The 65 th International Symposium on Molecular Spectroscopy









- Slides: 19

The 65 th International Symposium on Molecular Spectroscopy, June 2010 Optical Stark Spectroscopy of Iridium Monofluoride, Ir. F Xiujuan Zhuang & Timothy C. Steimle Dept. Chem. & Bio. Chem. Arizona State University, Tempe, AZ, USA Colan Linton Dept. Phys. & Chem, University of New Brunswick, Fredericton, NB, Canada Funded by: Do. E-BES

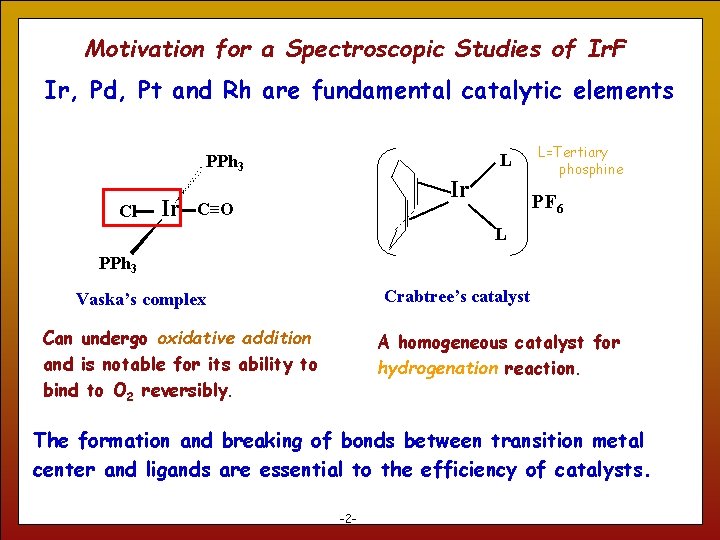
Motivation for a Spectroscopic Studies of Ir. F Ir, Pd, Pt and Rh are fundamental catalytic elements L PPh 3 Cl Ir Ir C≡O L=Tertiary phosphine PF 6 L PPh 3 Crabtree’s catalyst Vaska’s complex Can undergo oxidative addition and is notable for its ability to bind to O 2 reversibly. A homogeneous catalyst for hydrogenation reaction. The formation and breaking of bonds between transition metal center and ligands are essential to the efficiency of catalysts. 2 -2 -

Motivation for a Spectroscopic Studies of Ir. F (cont. ) Challenging Ir+ electron configurations: high density of states d 7 s 1 1, 3 D, 1, 3 P, 1, 3 D, 1, 3 F, 1, 3 G, 1, 3 H, 3, 5 P, 3, 5 F d 8 1 S, 1 D, 1 G, 3 P, 3 F Hund’s rules Low-lying states! Possible Ground & low-lying states: Ir+ (d 7 s 1) Ir+ (d 8) 3 F 5 F 5(E=0) 4(E=2260 cm 3Φ(d 8) Ir. F: 3, 5Φ, 3, 5Δ, 3, 5Π, 3, 5ΣIr. F: 3Φ, 3 Δ, 3Π, 3Σ- ) -1 3Φ i –- d 8+d 7 s 1 3Φ –- d 8+d 7 s 1 Spin-orbit 3Φ(d 7 s 1) 5Φ 5Φ(d 7 s 1) Hund’s rules ordering 3 -3 - i

Previous Related Work – • Ir. F not much UNB group: Visible LIF and DF in a free-jet expansion Fine structure parameters for numerous visible bands “red system” A 3Φ 4 -X 3Φ 4 Stark spectrum of Ir. F A 3Φ 4 -X 3Φ 4 No theoretical calculations! Today's talk • Ir-related molecules: Ir. C & Ir. N ASU group: Magnetic hyperfine interactions Permanent electric dipole J. Chem. Phys. 104, 8183, 1996 The goal: Determine μ for Ir. F, and compare with Ir. C, Ir. N and Co. F 4 Fe 26 Co 27 Ni 28 [Ar]3 d 64 s 2 [Ar]3 d 74 s 2 [Ar]3 d 84 s 2 Ru 44 Rh 45 Pd 46 [Kr]4 d 75 s 1 [Kr]4 d 85 s 1 [Kr]4 d 10 Os 76 Ir 77 Pt 78 [Xe]4 f 145 d 66 s 2 [Xe]4 f 145 d 76 s 2 [Xe]4 f 145 d 96 s 1 Extensive studies on isovalent Co. F -4 -

