Transcatheter aortic valve implantation for failed surgical aortic










- Slides: 27

Transcatheter aortic valve implantation for failed surgical aortic bioprostheses using a self-expanding device Early results from the prospective VIVA post-market study Ran Kornowski, MD, FESC, FACC Rabin Medical Center, Petah Tikva, Israel

Disclosure Statement of Financial Interest Within the past 12 months, I have had a financial interest/arrangement or affiliation with the organization listed below. Affiliation/Financial Relationship Company • • Research grant for the conducting the VIVA trial Medtronic

Background • Surgical aortic valve replacement has been the standard of care in symptomatic patients with aortic valve disease. • However, bioprosthetic valves degenerate over time, requiring re-do surgery to replace them. • As many patients are not candidates for reoperation, a less invasive valve-in-valve (Vi. V) procedure using transcatheter aortic valve implantation (TAVI) is an emerging alternative. • The VIVA trial was designed to create a large prospective dataset among Vi. V patients treated in clinical practice.

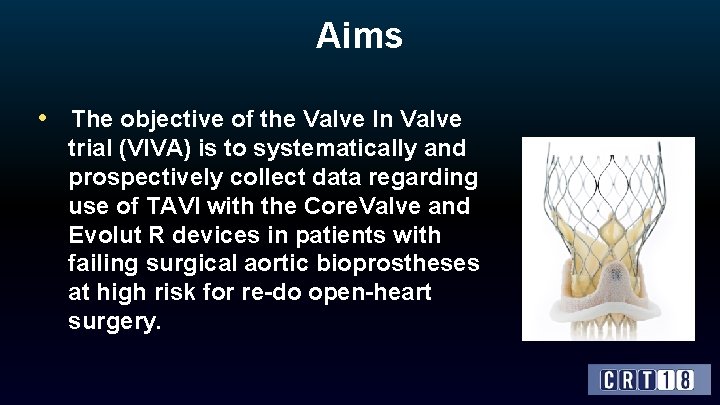
Aims • The objective of the Valve In Valve trial (VIVA) is to systematically and prospectively collect data regarding use of TAVI with the Core. Valve and Evolut R devices in patients with failing surgical aortic bioprostheses at high risk for re-do open-heart surgery.

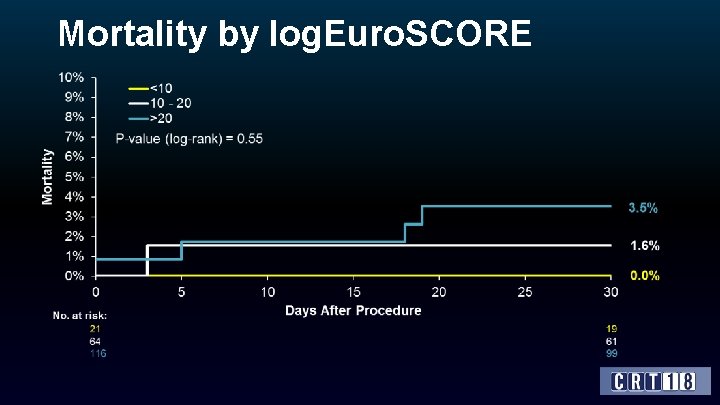
Methods • VIVA is an observational, single-arm, post-market multi-center study conducted at 23 sites in France, Germany, Israel, and Italy, which enrolled 202 patients. • Adults with symptomatic degeneration of an aortic bioprosthesis (stenosis and/or regurgitation) who were acceptable candidates for elective treatment with a selfexpanding transcatheter aortic valve were eligible for inclusion. • Patients were required to have a logistic Euro. SCORE >20% or STS score >10%, or presence of comorbidities that contraindicated redo surgery as assessed by the cardiologist and at least one cardiac surgeon OR deemed at high-risk for redo surgery by the heart team.

