TREATMENT OF HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS G E Janka Hamburg

TREATMENT OF HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS G. E. Janka Hamburg Iran November 2018

HAEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Diagnostic criteria 1 Familial disease/known genetic defect Clinical and laboratory criteria (5 out of 8) Fever Splenomegaly Cytopenia of => 2/3 cell lines Hemoglobin <90 g/l, platelets <100 x 109/l, ANC <1 x 109/l Hypertriglyceridemia and/or hypofibrinogenemia Fasting triglycerides =>3 mmol/l, fibrinogen <1. 5 g/l Haemophagocytosis in bone marrow, CSF, lymphnodes Ferritin =>500µg/l s. CD 25 =>2400 U/ml Decreased or absent natural killer cell activity Strong supportive evidence are cerebral symptoms with moderate pleocytosis and or elevated protein, elevated transaminases, bilirubin and LDH 1 HLH Study Group of the Histiocyte Society, Henter et al. PBC 2007

MAS - HLH New diagnostic criteria Multistep process: - Delphi survey to identify MAS features potentially suitable als criteria - Data collection on patients with s. JIA-associated MAS (362), patients with s. JIA not complicated by MAS (404), and patients with systemic infections (345) - Selection of candidate criteria from patients with consensus (391/428) in 28 experts (cut-off values calculated by ROC) - Selection of final criteria by consensus conference Final criteria Ravelli 2016 MAS is diagnosed in a febrile patient with s. JIA if the following criteria are met: Ferritin > 684 ng/m. L and any 2 of the following Platelet count ≤ 181 000/µL Aspartate aminotransferase (GOT) > 48 U/L Triglycerides > 156 mg/d. L Fibrinogen ≤ 360 mg/d. L
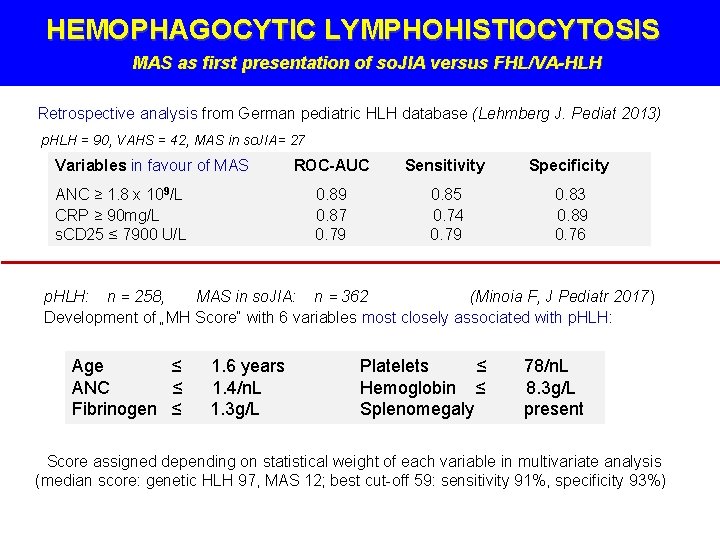
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS MAS as first presentation of so. JIA versus FHL/VA-HLH Retrospective analysis from German pediatric HLH database (Lehmberg J. Pediat 2013) p. HLH = 90, VAHS = 42, MAS in so. JIA= 27 Variables in favour of MAS ANC ≥ 1. 8 x 109/L CRP ≥ 90 mg/L s. CD 25 ≤ 7900 U/L ROC-AUC Sensitivity Specificity 0. 89 0. 87 0. 79 0. 85 0. 74 0. 79 0. 83 0. 89 0. 76 p. HLH: n = 258, MAS in so. JIA: n = 362 (Minoia F, J Pediatr 2017) Development of „MH Score“ with 6 variables most closely associated with p. HLH: Age ≤ ANC ≤ Fibrinogen ≤ 1. 6 years 1. 4/n. L 1. 3 g/L Platelets ≤ Hemoglobin ≤ Splenomegaly 78/n. L 8. 3 g/L present Score assigned depending on statistical weight of each variable in multivariate analysis (median score: genetic HLH 97, MAS 12; best cut-off 59: sensitivity 91%, specificity 93%)

