10292020 1 I Introduction II Classification of chromatographic















- Slides: 35

10/29/2020 1

I. Introduction II. Classification of chromatographic methods III. Principle of chromatography IV. High performance liquid chromatography (HPLC) 10/29/2020 V. Gas chromatography (GC) VI. Thin layer chromatography (TLC) 2

Definition: Chromatography is defined as a procedure by which solutes are separated by dynamic differential migration process in a system consisting of two or more phases, one of which moves continuously in a given direction and in which the individual substances exhibit different mobilities by reason of differences in adsorption, partition, solubility, vapor pressure, molecular size, or ionic charge density. 10/29/2020 3

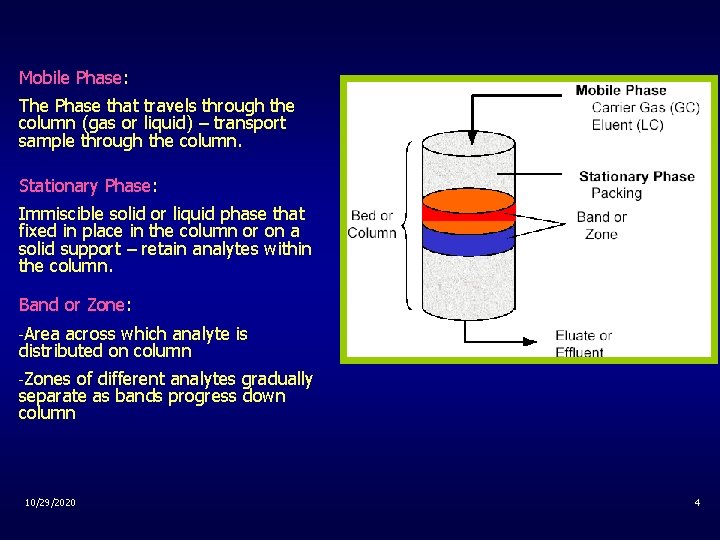
Mobile Phase: The Phase that travels through the column (gas or liquid) – transport sample through the column. Stationary Phase: Immiscible solid or liquid phase that fixed in place in the column or on a solid support – retain analytes within the column. Band or Zone: -Area across which analyte is distributed on column -Zones of different analytes gradually separate as bands progress down column 10/29/2020 4

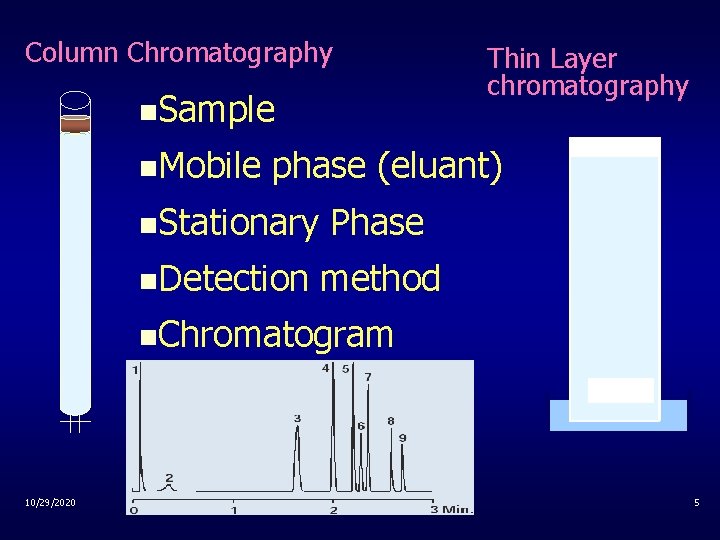
Column Chromatography n. Sample n. Mobile Thin Layer chromatography phase (eluant) n. Stationary n. Detection Phase method n. Chromatogram 10/29/2020 5

n n Method to separate components in a mixture based on different Distribution coefficients between the two phases. Chromatography categorized on the basis of interaction between solute and stationary phase Mobile phase either gas or liquid Stationary phase either liquid or solid – Liq/Liq (Partition) Liquid – Liq/Sol (Adsorption) Chromatography – Gas/Liq (Partition) – Gas/Sol (Adsorption) 10/29/2020 Gas Chromatography 6

