5 Phonons Thermal Properties Phonon Heat Capacity Anharmonic






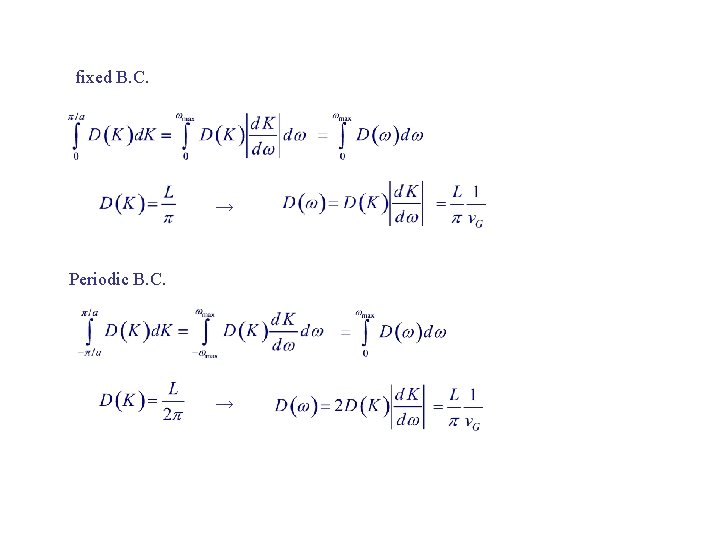








- Slides: 29

5. Phonons Thermal Properties • Phonon Heat Capacity • Anharmonic Crystal Interactions • Thermal Conductivity

Phonon Heat Capacity • • Planck Distribution Normal Mode Enumeration Density of States in One Dimension Density of States in Three Dimensions Debye Model for Density of States Debye T 3 Law Einstein Model of the Density of States General Result for D(ω)

where = Thermal expansivity = isothermal compressibility = 1/ B B = Bulk modulus α = linear (1 -D) thermal expansivity

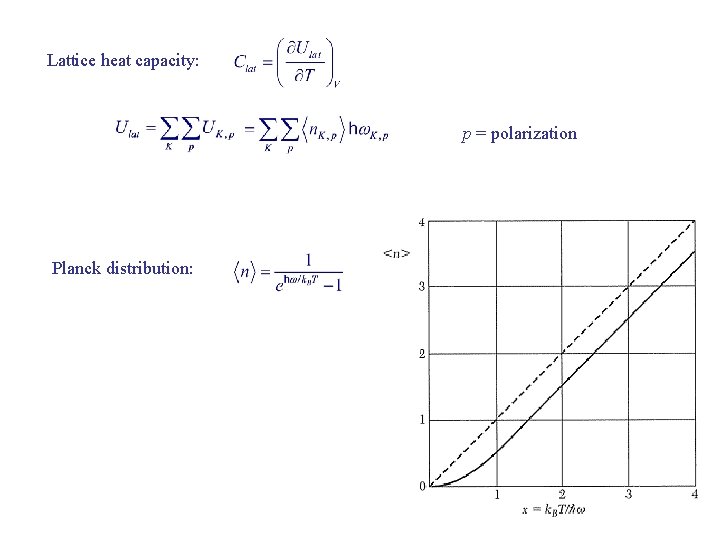
Lattice heat capacity: p = polarization Planck distribution:

Planck Distribution System at constant T Canonical ensemble : Boltzmann factor For a set of identical harmonic oscillators Nn = number of oscillators in the nth excited state when system is in thermal equilibrium Probability of an oscillator in the nth excited state: Occupation number:

Normal Mode Enumeration

Density of States in One Dimension Fixed boundary problem of N+1 particles. N = 10 → with Number of allowed K for non-stationary solutions is N– 1 = Number of mobile atoms Polarization p : 1 long, 2 trans