Experimental approach l Supersonic expansion /Ablation Source 532 nm Nd: YAG Pulse Laser Pulse Valve SF 6 (4%) + Ar Plasma Ir rod 5 -5 -

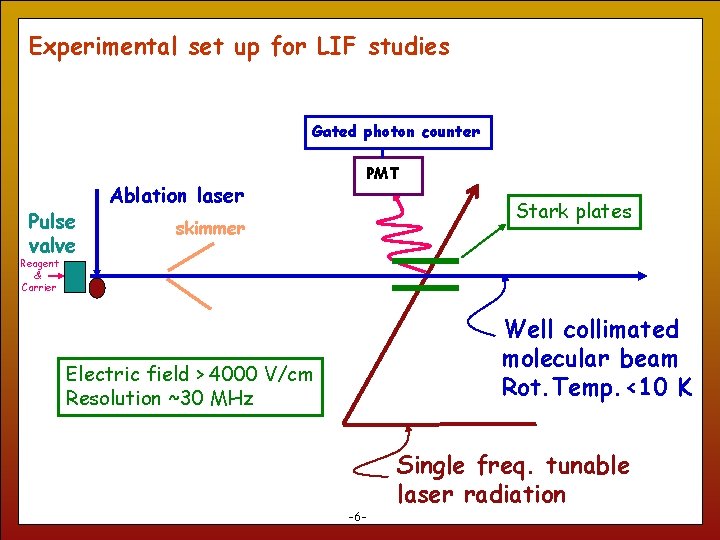
Experimental set up for LIF studies Gated photon counter Pulse valve Ablation laser PMT Stark plates skimmer Reagent & Carrier Well collimated molecular beam Rot. Temp. <10 K Electric field > 4000 V/cm Resolution ~30 MHz 6 -6 - Single freq. tunable laser radiation

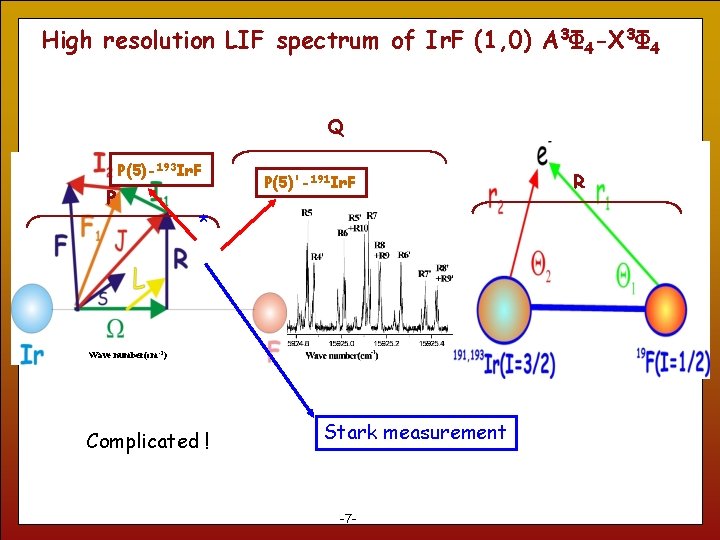
High resolution LIF spectrum of Ir. F (1, 0) A 3Φ 4 -X 3Φ 4 Q P(5)-193 Ir. F P P(5)'-191 Ir. F * Wave number(cm-1) Complicated ! 7 Stark measurement -7 - R