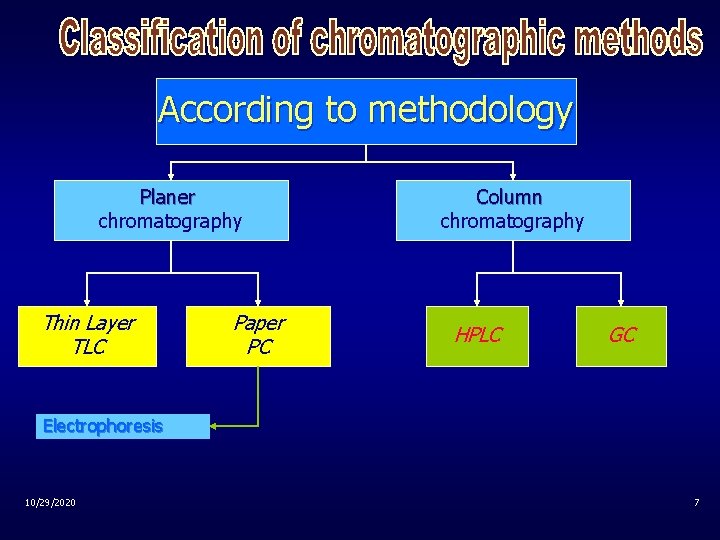
According to methodology Planer chromatography Thin Layer TLC Paper PC Column chromatography HPLC GC Electrophoresis 10/29/2020 7

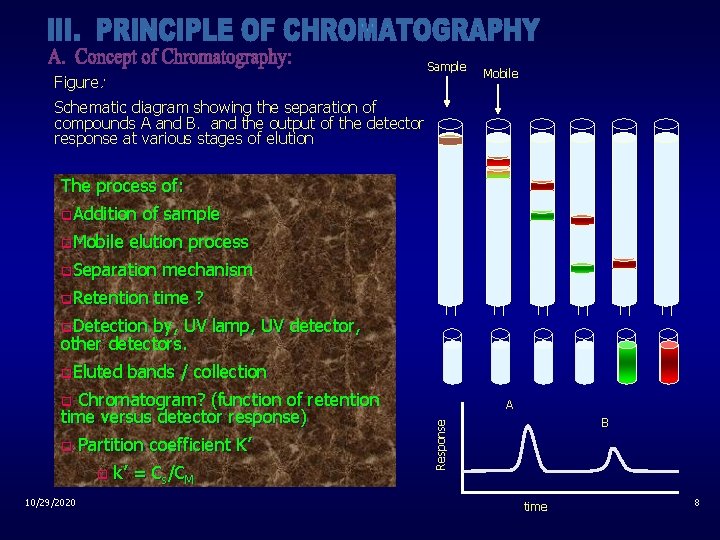
Sample Figure: Mobile Schematic diagram showing the separation of compounds A and B. and the output of the detector response at various stages of elution The process of: q. Addition q. Mobile of sample elution process q. Separation q. Retention mechanism time ? q. Detection by, UV lamp, UV detector, other detectors. q. Eluted bands / collection Chromatogram? (function of retention time versus detector response) q Partition coefficient K’ q 10/29/2020 k’ = Cs/CM B Response q A time 8

10/29/2020 9

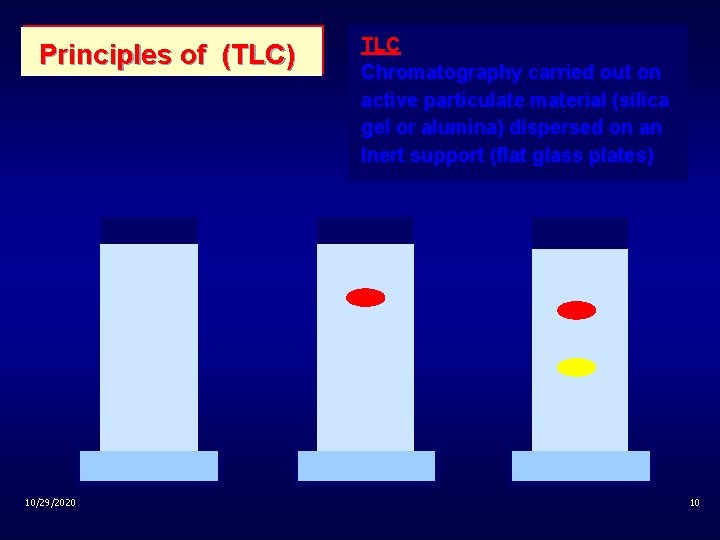
Principles of (TLC) 10/29/2020 TLC Chromatography carried out on active particulate material (silica gel or alumina) dispersed on an Inert support (flat glass plates) 10