Trial Organization Executive Committee/PIs: Publication Committee: R. Kornowski, D. Tchétché, JP Verhoye, B. Chevalier CERC, Massy, France CRO: Sponsor: 1) CEC: P. Ménasché (Chair), G. Sardella, N. Löffelhardt 2) Echo Corelab: M. Poupineau 3) Site management Medtronic

Endpoints and Compliance Primary Safety Endpoint: Cardiovascular death at 30 days post procedure; expected to be below 10%. Primary Efficacy Endpoint: Lack of significant aortic stenosis (mean gradient > 40 mm. Hg) or insufficiency (> moderate severity) at 1 year post procedure using clinical evaluation and echocardiography Secondary Endpoints: VARC-II endpoints: Access site complications, major bleeding, stroke, AKI stage III, new pacemaker implantation, and post-implantation aortic gradient 30 -day Compliance: 98. 5% (191/194)

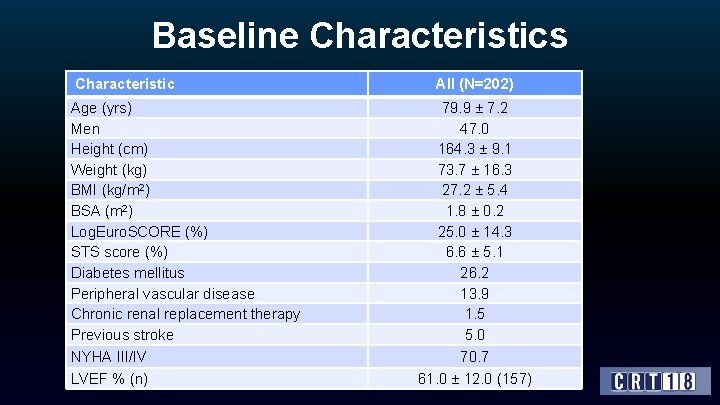
Baseline Characteristics Characteristic Age (yrs) Men Height (cm) Weight (kg) BMI (kg/m 2) BSA (m 2) Log. Euro. SCORE (%) STS score (%) Diabetes mellitus Peripheral vascular disease Chronic renal replacement therapy Previous stroke NYHA III/IV LVEF % (n) All (N=202) 79. 9 ± 7. 2 47. 0 164. 3 ± 9. 1 73. 7 ± 16. 3 27. 2 ± 5. 4 1. 8 ± 0. 2 25. 0 ± 14. 3 6. 6 ± 5. 1 26. 2 13. 9 1. 5 5. 0 70. 7 61. 0 ± 12. 0 (157)

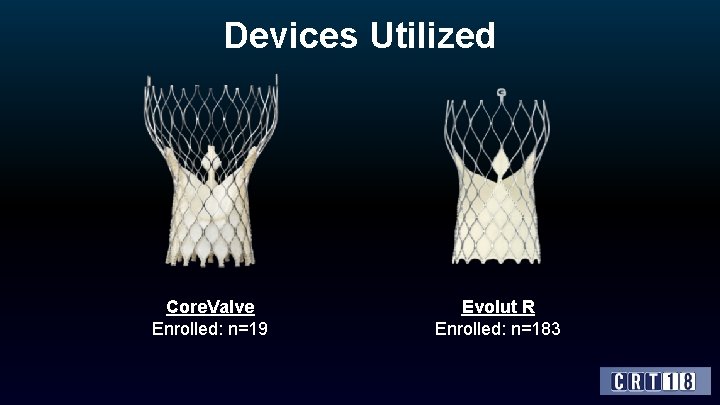
Devices Utilized Core. Valve Enrolled: n=19 Evolut R Enrolled: n=183