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS How to differentiate between genetic and acquired HLH? Identification of an infectious agent severity of disease not helpful Age helpful ~90% of children below 1 year will have genetic HLH Do not forget: mutations in HLH-related genes are being increasingly identified in adolescents or adult patients Functional tests (degranulation, expression of perforin, SAP and XIAP) or mutation analysis allow differentiation between genetic and acquired forms, but are not widely available or still too expensive. The distinction between genetic and acquired HLH is not important for initial treatment which is guided mainly by the severity of the disease!

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS General statement It is important to realize that HLH is a syndrome where often an underlying trigger/condition can be identified. In children infections (mostly viral), autoinflammatory/ autoimmune diseases, malignant diseases and (rarely) metabolic diseases have to be considered. The search for a trigger is of utmost importance! Especially in adults and older children lymphomas (ultrasound, x-ray, CT, MRI) must be ruled out. Usually HLH-directed therapy is needed in addition to the specific treatment of the triggering condition (exceptions: leishmaniasis, tuberculosis).
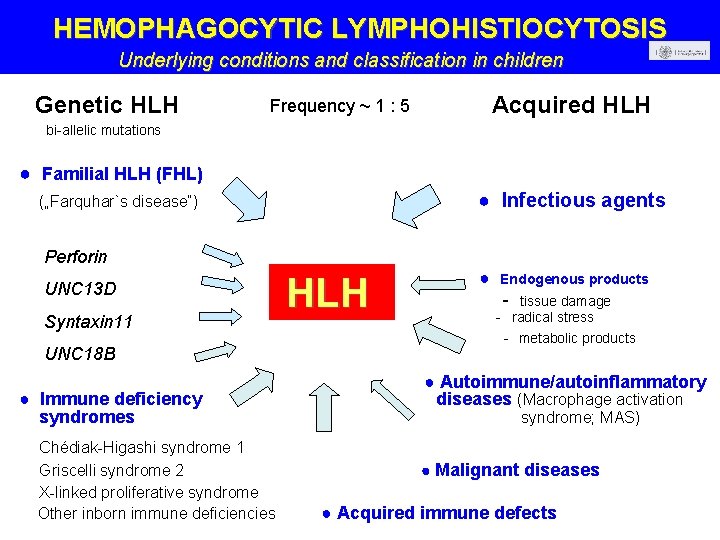
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Underlying conditions and classification in children Genetic HLH Acquired HLH Frequency ~ 1 : 5 bi-allelic mutations ● Familial HLH (FHL) ● Infectious agents („Farquhar`s disease“) Perforin UNC 13 D Syntaxin 11 UNC 18 B ● Immune deficiency syndromes Chédiak-Higashi syndrome 1 Griscelli syndrome 2 X-linked proliferative syndrome Other inborn immune deficiencies HLH ● Endogenous products - tissue damage - radical stress - metabolic products ● Autoimmune/autoinflammatory diseases (Macrophage activation syndrome; MAS) Malignant diseases ● Acquired immune defects

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Principles of treatment Treatment for HLH depends on the underlying disease and severity of symptoms. Children with MAS are usually treated successfully with high-dose corticosteroids ± ciclosporin A. In non-responders anakinra is effective. HLH cases with less severe symptoms may only need corticosteroids ± immunoglobulins – but be aware of the dynamics of the disease!