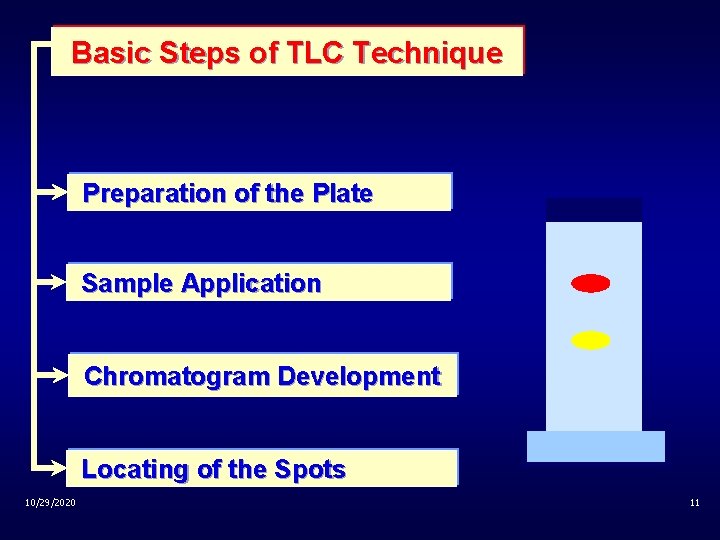
Basic Steps of TLC Technique Preparation of the Plate Sample Application Chromatogram Development Locating of the Spots 10/29/2020 11

Preparation of the Plate § Slurry of the active material is uniformly spread over the plate by means of a commercially available spreader. § Air-drying overnight, or oven-drying at 80 -90 C for about 30 minutes. § Ready to use thin layers (pre-coated plates) are commercially available. 10/29/2020 12

Sample Application 1 -2 cm Base line 2 -2. 5 cm 10/29/2020 13

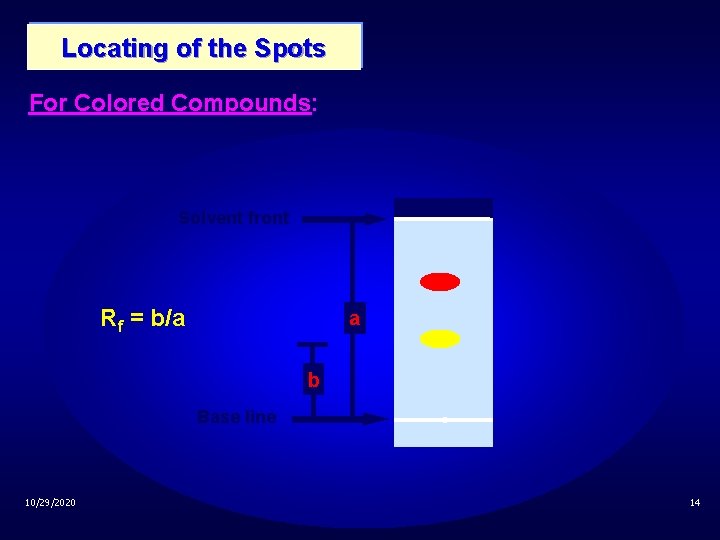
Locating of the Spots For Colored Compounds: Solvent front Rf = b/a a b Base line 10/29/2020 14

For Colorless Compounds: Where is the spots ? ? We do not know. Solvent front Rf = b/a a b Base line • Iodine or sulphuric acid is used for most organic mixtures. • Ninhydrin is used for amino acids. 10/29/2020 • 2, 4 -Dinitrophenylhydrazine is used for aldehydes and ketones 15

Applications of TLC Technique Co-spo t Authentic Unknown Co-spo t Authentic 10/29/2020 Unknown Identification of Unknown Compounds 16