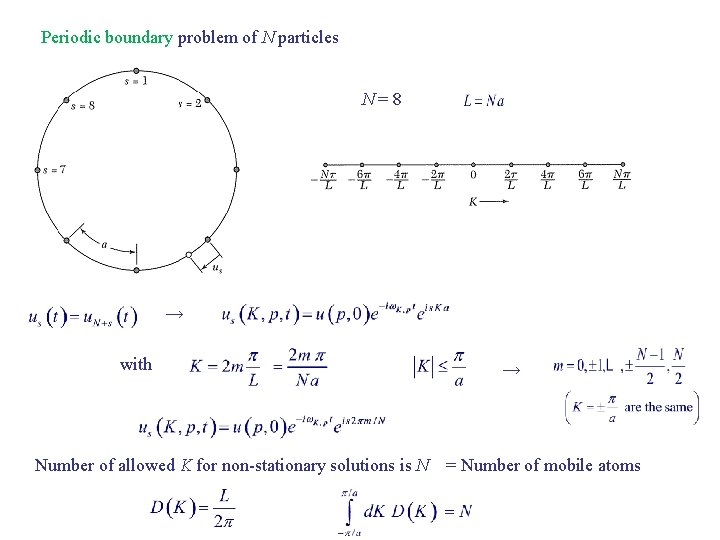
Periodic boundary problem of N particles N=8 → with → Number of allowed K for non-stationary solutions is N = Number of mobile atoms
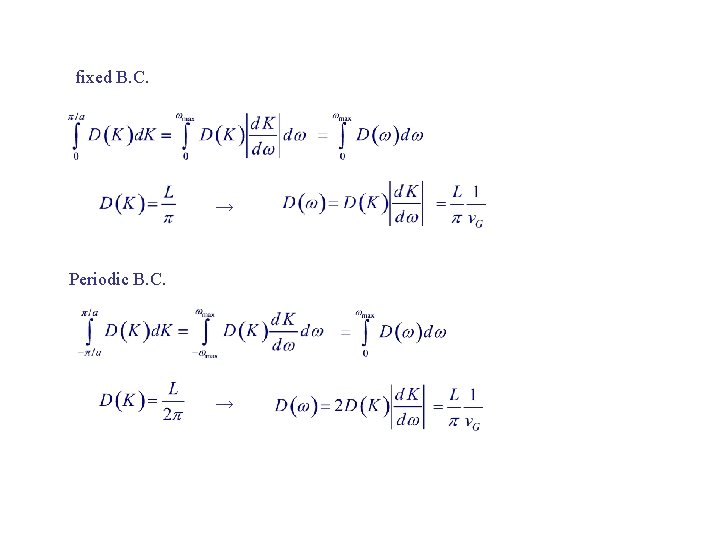
fixed B. C. → Periodic B. C. →

Density of States in Three Dimensions Periodic B. C. ; N 3 cells in cube of side L → density of states in K-space is Number of modes per polarization lying between ω and ω + d ω is → density of states in ω-space is For isotropic materials, →

Debye Model for Density of States Debye model: v velocity of sound (for a given type of polarization) For a crystal of N primitive cells: → Debye frequency

Thermal (vibrational) energy Debye integrals: See Ex on Zeta functions, Arfken where θ Debye temperature for each acoustic branch