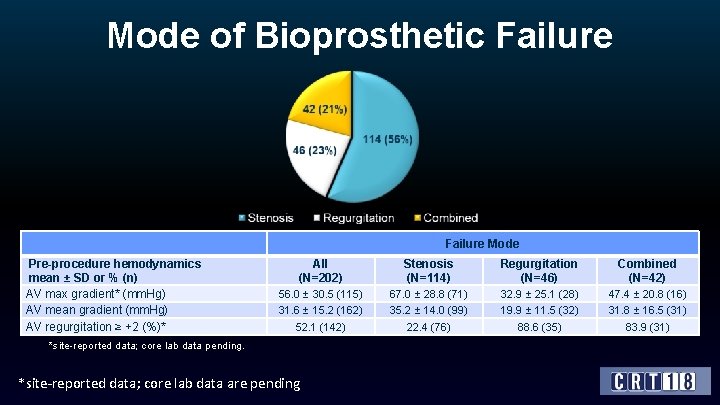
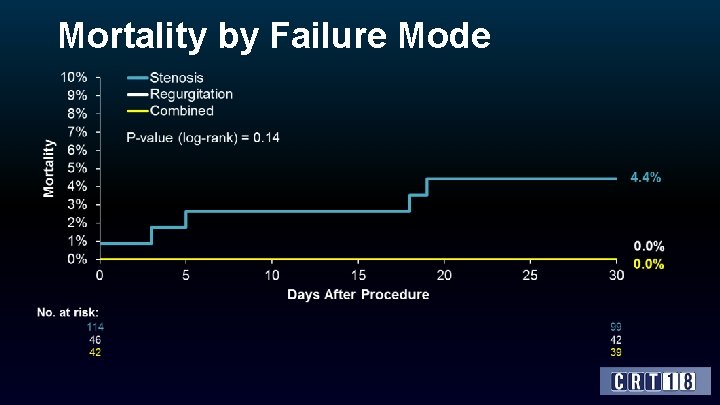
Mode of Bioprosthetic Failure Mode Pre-procedure hemodynamics mean ± SD or % (n) AV max gradient* (mm. Hg) AV mean gradient (mm. Hg) AV regurgitation ≥ +2 (%)* All (N=202) Stenosis (N=114) Regurgitation (N=46) Combined (N=42) 56. 0 ± 30. 5 (115) 67. 0 ± 28. 8 (71) 32. 9 ± 25. 1 (28) 47. 4 ± 20. 8 (16) 31. 6 ± 15. 2 (162) 35. 2 ± 14. 0 (99) 19. 9 ± 11. 5 (32) 31. 8 ± 16. 5 (31) 52. 1 (142) 22. 4 (76) 88. 6 (35) 83. 9 (31) *site-reported data; core lab data pending. *site-reported data; core lab data are pending

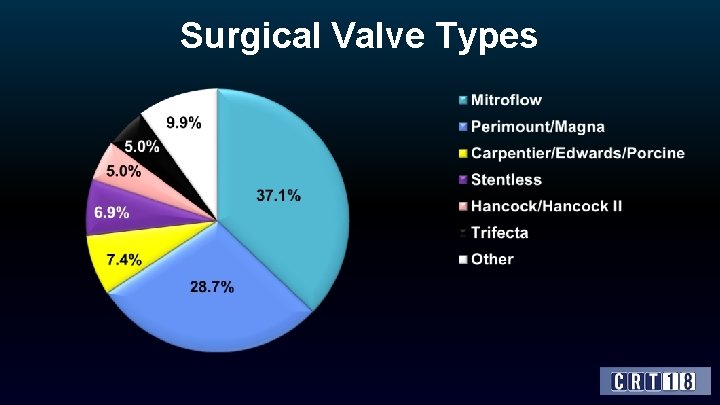
Surgical Valve Types

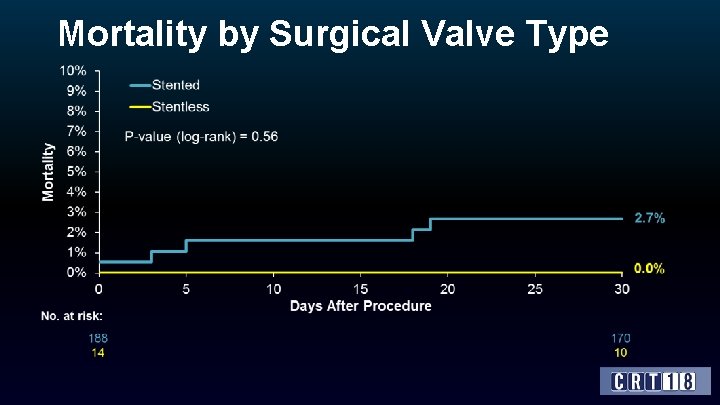
Surgical Valve Characteristics Characteristic Time since last SAVR, yrs (mean ± SD) All (N=202) 9. 3 ± 4. 4 (median) Surgical valve type (%) 8. 8 Stented 93. 1 Stentless Label size (%) 6. 9 ≤ 21 mm 41. 3 > 21 mm and < 25 mm 32. 8 ≥ 25 mm Internal diameter (%) 25. 9 < 20 mm 40. 9 ≥ 20 mm and < 23 mm 35. 1 ≥ 23 mm 24. 0