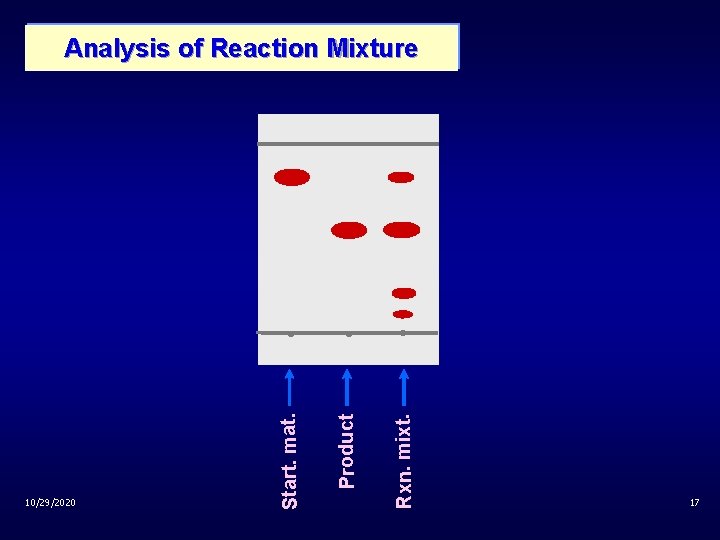
Product Rxn. mixt. 10/29/2020 Start. mat. Analysis of Reaction Mixture 17

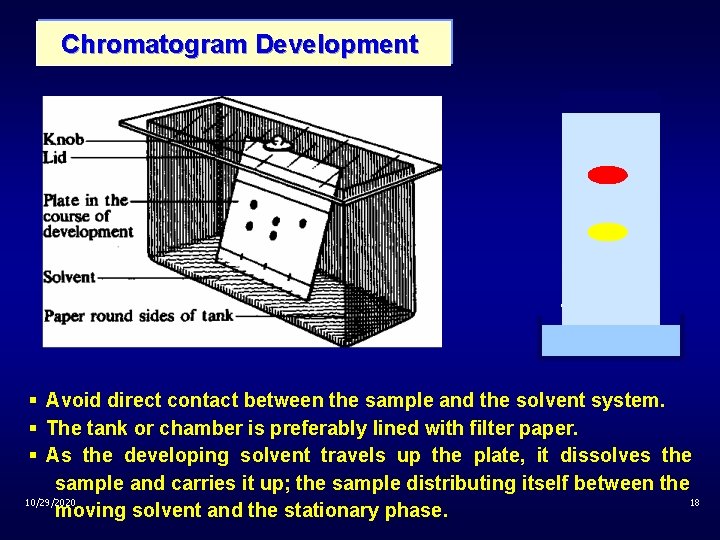
Chromatogram Development § Avoid direct contact between the sample and the solvent system. § The tank or chamber is preferably lined with filter paper. § As the developing solvent travels up the plate, it dissolves the sample and carries it up; the sample distributing itself between the 10/29/2020 18 moving solvent and the stationary phase.

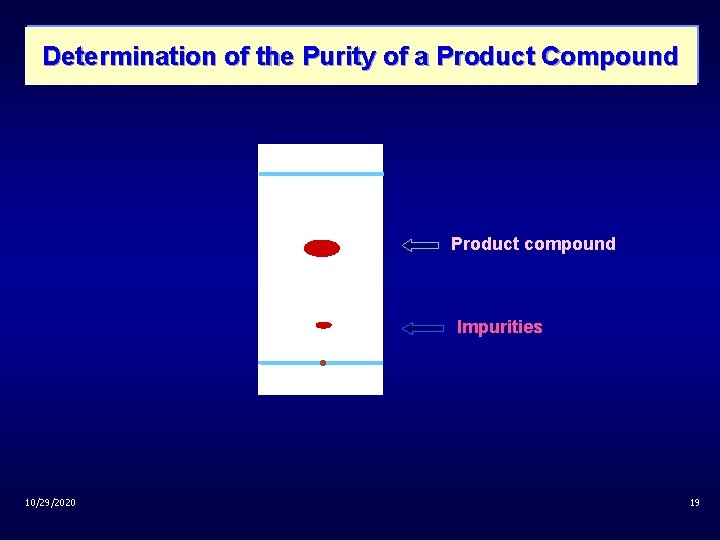
Determination of the Purity of a Product Compound Product compound Impurities 10/29/2020 19