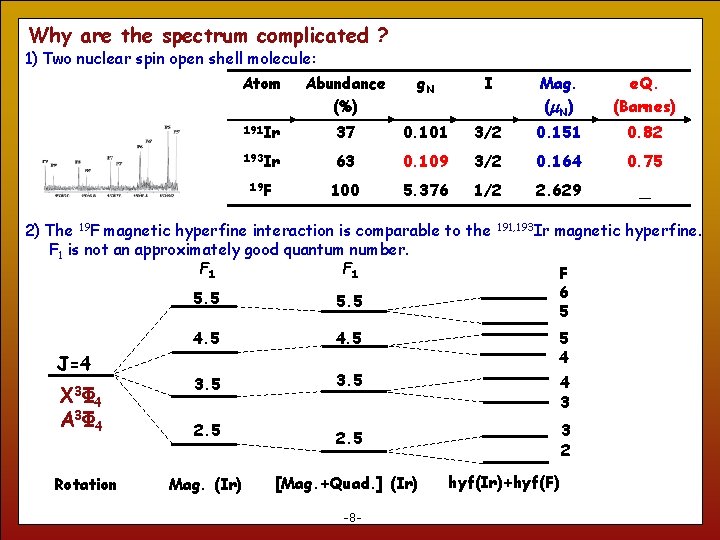
Why are the spectrum complicated ? 1) Two nuclear spin open shell molecule: Atom Abundance (%) g. N I Mag. (m. N ) e. Q. (Barnes) 191 Ir 37 0. 101 3/2 0. 151 0. 82 193 Ir 63 0. 109 3/2 0. 164 0. 75 19 F 100 5. 376 1/2 2. 629 _ 2) The 19 F magnetic hyperfine interaction is comparable to the F 1 is not an approximately good quantum number. F 1 J=4 X 3Φ 4 A 3Φ 4 8 Rotation 191, 193 Ir magnetic hyperfine. F 6 5 5. 5 4. 5 5 4 3 2. 5 3 2 2. 5 Mag. (Ir) [Mag. +Quad. ] (Ir) -8 - hyf(Ir)+hyf(F)

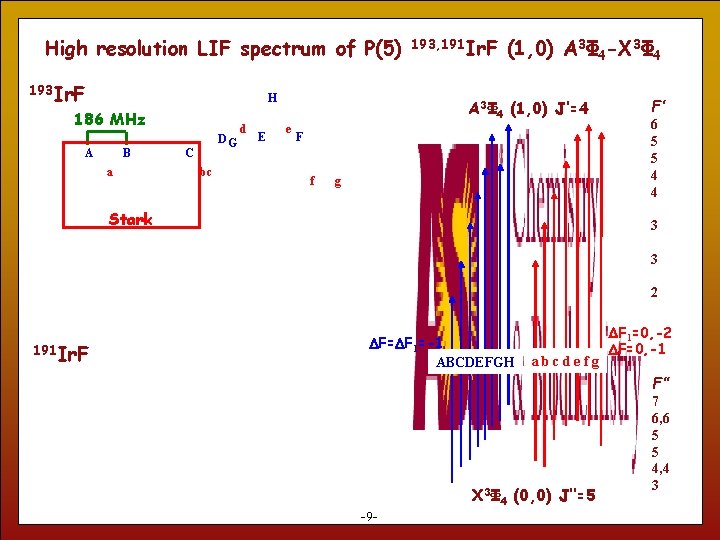
High resolution LIF spectrum of P(5) 193 Ir. F H 186 MHz A B a DG C bc d E 193, 191 Ir. F (1, 0) A 3Φ 4 -X 3Φ 4 A 3Φ 4 (1, 0) J’=4 e F f g Stark F’ 6 5 5 4 4 3 3 2 191 Ir. F DF=DF 1=-1 ABCDEFGH abcdefg DF 1=0, -2 DF=0, -1 F’’ 9 -9 - X 3Φ 4 (0, 0) J’’=5 7 6, 6 5 5 4, 4 3