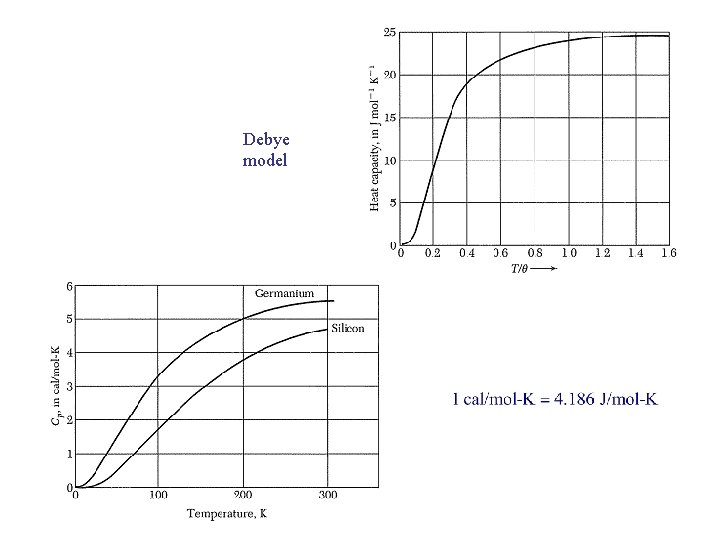
Debye model

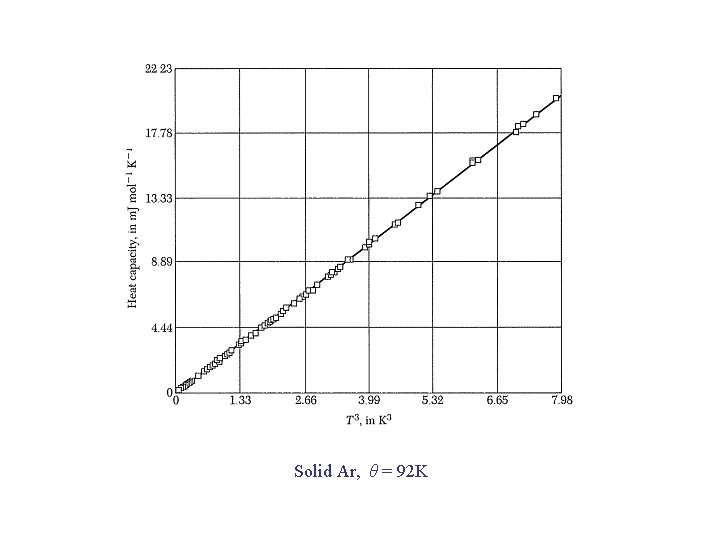
Debye T 3 Law For low T, x. D → : → for each acoustic branch To account for all 3 acoustic branches, we set and so that Good for T < θ /50

Solid Ar, θ = 92 K

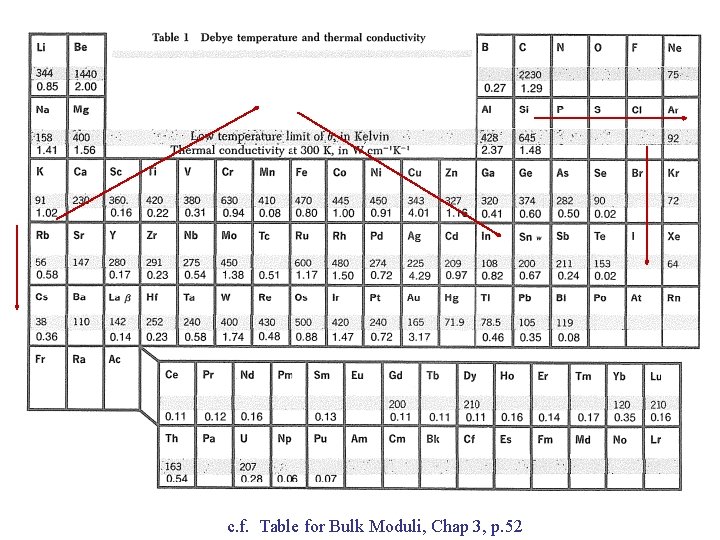
c. f. Table for Bulk Moduli, Chap 3, p. 52

Qualitative Explanation of the T 3 Law Of the 3 N modes, only a fraction (KT /KD ) 3 = ( T / θ )3 is excited. →

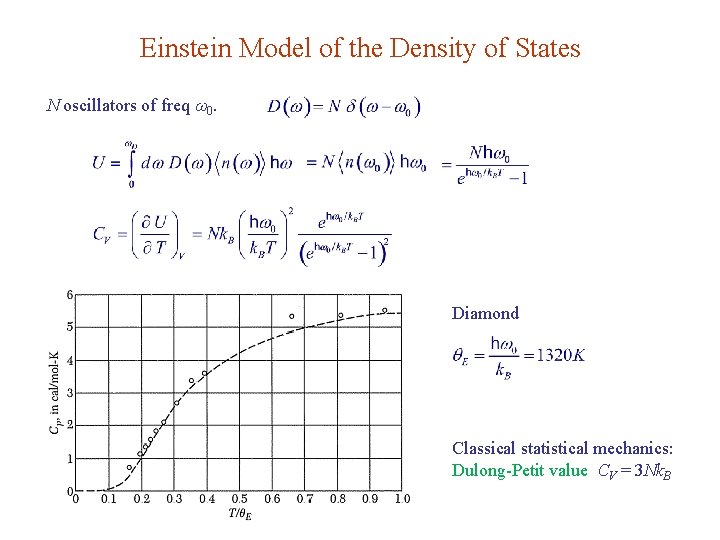
Einstein Model of the Density of States N oscillators of freq ω0. Diamond Classical statistical mechanics: Dulong-Petit value CV = 3 Nk. B