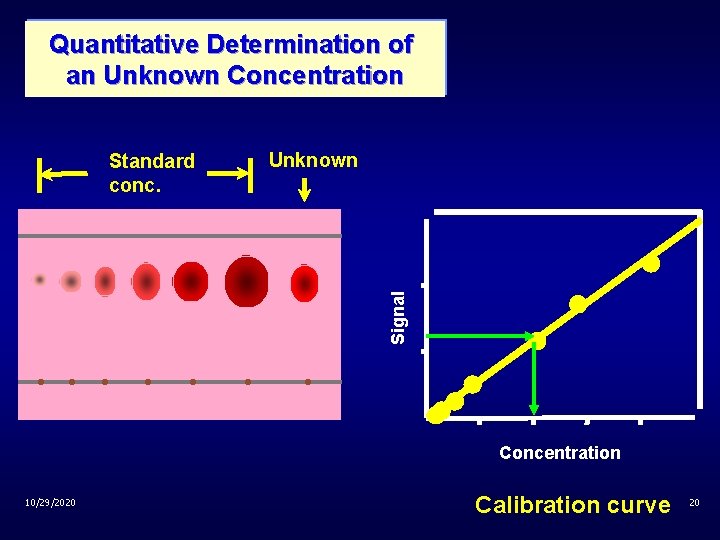
Quantitative Determination of an Unknown Concentration Unknown Signal Standard conc. Concentration 10/29/2020 Calibration curve 20

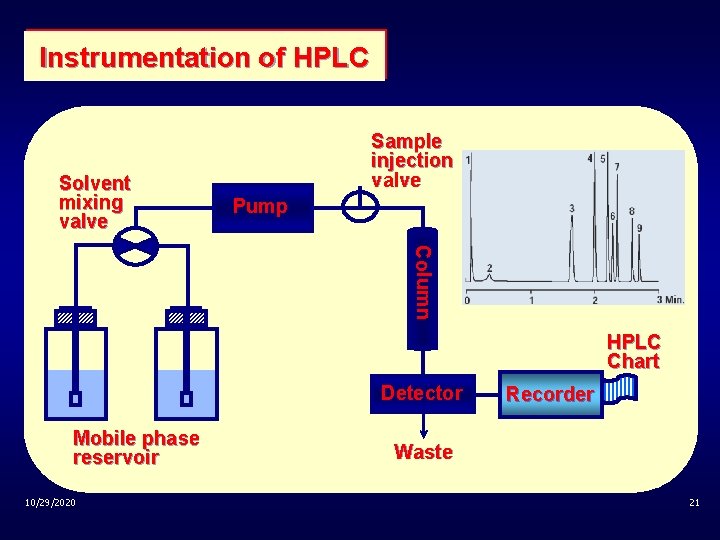
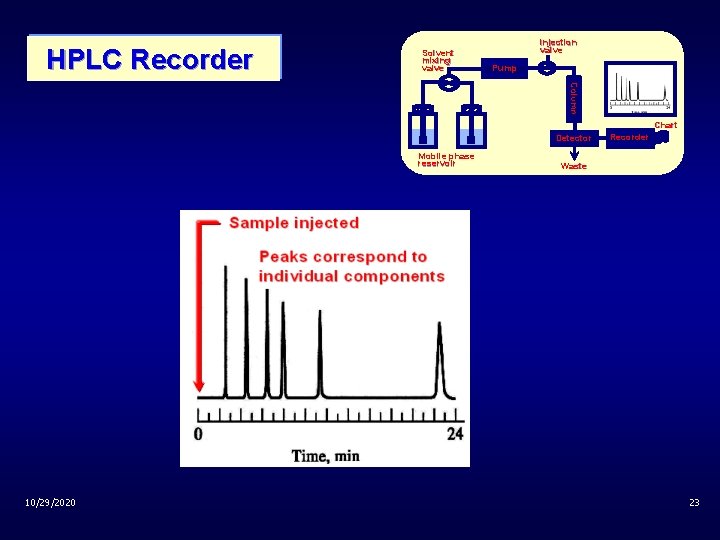
Instrumentation of HPLC Solvent mixing valve Sample injection valve Pump Column HPLC Chart Detector Mobile phase reservoir 10/29/2020 Recorder Waste 21