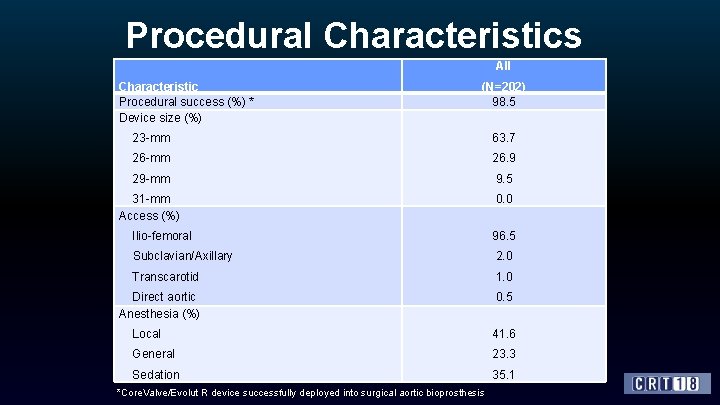
Procedural Characteristics All Characteristic Procedural success (%) * Device size (%) (N=202) 98. 5 23 -mm 63. 7 26 -mm 26. 9 29 -mm 9. 5 31 -mm Access (%) 0. 0 Ilio-femoral 96. 5 Subclavian/Axillary 2. 0 Transcarotid 1. 0 Direct aortic Anesthesia (%) 0. 5 Local 41. 6 General 23. 3 Sedation 35. 1 *Core. Valve/Evolut R device successfully deployed into surgical aortic bioprosthesis

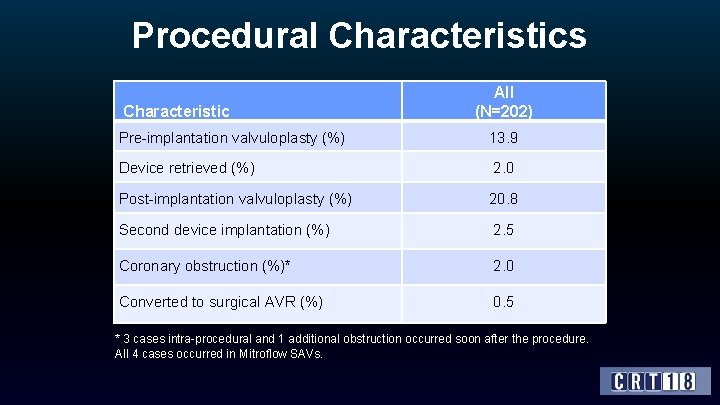
Procedural Characteristics Characteristic All (N=202) Pre-implantation valvuloplasty (%) 13. 9 Device retrieved (%) 2. 0 Post-implantation valvuloplasty (%) 20. 8 Second device implantation (%) 2. 5 Coronary obstruction (%)* 2. 0 Converted to surgical AVR (%) 0. 5 * 3 cases intra-procedural and 1 additional obstruction occurred soon after the procedure. All 4 cases occurred in Mitroflow SAVs.

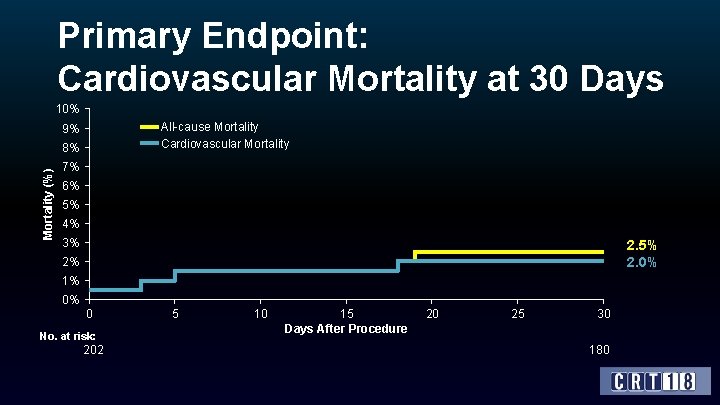
Primary Endpoint: Cardiovascular Mortality at 30 Days 10% All-cause Mortality Cardiovascular Mortality 9% Mortality (%) 8% 7% 6% 5% 4% 3% 2. 5% 2. 0% 2% 1% 0% 0 No. at risk: 202 5 10 15 Days After Procedure 20 25 30 180