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Principles of treatment HLH therapy is a two-sided sword On one hand hyperinflammation has to be suppressed to prevent the dangerous consequences of hyperinflammation; on the other hand immunesuppressive and cytostatic treatment may be counterproductive for control of infectious triggers and recovery of the bone marrow. In contrast to malignant diseases the aim is not to destroy as many cells as possible by intensive therapy but just to down-regulate the hyperactive immune response and to reduce the number of activated (infected) cells in the hope to achieve a new balance.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Principles of treatment Suppression of hyperinflammation corticosteroids immunoglobulins cyclosporin A anti-cytokine treatments antibodies Elimination of (infected) cells: APCs, lymphocytes etoposide, rituximab, corticosteroids antithymocyte globulin/alemtuzumab Treatment of the trigger Replacement of the defective immune system by stem cell transplantation in genetic cases

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Treatment Immunosuppressive/immunomodulatory agents - Corticosteroids - Intravenous immunoglobulins Cyclosporin A Cytokine removal (plasmapheresis, cytokine-absorbers) Antagonists of single cytokines: IL-1, IL-6, INFγ Inhibition of Janus kinase 1/2: ruxolitinib Antibody against IL-2 receptor: basiliximab Cytotoxic drugs - Corticosteroids - Etoposide (Ambruso 1980) - T-and B-cell antibodies (ATG, alemtuzumab, rituximab)

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Founding of the Histiocyte Society in 1985 Founding of the HLH Study Group in 1989 Founding members Maurizio Aricò Jan-Inge Henter Blaise Favara Diane Komp Jon Pritchard Christian Nezelof Göran Elinder Gritta Janka

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Two international studies: HLH-1994 and HLH-2004 Follow-up publication by Trottestam H, Blood 2011
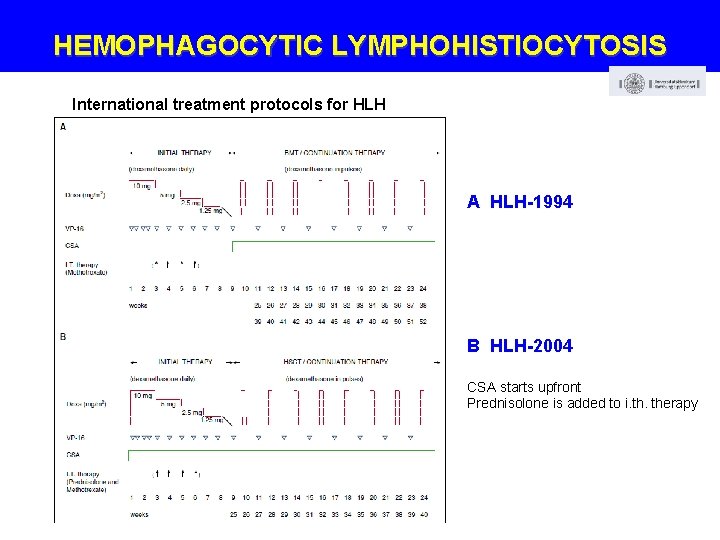
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS International treatment protocols for HLH A HLH-1994 B HLH-2004 CSA starts upfront Prednisolone is added to i. therapy
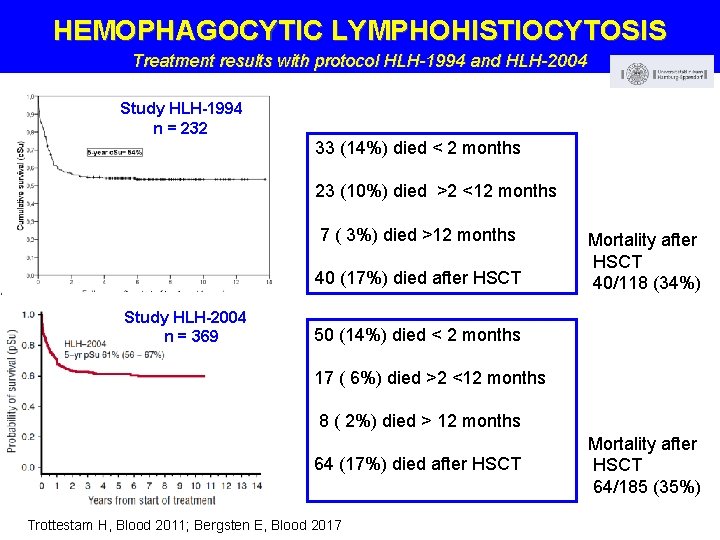
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Treatment results with protocol HLH-1994 and HLH-2004 Study HLH-1994 n = 232 33 (14%) died < 2 months 23 (10%) died >2 <12 months 7 ( 3%) died >12 months 40 (17%) died after HSCT Study HLH-2004 n = 369 Mortality after HSCT 40/118 (34%) 50 (14%) died < 2 months 17 ( 6%) died >2 <12 months 8 ( 2%) died > 12 months 64 (17%) died after HSCT Trottestam H, Blood 2011; Bergsten E, Blood 2017 Mortality after HSCT 64/185 (35%)
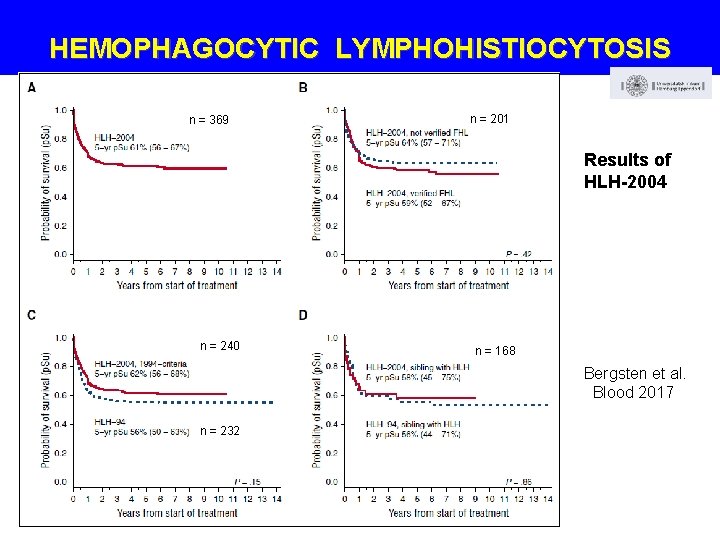
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS n = 369 n = 201 Results of HLH-2004 n = 369 n = 168 n = 240 n = 168 Bergsten et al. Blood 2017 n = 232