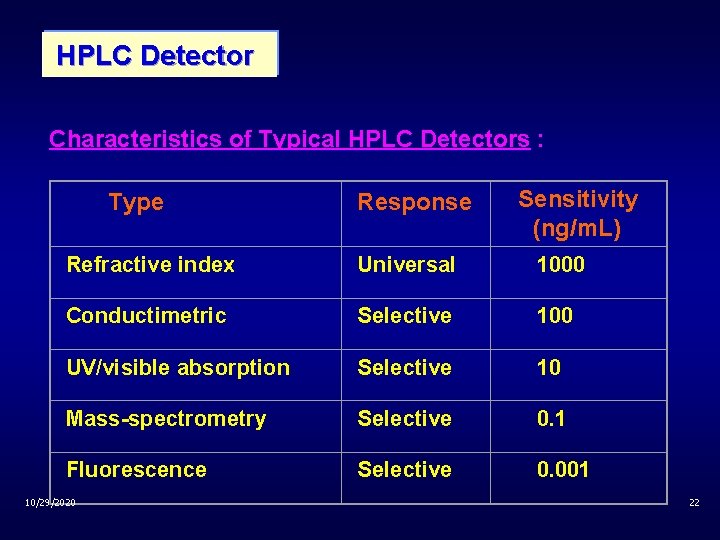
HPLC Detector Characteristics of Typical HPLC Detectors : Type Response Sensitivity (ng/m. L) Refractive index Universal 1000 Conductimetric Selective 100 UV/visible absorption Selective 10 Mass-spectrometry Selective 0. 1 Fluorescence Selective 0. 001 10/29/2020 22

HPLC Recorder Solvent mixing valve injection valve Pump Column Chart Detector Mobile phase reservoir 10/29/2020 Recorder Waste 23

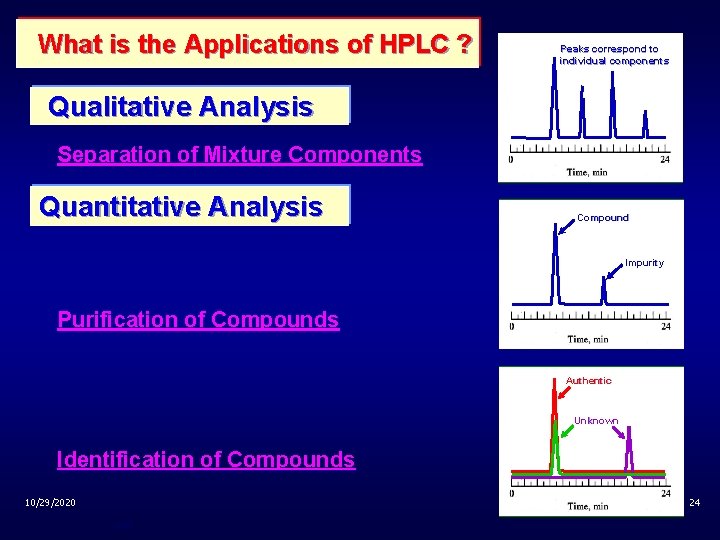
What is the Applications of HPLC ? Peaks correspond to individual components Qualitative Analysis Separation of Mixture Components Quantitative Analysis Compound Impurity Purification of Compounds Authentic Unknown Identification of Compounds 10/29/2020 24

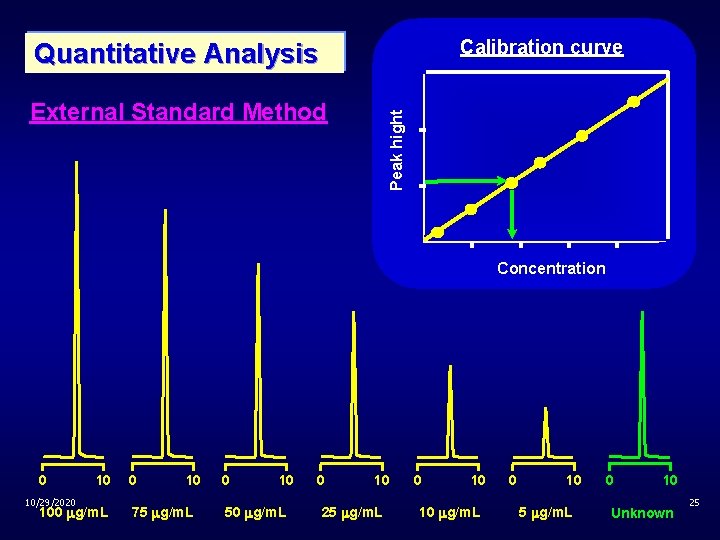
Calibration curve Quantitative Analysis Peak hight External Standard Method Concentration 0 10/29/2020 10 100 g/m. L 0 10 75 g/m. L 0 10 50 g/m. L 0 10 25 g/m. L 0 10 10 g/m. L 0 10 5 g/m. L 0 10 Unknown 25