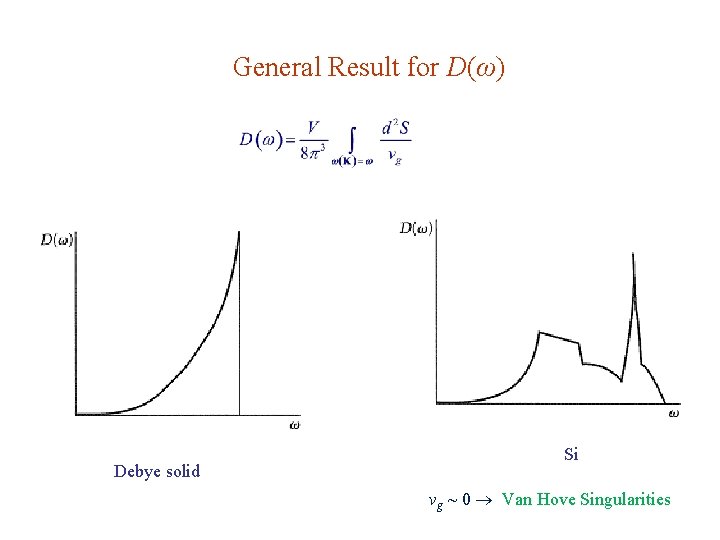
General Result for D(ω) Debye solid Si vg ~ 0 Van Hove Singularities

Anharmonic Crystal Interactions Harmonic (Linear) Waves: • Normal modes do not decay. • Normal modes do not interact. • No thermal expansion. • Adiabatic & isothermal elastic constants are equal. • Elastic constants are independent of P and T. • C → constant for T > θ. Deviation from harmonic behavior → Anharmonic effects

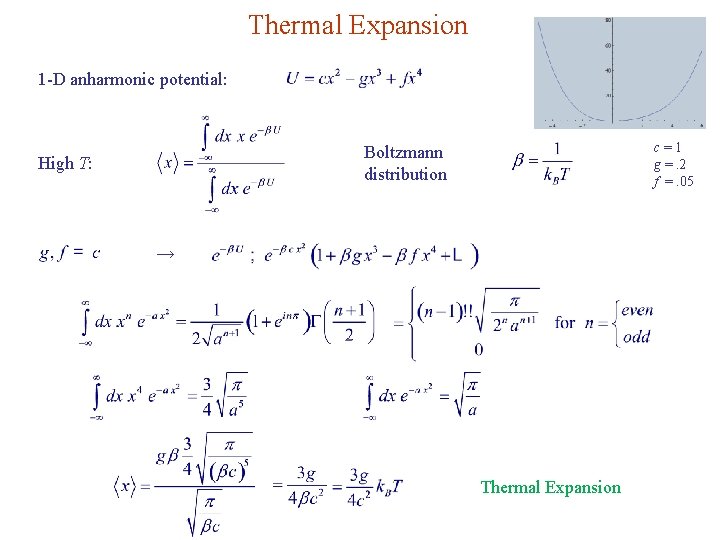
Thermal Expansion 1 -D anharmonic potential: c=1 g =. 2 f =. 05 Boltzmann distribution High T: → Thermal Expansion