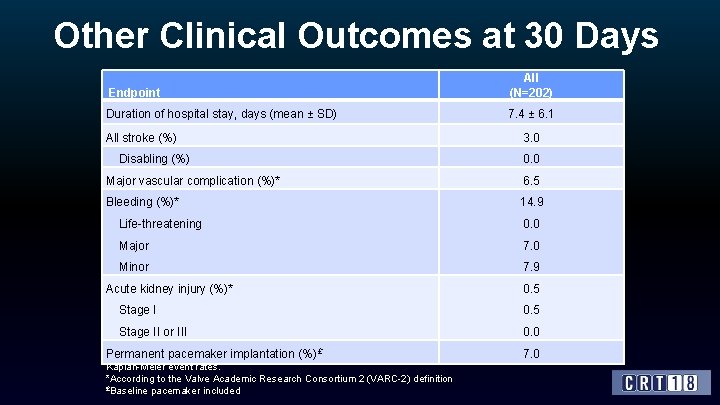
Other Clinical Outcomes at 30 Days Endpoint All (N=202) Duration of hospital stay, days (mean ± SD) 7. 4 ± 6. 1 All stroke (%) 3. 0 Disabling (%) 0. 0 Major vascular complication (%)* 6. 5 Bleeding (%)* 14. 9 Life-threatening 0. 0 Major 7. 0 Minor 7. 9 Acute kidney injury (%)* 0. 5 Stage II or III 0. 0 Permanent pacemaker implantation (%) £ 7. 0 Kaplan-Meier event rates. *According to the Valve Academic Research Consortium 2 (VARC-2) definition £Baseline pacemaker included

Mortality by log. Euro. SCORE

Mortality by Failure Mode

Mortality by Surgical Valve Type

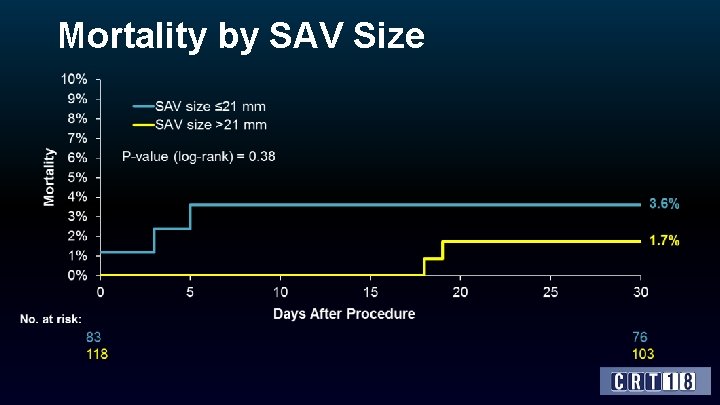
Mortality by SAV Size

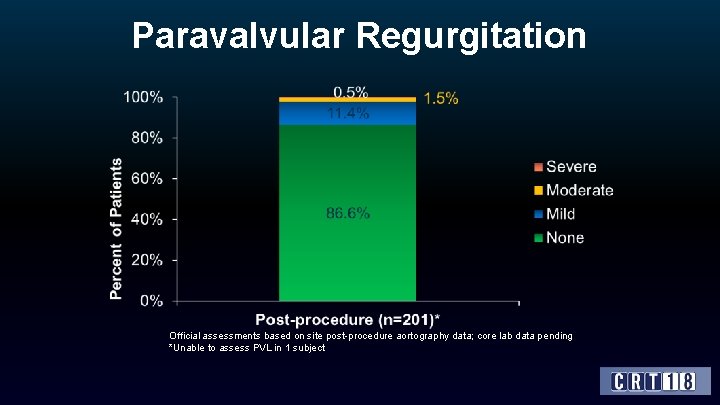
Paravalvular Regurgitation Official assessments based on site post-procedure aortography data; core lab data pending *Unable to assess PVL in 1 subject