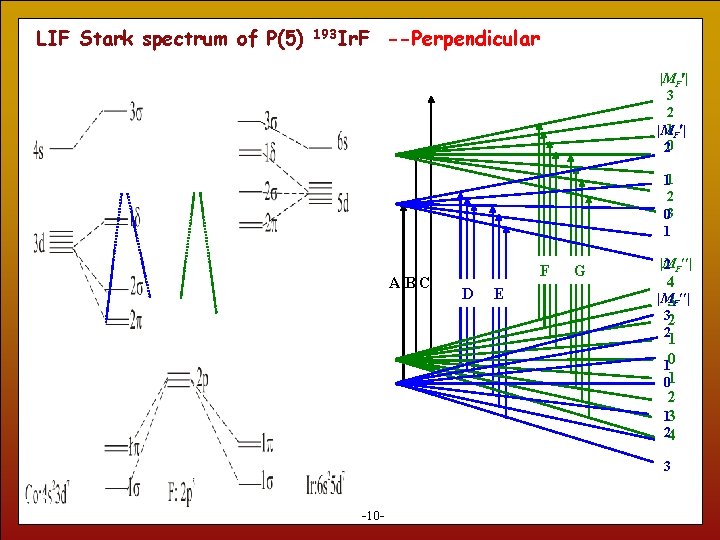
LIF Stark spectrum of P(5) 193 Ir. F --Perpendicular |MF | 3 2 |M 1 F | 20 11 2 03 1 ABC F D E G |M 2 F | 4 |M 3 F | 32 2 1 10 01 2 13 24 3 10 -10 -

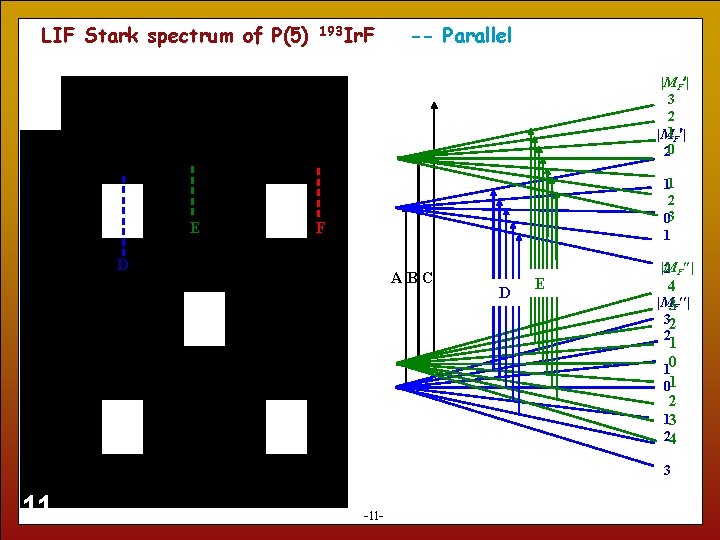
LIF Stark spectrum of P(5) 193 Ir. F -- Parallel |MF | 3 2 |M 1 F | 20 E 11 2 03 1 F D ABC D E |M 2 F | 4 |M 3 F | 32 2 1 10 01 2 13 24 3 11 -11 -

Modeling the Field Free spectrum 193 Ir. F A 3Φ 4 -X 3Φ 4 e. Qq 0 Field free Hamiltonian Heff = ALz. Sz+BR 2 + (3 I 2 – I 2) +h 4(Ir) Iz Lz +b. F(F) I S 4 I(2 I-1) z Matrix representation in a Hund’s sequentially coupled basis set: 16 16 Diagonalize Eigenvalues Matrix & (| | =4) Eigenvectors A direct least squares fit to the observed field-free wavenumbers was performed. # data points: 50 for 193 Ir. F rms of fits: 0. 0004 cm-1 12 -12 - ; 38 for 191 Ir. F

