Trends in the Periodic Table and Bonding Periodic

Trends in the Periodic Table and Bonding Periodic Trends in Ionisation Energies and Covalent Radii

Ionisation Energy The ionisation energy is the energy required to remove one mole of electrons from one mole of atoms in the gaseous state. The first ionisation energy of magnesium: Mg (g) Mg+ (g) + e- 744 k. Jmol-1 Values for ionisation energies are given to you

Ionisation Energy The second ionisation energy of magnesium: Mg+ (g) Mg 2+ (g) + e- 1460 k. Jmol-1 The third ionisation enthalpy shows a massive increase because it requires an electron to be removed from magnesium’s second energy level. Mg 2+ (g) Mg 3+ (g) + e- 7750 k. Jmol-1
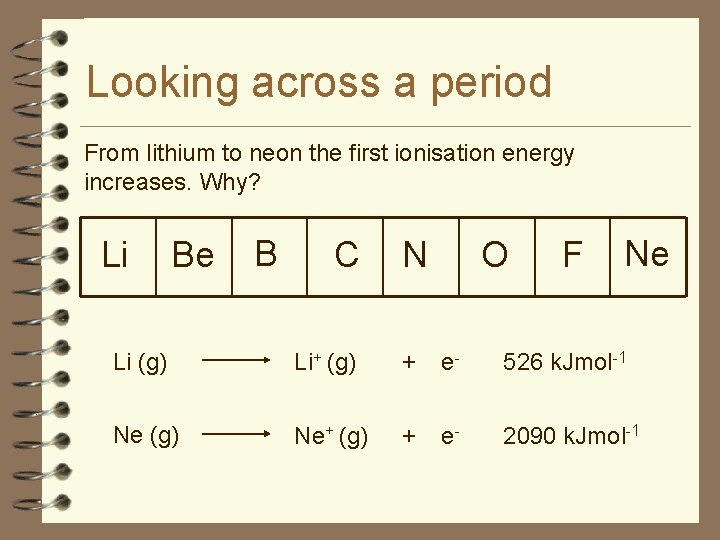
Looking across a period From lithium to neon the first ionisation energy increases. Why? Li Be B C N O F Ne Li (g) Li+ (g) + e- 526 k. Jmol-1 Ne (g) Ne+ (g) + e- 2090 k. Jmol-1
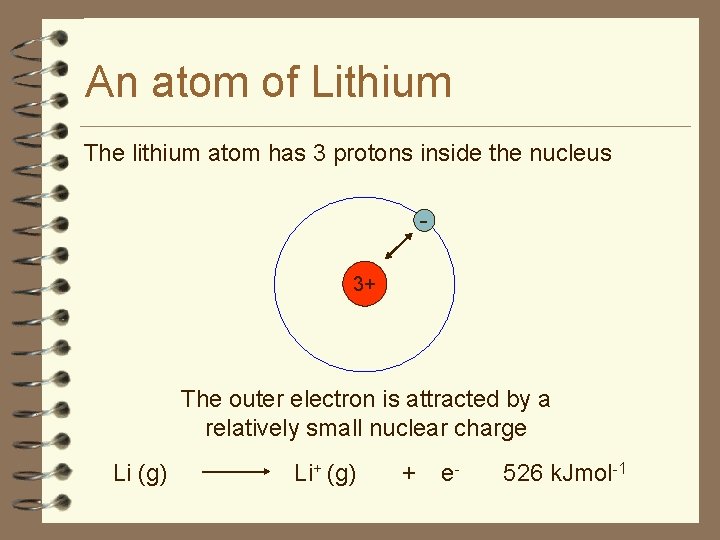
An atom of Lithium The lithium atom has 3 protons inside the nucleus 3+ The outer electron is attracted by a relatively small nuclear charge Li (g) Li+ (g) + e- 526 k. Jmol-1
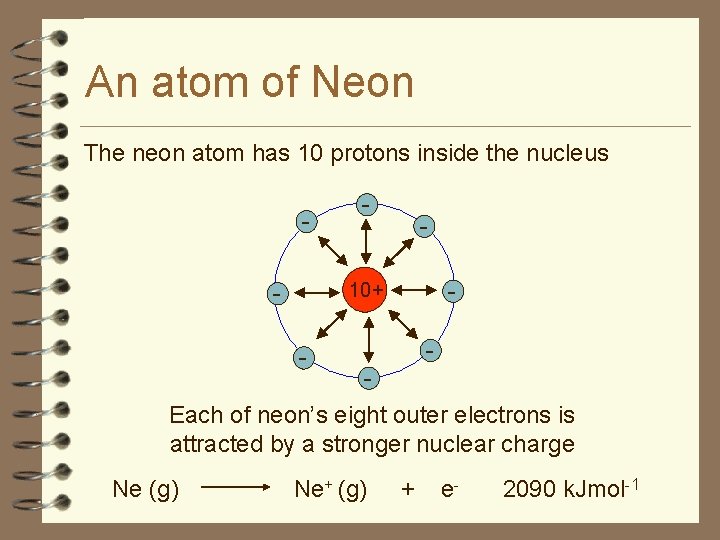
An atom of Neon The neon atom has 10 protons inside the nucleus - - - 10+ - - Each of neon’s eight outer electrons is attracted by a stronger nuclear charge Ne (g) Ne+ (g) + e- 2090 k. Jmol-1

Looking down a group The first ionisation energy decreases down a group in the periodic table. Why? Li (g) Li+ (g) + e- 526 k. Jmol-1 Cs (g) Cs+ (g) + e- 382 k. Jmol-1

2. 1. Screening More Energy Effect Levels As we saw with atomicforsize, the single electron in theby Caesium’s attraction its outer electron is lowered outermost energy level is much fromelectrons. the nucleus in the screening effect caused by further all its other caesium than in lithium. - Li - - -- - Cs ------- - - -

Ionisation Energy Summary Increasing Ionisation Energy Across a period from left to right ionisation energy increases This is due to the increase in atomic charge having a greater pull on the electrons and therefore more energy is required to remove electrons.
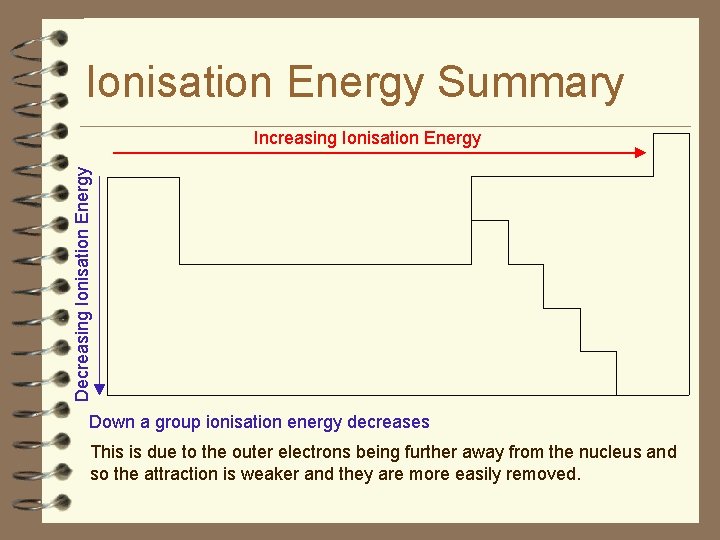
Ionisation Energy Summary Decreasing Ionisation Energy Increasing Ionisation Energy Down a group ionisation energy decreases This is due to the outer electrons being further away from the nucleus and so the attraction is weaker and they are more easily removed.
- Slides: 10