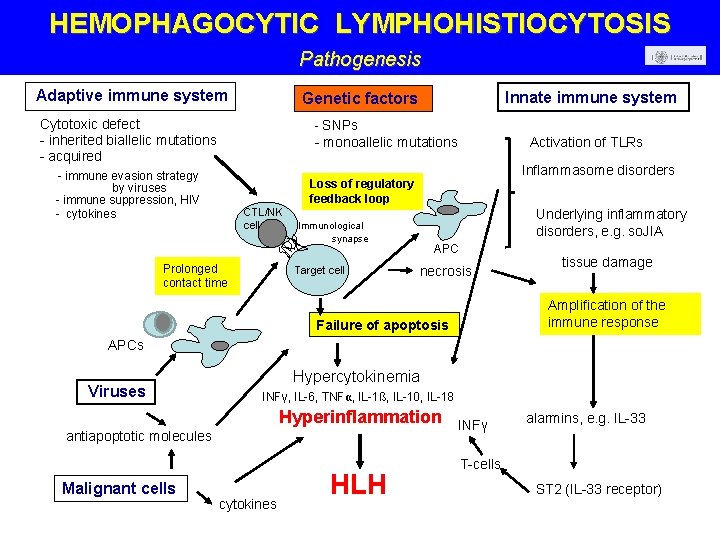
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Adaptive immune system Innate immune system Genetic factors Cytotoxic defect - inherited biallelic mutations - acquired - SNPs - monoallelic mutations - immune evasion strategy by viruses - immune suppression, HIV - cytokines Activation of TLRs Inflammasome disorders Loss of regulatory feedback loop CTL/NK cell Prolonged contact time Underlying inflammatory disorders, e. g. so. JIA Immunological synapse Target cell APC necrosis tissue damage Amplification of the immune response Failure of apoptosis APCs Viruses Hypercytokinemia INFγ, IL-6, TNFα, IL-1ß, IL-10, IL-18 Hyperinflammation antiapoptotic molecules Malignant cells cytokines HLH INFγ alarmins, e. g. IL-33 T-cells ST 2 (IL-33 receptor)
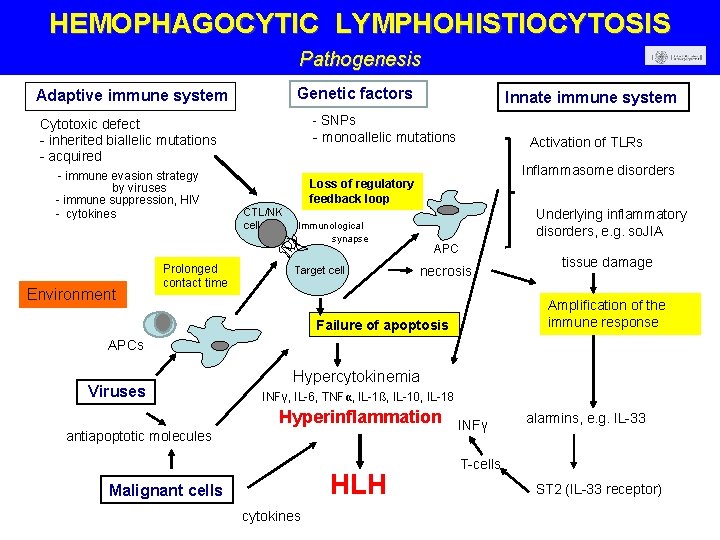
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathogenesis Genetic factors Adaptive immune system - SNPs Cytotoxic defect - inherited biallelic mutations - acquired - immune evasion strategy by viruses - immune suppression, HIV - cytokines Environment Prolonged contact time Innate immune system - monoallelic mutations Activation of TLRs Inflammasome disorders Loss of regulatory feedback loop CTL/NK cell Underlying inflammatory disorders, e. g. so. JIA Immunological synapse Target cell APC necrosis tissue damage Amplification of the immune response Failure of apoptosis APCs Viruses Hypercytokinemia INFγ, IL-6, TNFα, IL-1ß, IL-10, IL-18 Hyperinflammation antiapoptotic molecules HLH Malignant cells cytokines INFγ alarmins, e. g. IL-33 T-cells ST 2 (IL-33 receptor)

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS PATHOPHYSIOLOGY Which physiological processes explain clinical and laboratory findings?