G C 10/29/2020 26

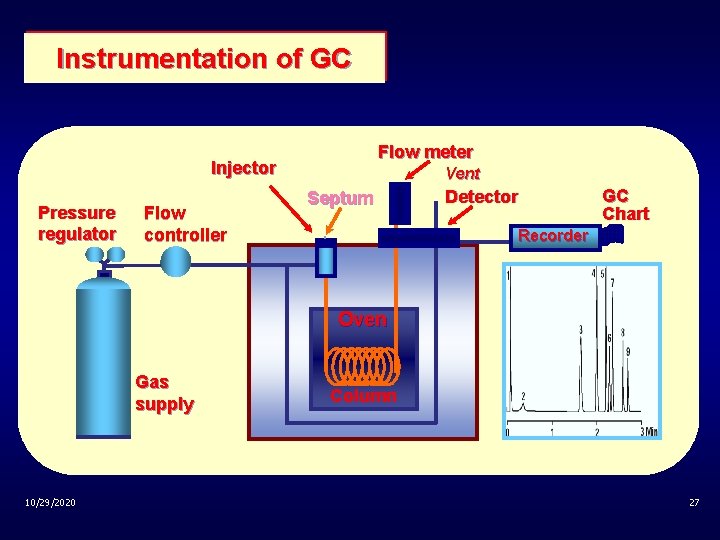
Instrumentation of GC Flow meter Injector Pressure regulator Flow controller Vent Septum Detector GC Chart Recorder Oven Gas supply 10/29/2020 Column 27

GC Column n Packed column * * n ~ 3 -6 mm inner diameter tubing, 1 -5 m long used for preparative separations or to separate gases that are poorly retained lower resolution small, uniform particle size decreases Eddy diffusion (requiring higher pressures) open tubular (more common): * * * 10/29/2020 0. 1 -0. 5 mm inner dia. , 10 -100 m long 0. 1 -5 mm thick sp coated on inner walls higher resolution, shorter analysis times, greater sensitivity compared to packed columns 28

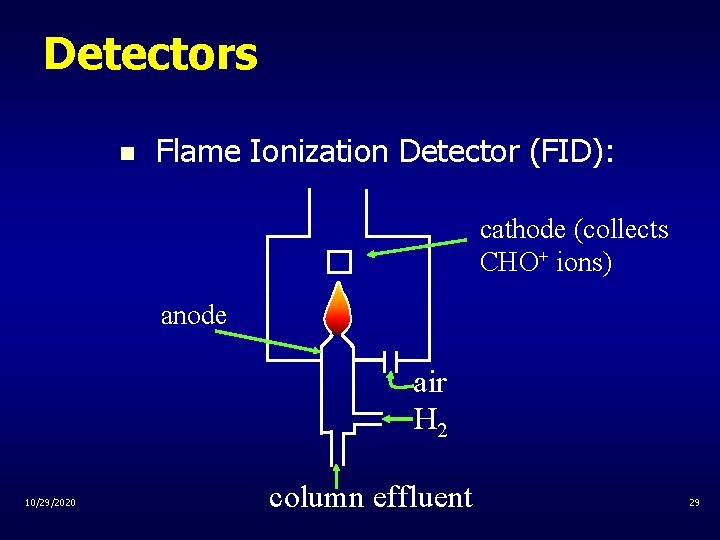
Detectors n Flame Ionization Detector (FID): cathode (collects CHO+ ions) anode air H 2 10/29/2020 column effluent 29

Detectors n Flame Ionization Detector (FID): – organic solutes are burned in flame producing CH radicals and eventually CHO+ . . – CH + O CHO+ + e– CHO+ ions are collected by cathode, produces current as the response 10/29/2020 30