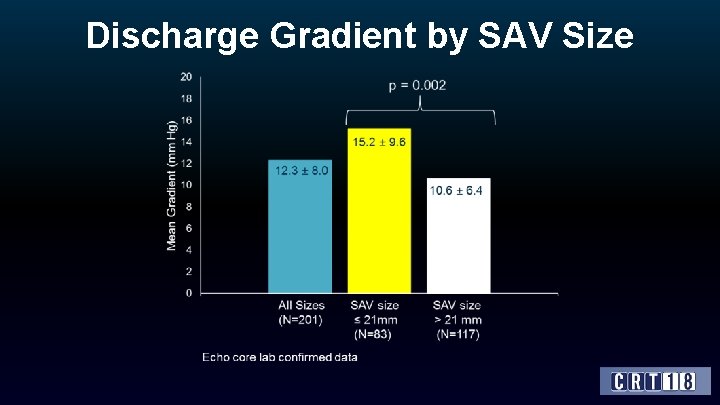
Discharge Gradient by SAV Size

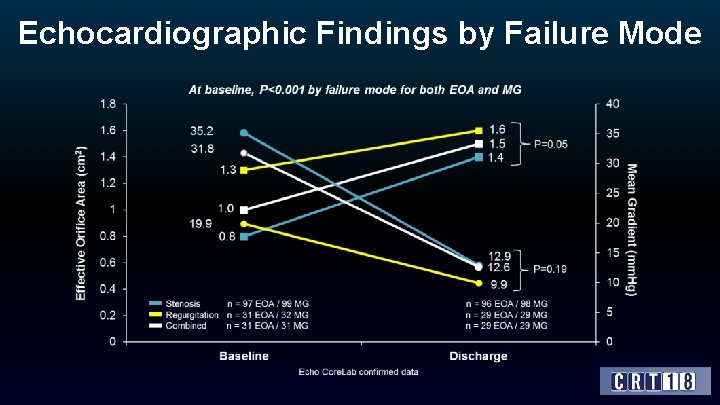
Echocardiographic Findings by Failure Mode

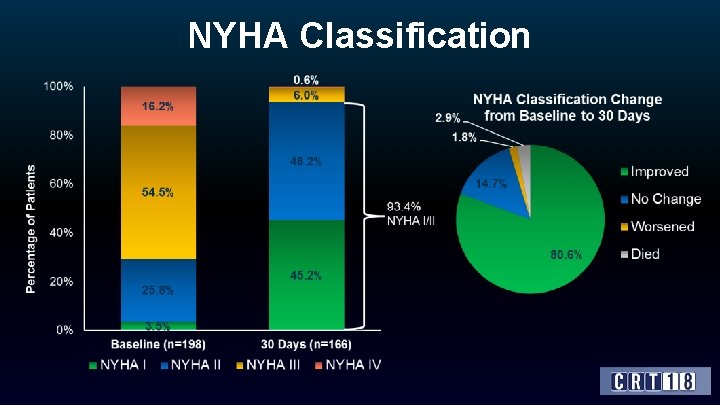
NYHA Classification

Conclusions • The VIVA trial confirmed the feasibility, safety and effectiveness of the TAVI Vi. V intervention using the Core. Valve/Evolut R devices in high-risk patients with failing surgical aortic bioprostheses. • 30 -day mortality/CV mortality was 2. 5%/2. 0% among patients who had average Log. Euro. SCORE 25% and mean STS 6. 6%. In this respect, the study met its primary safety endpoint at 30 -days (which was pre-defined as 30 -day CV mortality rate <10%). • Complications were mostly minor (i. e. not life threatening) and at a relatively low rate. • Echocardiography data at discharge and NYHA functional class after 30 -days are favorable, which indicates the short-term effectiveness of this mode of treatment. • The one-year clinical and echocardiographic efficacy data are awaited and will be reported in the near-future.

Please note

Participating Centers France Clinique Pasteur; Toulouse - Dr. Didier Tchétché Hopital Jacques Cartier; Massy – Dr. Bernard Chevalier CHU Mondor; Créteil - Prof. Emmanuel Teiger CHU de Nantes - Dr. Thibaut Manigold CHU Lille – Dr. Thomas Modine CHU Clermont; Clermont-Ferrand – Dr. Geraud Souteyrand Tonkin Clinic; Villeurbanne – Dr. Didier Champagnac CHU Rennes - Prof. Jean Philippe Verhoye CHU Bordeaux; Pessac - Dr Lionel Leroux CHU Brest - Prof. Martine Gilard CHU Rangueil; Toulouse - Dr. Bertrand Marcheix Clinique Parly 2; Le Chesnay - Dr. Gregoire Dambrin CHU La Timone; Marseille - Dr. Dominique Grisoli Israel Rabin Medical Center; Petah Tikva - Prof. Ran Kornowski Sheba Medical Center; Tel Hashomer - Prof. Victor Guetta Germany Herzzentrum Leipzig - Dr. David Holzhey Sana-Herzzentrum Cottbus - Dr. Axel Harnath UK Hamburg Eppendorf - Prof. Ulrich Schäfer Herz-und Diabeteszentrum NRW; Bad Oeynhausen - Dr. Werner Scholtz Kerckhoff Klinik; Bad Nauheim - Dr. Won-Keun Kim Italy Brescia Hospital - Dr. Federica Ettori Pisa Hospital - Prof. Anna Sonia Petronio San Donato; Milano - Prof. Francesco Bedogni