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Clinical symptoms and laboratory findings Fever, hepatosplenomegaly, cytopenias Neurological symptoms High ferritin, low fibrinogen, high triglycerides, transaminases, bilirubin, LDH CSF pleocytosis and/or elevated protein Hemophagocytosis

HLH: SYMPTOMS AND CYTOKINES Symptoms of HLH can be explained by hypercytokinemia and organ infiltration by histiocytes and lymphocytes • fever (interleukins) • pancytopenia (TNF- , INF- , heavy subunit of ferritin, phagocytosis) • high triglycerides (TNF- , INF- • low fibrinogen (plasminogen activator lipoprotein lipase ) hyperfibrinolysis) • high ferritin (activated macrophages), high levels of soluble interleukin-2 receptor (s. CD 25) (activated lymphocytes) • hepatosplenomegaly, elevated transaminases/bilirubin, neurological symptoms (infiltration by activated lymphocytes and histiocytes; cytokines and chemokines) All these symptoms and laboratory values can also be present in infections which take a normal course; however, in HLH these findings are abnormally out of proportion
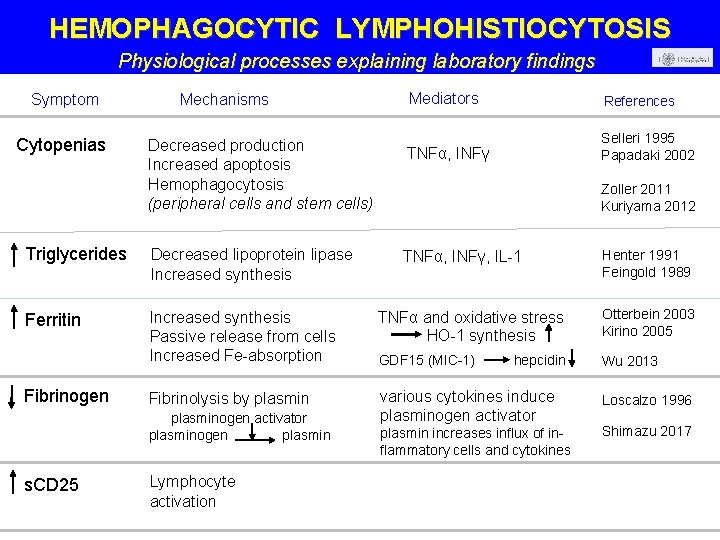
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Physiological processes explaining laboratory findings Symptom Cytopenias Mechanisms Decreased production Increased apoptosis Hemophagocytosis (peripheral cells and stem cells) Mediators References TNFα, INFγ Selleri 1995 Papadaki 2002 Zoller 2011 Kuriyama 2012 Triglycerides Decreased lipoprotein lipase Increased synthesis Ferritin Increased synthesis Passive release from cells Increased Fe-absorption TNFα and oxidative stress HO-1 synthesis Otterbein 2003 Kirino 2005 GDF 15 (MIC-1) Wu 2013 Fibrinolysis by plasmin various cytokines induce plasminogen activator Fibrinogen plasminogen activator plasminogen plasmin s. CD 25 Lymphocyte activation TNFα, INFγ, IL-1 hepcidin plasmin increases influx of inflammatory cells and cytokines Henter 1991 Feingold 1989 Loscalzo 1996 Shimazu 2017

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathophysiology HLH and bone marrow stem cells Kuriyama T, Blood 2012 Self-recognition to prevent phagocytosis is regulated by interaction of CD 47 (expressed in hematopoietic cells) and SIRPA (expressed in macrophages). Inflammatory cytokines downregulate CD 47 specifically in HSCs. CD 47 is downregulated in stem cells of HLH patients with active disease, leading to increased phagocytosis. The number of HSCs in HLH patients was reduced to 23% of those in healthy adults. Pascutti MF, Frontiers Immunol 2016 Several viruses infect HSCs or supportive stromal cells in the bone marrow.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Pathophysiology HLH and role of plasmin Shimazu H, Blood 2017 Activated macrophages produce plasminogen activator, leading to increased levels of plasmin. Plasmin can degrade fibrin clots. Plasmin regulates the influx of inflammatory cells and the inflammatory response by inducing transcription of cytokines. In a mouse model of HLH, established by repeated TLR-9 stimulation (Cp. G + DG), plasmin levels were excessively high. Genetic (plasminogen knock-out mice) and pharmacological (plasmin antibody) inhibition of plasmin reduced mortality and HLH symptoms.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Summary Knowledge of HLH has increased considerably over the past years. Discovery of mutation of genes involved in the cytolytic granule pathway has given important insights into the pathogenesis of familial HLH and of the immune system in general. Pathogenesis of acquired HLH in children and adults is still poorly understood. HLH in these patients is probably due to a combination of host factors, pathogen-associated factors and other triggers. Better insight into the pathogenesis and pathophysiology of HLH may show new ways for treatment.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Thank you for your attention! There is still much to learn about HLH!
- Slides: 31