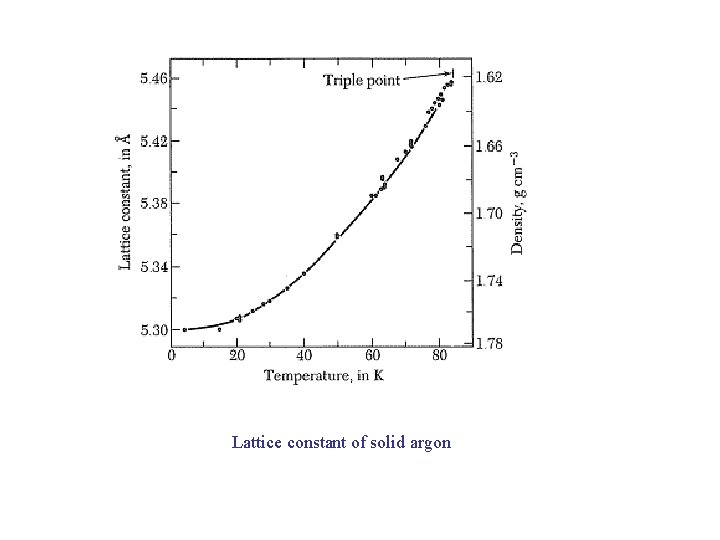
Lattice constant of solid argon

Thermal Conductivity Heat current density: κ = Thermal conductivity coefficient For phonons, JQ = JU. Key features of kinetic theory (see L. E. Reichl, “A Modern Course of Statistical Mechanics”, § 13. 4 ): 1. Quantities not conserved in particle collisions are quickly thermalized to (global) equilibrium values. (e. g. , velocity directions & magnitudes ) 2. Conserved quantities can remain out of global equilibrium (e. g. , stay in local equilibrium. They get transported spatially in the presence of a “gradient”. 3. MFP is determined by collisions that do not conserve the total momenta of particles. Net amount of A(z) transported across the x-y plane at z 0 in the +z direction per unit area per unit time: Δz = distance above/below plane at which particle suffered last collision. n = particle density, l = mean free path, a = some constant. b. A = 1/3 is determined from self diffusion

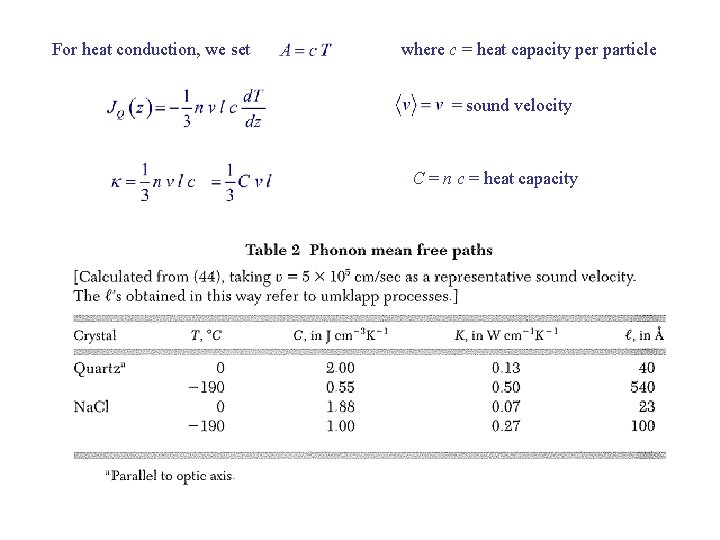
For heat conduction, we set where c = heat capacity per particle = sound velocity C = n c = heat capacity

Thermal Resistivity of Phonon Gas Harmonic phonons: mfp l determined by collisions with boundaries & imperfections. Anharmonic phonons: only U-processes contribute.

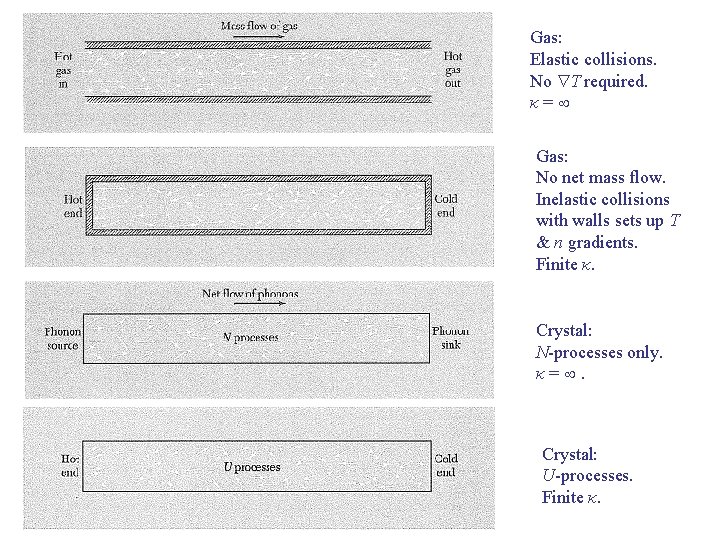
Gas: Elastic collisions. No T required. κ= Gas: No net mass flow. Inelastic collisions with walls sets up T & n gradients. Finite κ. Crystal: N-processes only. κ=. Crystal: U-processes. Finite κ.