Field free parameters used to predict P(5) lines of (1, 0) A 3 4 -X 3 4 band. Parameters a X 3 4(0, 0) A 3 4(1, 0) m B(193 Ir. F) 0. 28358(1)b 0. 25494(1)b B(191 Ir. F) 0. 28388(1)b 0. 25520(1)b b. F(19 F) 0. 00860(44) 0. 01428(27) (193 Ir) 0. 00906(18) 0. 01512(18) h 4(191 Ir) 0. 00795(21) 0. 01362(21) -0. 00972(83) -0. 03859(66) -0. 01167(100) -0. 04265(76) h 4 e. Qq 0(193 Ir) e. Qq 0 (191 Ir) h 4(193 Ir) h 4(191 Ir) e. Qq 0(193 Ir) e. Qq 0(191 Ir) 1. 14 1. 11 0. 83 0. 91 0 a = 4 p 2 g. Nm. Bm. N<L|Slziri-3|L>l /L m 0 8 p b. F = 4 p 3 geg. Nm. Bm. N<L|Sszid(r)|L>s/S m 0 2 2 c = 4 p 3 geg. Nm. Bm. N<L|Sszi(3 cos qi-1) ri-3|L>s/S h 4=3 a+b. F+ 1. 09 0. 92 Optimized by least square fitting program, r. m. s. =0. 0004 cm-1 Units in cm-1 b Ref. Can. J. Phys. Vol 87, 2009 A. G. Adam et al. a 13 -13 - 2 C 3 g. N(193 Ir) g. N(191 Ir) Q(193 Ir) Q(191 Ir) h 4 ~ g. N

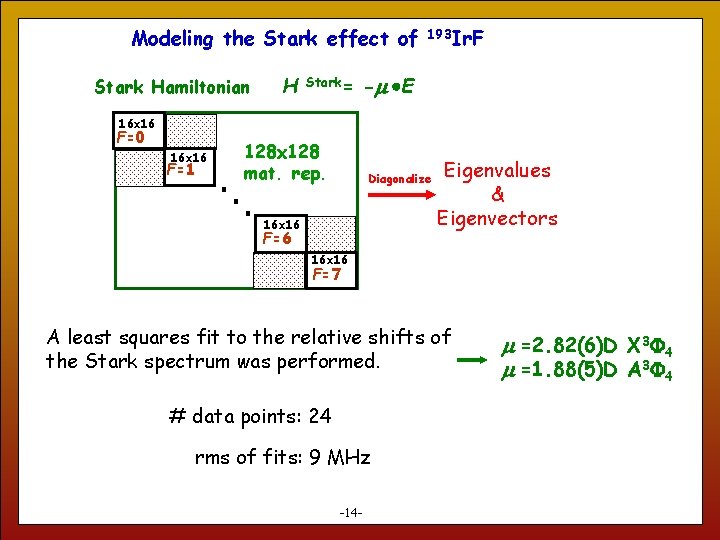
Modeling the Stark effect of Stark Hamiltonian H Stark= 193 Ir. F -m E 16 x 16 F=0 16 x 16 F=1 128 x 128 mat. rep. Diagonalize 16 x 16 F=6 Eigenvalues & Eigenvectors 16 x 16 F=7 A least squares fit to the relative shifts of the Stark spectrum was performed. # data points: 24 rms of fits: 9 MHz 14 -14 - m =2. 82(6)D X 3 4 m =1. 88(5)D A 3 4

Comparison of ground state dipole moments a b Molecules Ir. C Ir. N Ir. F Co. F m (D) 1. 60(7) 1. 66(1) 2. 82(6) 4. 51(5) state X 2 D 5/2 X 1 S + X 3 4 Re(Å) 1. 6086 1. 6094 1. 8507 b 1. 7358 m/Re 0. 99 1. 03 1. 52 2. 60 All the m and Re come from experimental measurements of Prof. Tim Steimle’s group Ref. Can. J. Phys. Vol 87, 2009 A. G. Adam et al. Electronegativity: (in Pauling scale) Ir F Ir N 2. 20 3. 98 2. 20 3. 04 Ir C 2. 20 2. 55 Co 1. 88 15 a F 3. 98 Consistent with electronegativity ! -15 -