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS HLH Steering Committee of the Histiocyte Society Recommendations for the Use of Etoposide-Based Therapy and Bone Marrow Transplantation for the Treatment of HLH Ehl S. et al. Journal of Allergy Clinical Immunology Practice 2018 Recommendation of HLH Study Group to use the HLH-94 protocol with HLH-2004 diagnostic criteria

Authors of „Recommendations for use of protocol HLH-1994 Anna. Carin Horne Elena Sieni Jan-Inge Henter Michael Jordan Paul La Rosée Itziar Astigarraga Eiichi Ishii Stephan Ehl Melissa Hines Kim Nichols Tatiana Greenwood Kai Lehmberg Zhao Wang Gritta Janka Rafal Machowicz
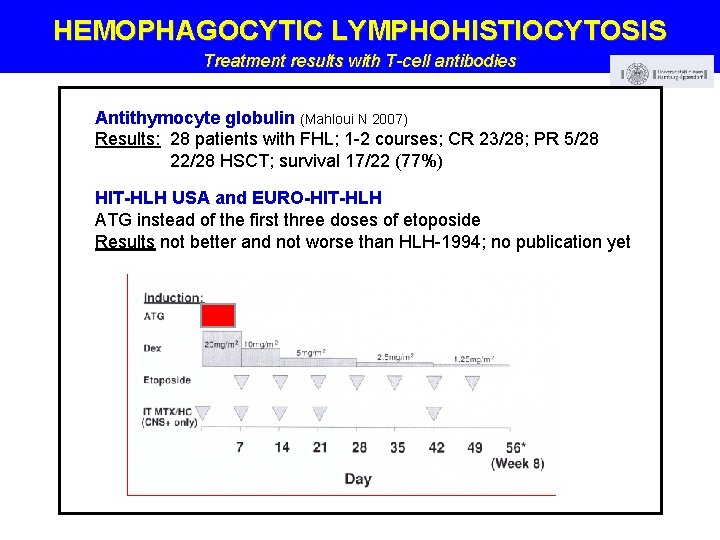
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Treatment results with T-cell antibodies Antithymocyte globulin (Mahloui N 2007) Results: 28 patients with FHL; 1 -2 courses; CR 23/28; PR 5/28 22/28 HSCT; survival 17/22 (77%) HIT-HLH USA and EURO-HIT-HLH ATG instead of the first three doses of etoposide Results not better and not worse than HLH-1994; no publication yet