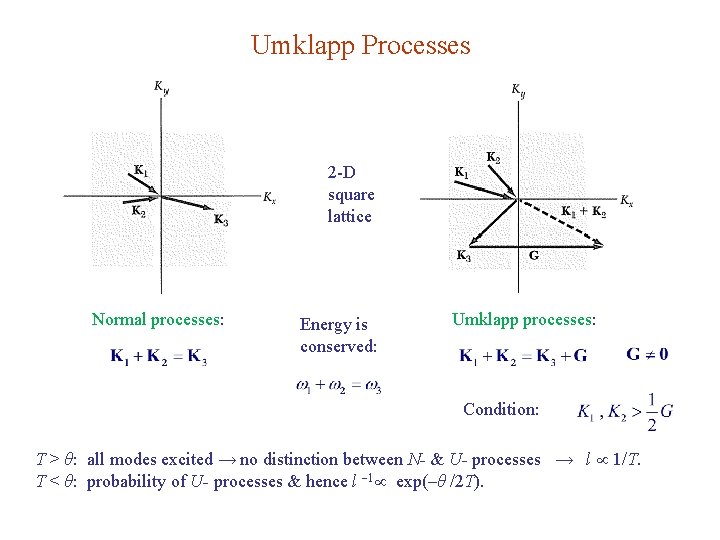
Umklapp Processes 2 -D square lattice Normal processes: Energy is conserved: Umklapp processes: Condition: T > θ: all modes excited → no distinction between N- & U- processes → l 1/T. T < θ: probability of U- processes & hence l 1 exp(–θ /2 T).

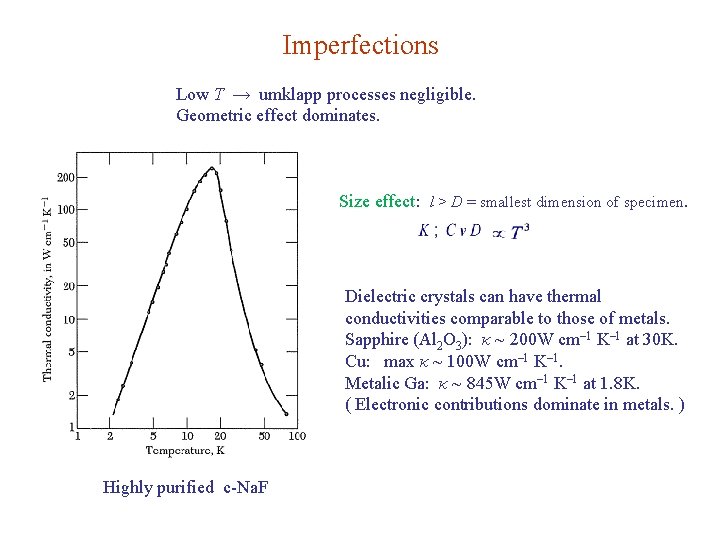
Imperfections Low T → umklapp processes negligible. Geometric effect dominates. Size effect: l > D = smallest dimension of specimen. Dielectric crystals can have thermal conductivities comparable to those of metals. Sapphire (Al 2 O 3): κ ~ 200 W cm– 1 K– 1 at 30 K. Cu: max κ ~ 100 W cm– 1 K– 1. Metalic Ga: κ ~ 845 W cm– 1 K– 1 at 1. 8 K. ( Electronic contributions dominate in metals. ) Highly purified c-Na. F

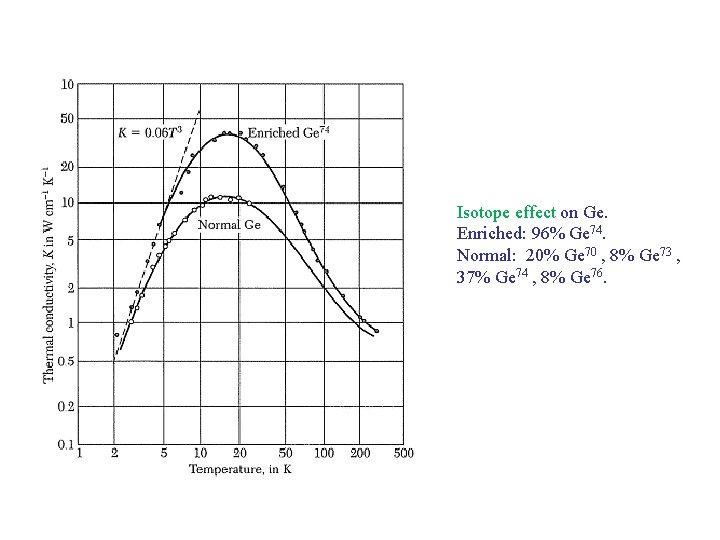
Isotope effect on Ge. Enriched: 96% Ge 74. Normal: 20% Ge 70 , 8% Ge 73 , 37% Ge 74 , 8% Ge 76.