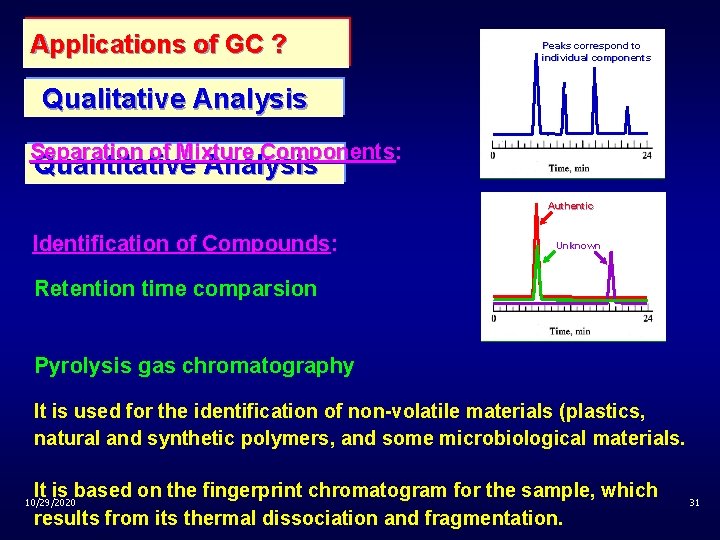
Applications of GC ? Peaks correspond to individual components Qualitative Analysis Separation of Mixture Components: Quantitative Analysis Authentic Identification of Compounds: Unknown Retention time comparsion Pyrolysis gas chromatography It is used for the identification of non-volatile materials (plastics, natural and synthetic polymers, and some microbiological materials. It is based on the fingerprint chromatogram for the sample, which results from its thermal dissociation and fragmentation. 10/29/2020 31

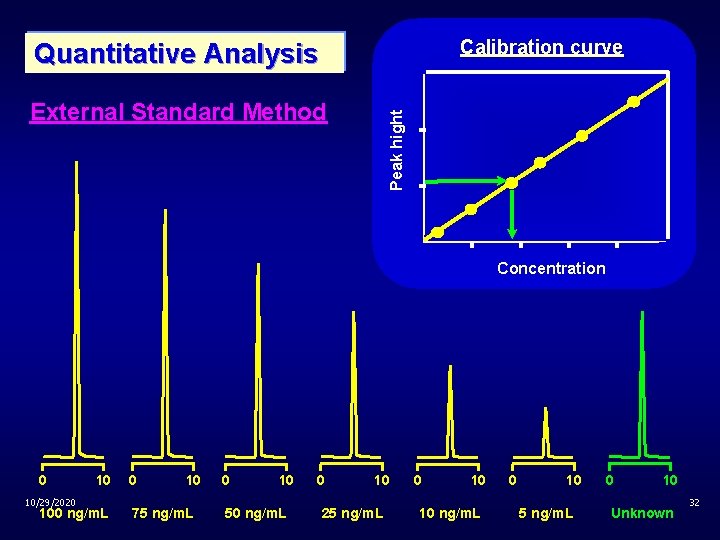
Calibration curve Quantitative Analysis Peak hight External Standard Method Concentration 0 10/29/2020 10 100 ng/m. L 0 10 75 ng/m. L 0 10 50 ng/m. L 0 10 25 ng/m. L 0 10 10 ng/m. L 0 10 5 ng/m. L 0 10 Unknown 32

Aspects of GC Applications: Food Analysis of foods is concerned with confirm the presence and determination the quantities of the analytes (lipids, proteins, carbohydrates, preservatives, flavours, colorants, and also vitamins, steroids, and pesticide residues). Drug Analysis GC is widely applied to identification of the active components, possible impurities as well as the metabolites. 10/29/2020 33

Environmental Analysis The environmental contaminants; e. g. dichlorodiphenyltrichloroethane (DDT) and the polychlorinated biphenyls (PCBs) are present in the environment at very low concentrations and are found among many of other compounds. GC, with its high sensitivity and high separating power, is mostly used in the analysis of environmental samples. Forensic Analysis In forensic cases, very little sample is available, and the concentration of the sample components may be very low. GC is a useful due to its high sensitivity and separation efficiency. 10/29/2020 34

10/29/2020 35