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Treatment results with T-and B-cell antibodies Alemtuzumab Pilot study (2006 -2015) (Moushous, HS meeting 2016) 23 patients with FHL, 1 -3 courses Results: CR: 87%, PR 9%, 1 nonresponder; 22/23 patients HSCT Ongoing study (NCT 02472954) (Moushous, HS meeting 2018) so far 20 patients with FHL; planned 30; Campath d 1 0. 5 mg/kg; d 2+3 1 mg/kg + Pred d 4 -14 2 mg/kg, then taper, + CSA same response rate; ~50% received haplo-BMT Rituximab (Chellapandian, Rituximab Study Group 2013) Retrospective survey; 42 patients with EBV-HLH; median 3 doses (2/3 within 1 mos after diagnosis; combination with other HLH drugs) Results: significant decrease in EBV load and ferritin; significant increase in platelets and SGOT.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Salvage therapy Literature survey by Working Group on Second-line Treatment in HLH (R. Marsh, PBC 2017) Anakinra: 3 patients with MAS (3 CR) Patients with refractory/relapsed HLH, previously treated with steroids + etoposide or steroids + ATG. At least 2 patients treated. (Behrens E, Miettunen PM ) ATG: 2 patients with FHL (2 CR) (Mahlaoui N) Alemtuzumab: 24 patients with HLH (16 PR, 8 NR) (Marsh R, Strout MP, Gerard LM) Liposomal doxorubin, etoposide and methyl-prednisolone Thirty-four adult patients without lymphoma: 12 CR, 14 PR, 8 NR (Wang Z)

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS New drugs Interferon-γ antibody Emapalumab (NI-0501) NCT 01818492: phase II/III study, 20 countries, still recruiting Inclusion: children with primary HLH; reactivated disease, or unsatisfactory response, or intolerant to standard therapy Exclusion: secondary HLH, HLH in rheumatic or malignant diseases Treatment: antibody 1 mg/kg every 3 days + dexamethasone Results: Eleven of 13 patients alive after 8 weeks (1 first-line treatment) - Satisfactory response in 9/13 patients (improvement in HLH parameters) - Seven of 13 patients proceeded to HSCT - Resolution of CNS symptoms in 2 evaluable patients - Reduction of DEX in 50% of patients in first 4 weeks. Update HS meeting 2016: 31 patients recruited, 25 with mutations; 5 first line-treatment Jordan M, Blood LBA 2015

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS New drugs Ruxolitinib is an oral anti-inflammatory and immunosuppressive Janus kinase (JAK) inhibitor, approved for myelofibrosis. Activity has been shown in rheumatoid arthritis, Gv. HD, psoriasis, ulcerative colitis, and other diseases. JAK 1 and JAK 2 control signaling of many cytokines by transmitting cytokine-induced signals, notably from INF-γ, IL-2 and IL-6. Ruxolitinib is being evaluated in 2 ongoing clinical trial for HLH in adults). There are several recent case reports.