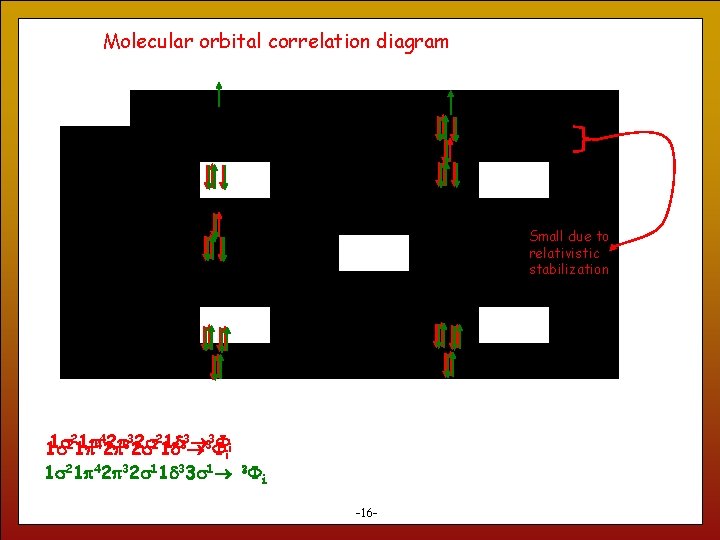
Molecular orbital correlation diagram Small due to relativistic stabilization 1 s 221 p 1 p 442 p 2 p 332 s 2 s 221 d 1 d 33 i 1 s 21 p 42 p 32 s 11 d 33 s 1 3 i 16 -16 -

Comparison of 3 s and 2 s contribution to m 1 s 21 p 42 p 32 s 21 d 3 3 i 1 s 21 p 42 p 32 s 11 d 33 s 1 3 i 2 s 3 s e- Ir/Co m ind mbond m ind 17 Ir/Co F F mbond mtot Small m Large m -17 -

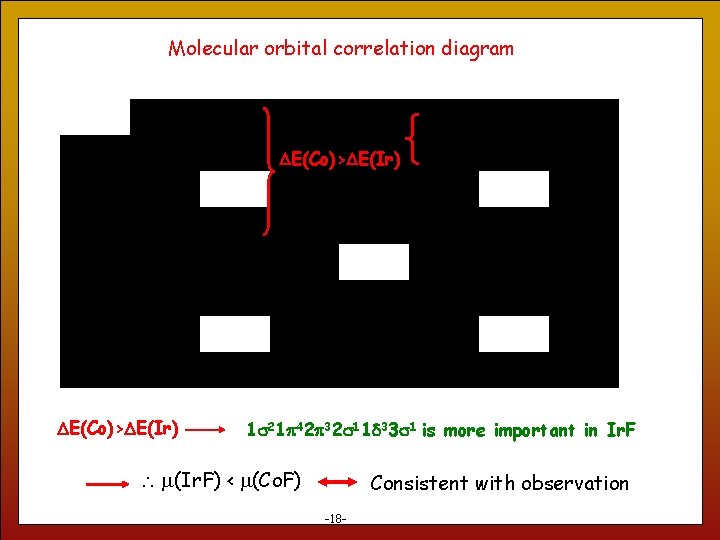
Molecular orbital correlation diagram DE(Co)>DE(Ir) 18 1 s 21 p 42 p 32 s 11 d 33 s 1 is more important in Ir. F m(Ir. F) < m(Co. F) Consistent with observation -18 -

Conclusion a) The complicated hyperfine structure of low-J lines has been analyzed. b) The permanent electric dipole moment were obtained form optical Stark spectrum. Still to do: a) Interpret the hyperfine parameters. b) Theoretical predictions (by others). Thank you 19 -19 -