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Ruxolitinib Das et al. (Kim Nichols` group) Blood 2016 Two mouse models for HLH: PRF 1 -/- mouse and Cp. G induced model for secondary HLH Preemptive treatment with ruxolitinib starting day 4 (to day 9) Prevention of HLH symptoms, hypercytokinemia and tissue inflammation, enhancement of survival Maschalidi et al. (Alain Fischer`s group) Blood 2016 Two mouse models for HLH: PRF 1 -/- mouse and RAB 27 a -/Treatment of manifest HLH with ruxolitinib for 14 days starting day 7 (PRF 1), or day 10 (RAB 27 A) post infection Enhancement of survival (comparable with INF-γ antibody treated mice) Correction of cytopenias, reduction of tissue inflammation, reversal of CNS symptoms (RAB 27 A-/- mouse)
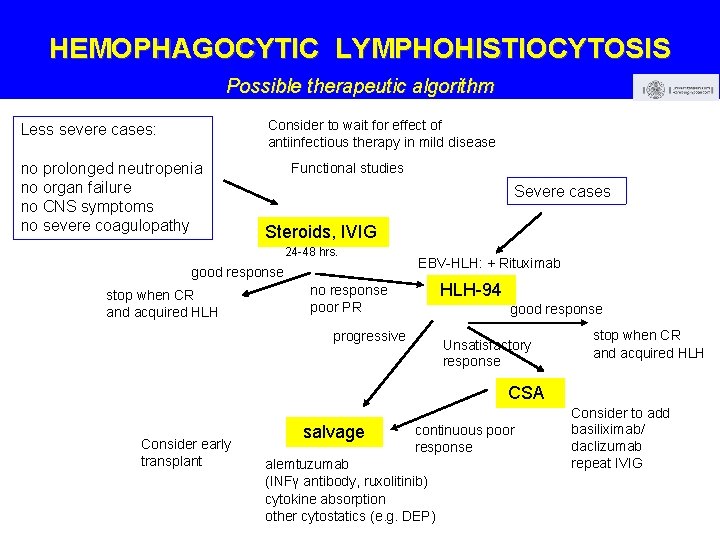
HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Possible therapeutic algorithm Consider to wait for effect of antiinfectious therapy in mild disease Less severe cases: no prolonged neutropenia no organ failure no CNS symptoms no severe coagulopathy Functional studies Severe cases Steroids, IVIG 24 -48 hrs. good response stop when CR and acquired HLH EBV-HLH: + Rituximab HLH-94 no response poor PR good response progressive Unsatisfactory response stop when CR and acquired HLH CSA Consider early transplant salvage continuous poor response alemtuzumab (INFγ antibody, ruxolitinib) cytokine absorption other cytostatics (e. g. DEP) Consider to add basiliximab/ daclizumab repeat IVIG

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Stem cell transplantation: Reduced intensity conditioning Author number conditioning regime survival Horne A, BJH 2005 86 BU, CYT, VP 16 ± ATG 64% Cooper N BMT 2008 25 5 -FU, MEL, alemtuzumab 84% (2 -105 mos, med 36) Marsh R Blood 2010 26 5 -FU, MEL, alemtuzumab p. OS 92% (at 3 years) Lehmberg K Haemat. 2013 19 60 5 -FU, TREO, THIO (14), alemtu 100% (7 -31, med 16 mos) manuscript in preparation 78% Allen C, Blood 2018 5 -FU, MEL, alemtuzumab 34 68% at 18 mos RIC conditioning is associated with less toxicity and better survival, but a with a higher frequency of mixed chimerism, necessitating intervention with DLIs or second transplants

HEMOPHAGOCYTIC LYMPHOHISTIOCYTOSIS Stem cell transplantation with RIC: mixed donor chimerisms (DC) (Hartz B HS Blood 2016) 103 patients with FHL from 7 countries, DC < 75%; overall survival 82% 17 (16%) reactivations (10 before d 180); overall survival 9/17 18 pts. 2 nd transplants (9 with previous reactivations); mortality 33% Key messages: ● Patients with reactivations before d 180 have variable DC and frequently viral triggers; underlying immunosuppression plays a role. ● A DC of 20 -30% protects from reactivation after d 180 ● DC below 30% does not always result in reactivation of HLH ► 5 pts. line-specific DC (CD 3) <10% 1. 1. -10 yrs (med. 5. 1) without relapse ● In patients with low DC, risk of 2 nd HSCT must be weighed against risk of reactivation

Thank you for your attention! Our HLH Team Hamburg: Kai Lehmberg Udo zur Stadt Ingo Müller Anke Clodius Manuela Adao Gritta Janka München: Karin Beutel Freiburg: Stephan Ehl Miriam Heizmann Carsten Speckmann Sandra Amman Ilka Fuchs

- Slides: 29