64 th OSU International Symposium on Molecular Spectroscopy




- Slides: 16

64 th OSU International Symposium on Molecular Spectroscopy Assignment of mm- and smm-wave spectra of rare isotopologues of cyanamide and the rm(1) geometry of NH 2 CN Adam Krasnicki, Zbigniew Kisiel Institute of Physics, Polish Academy of Sciences Brenda P. Winnewisser, Manfred Winnewisser Department of Physics, The Ohio State University

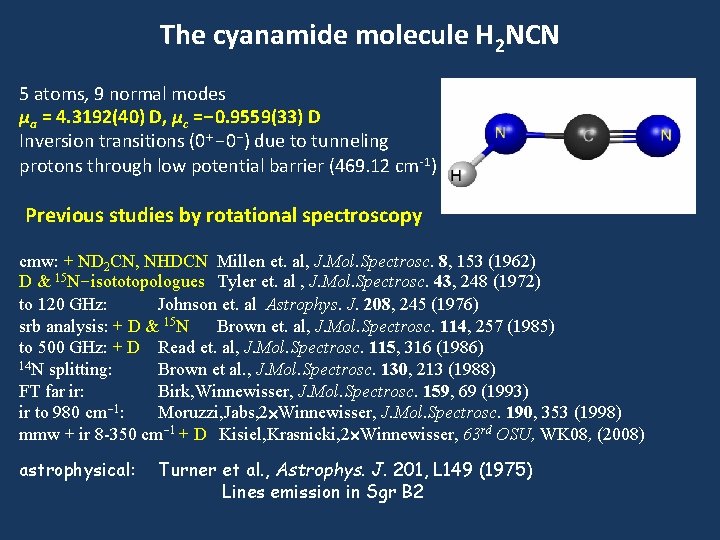
The cyanamide molecule H 2 NCN 5 atoms, 9 normal modes μa = 4. 3192(40) D, μc =-0. 9559(33) D Inversion transitions (0+-0 -) due to tunneling protons through low potential barrier (469. 12 cm-1) Previous studies by rotational spectroscopy cmw: + ND 2 CN, NHDCN Millen et. al, J. Mol. Spectrosc. 8, 153 (1962) D & 15 N-isototopologues Tyler et. al , J. Mol. Spectrosc. 43, 248 (1972) to 120 GHz: Johnson et. al Astrophys. J. 208, 245 (1976) srb analysis: + D & 15 N Brown et. al, J. Mol. Spectrosc. 114, 257 (1985) to 500 GHz: + D Read et. al, J. Mol. Spectrosc. 115, 316 (1986) 14 N splitting: Brown et al. , J. Mol. Spectrosc. 130, 213 (1988) FT far ir: Birk, Winnewisser, J. Mol. Spectrosc. 159, 69 (1993) ir to 980 cm-1: Moruzzi, Jabs, 2 Winnewisser, J. Mol. Spectrosc. 190, 353 (1998) mmw + ir 8 -350 cm-1 + D Kisiel, Krasnicki, 2 Winnewisser, 63 rd OSU, WK 08, (2008) astrophysical: Turner et al. , Astrophys. J. 201, L 149 (1975) Lines emission in Sgr B 2

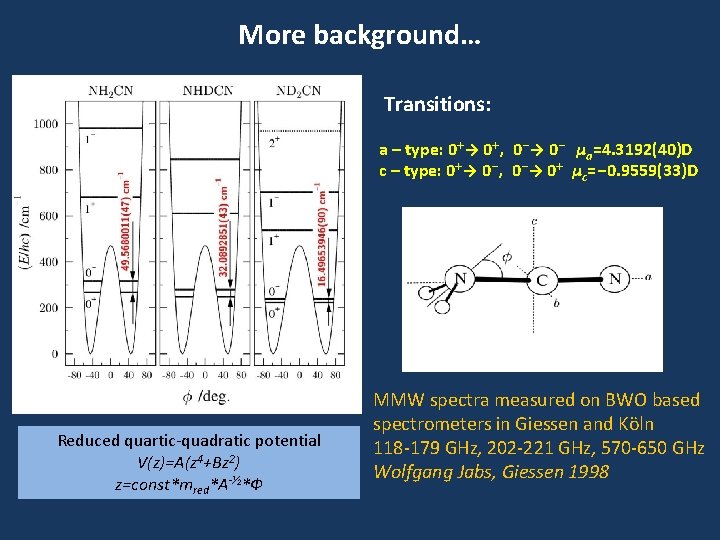
More background… Transitions: a – type: 0+→ 0+, 0 -→ 0 - μa=4. 3192(40)D c – type: 0+→ 0 -, 0 -→ 0+ μc=-0. 9559(33)D Reduced quartic-quadratic potential V(z)=A(z 4+Bz 2) z=const*mred*A-⅟ 2*Ф MMW spectra measured on BWO based spectrometers in Giessen and Köln 118 -179 GHz, 202 -221 GHz, 570 -650 GHz Wolfgang Jabs, Giessen 1998

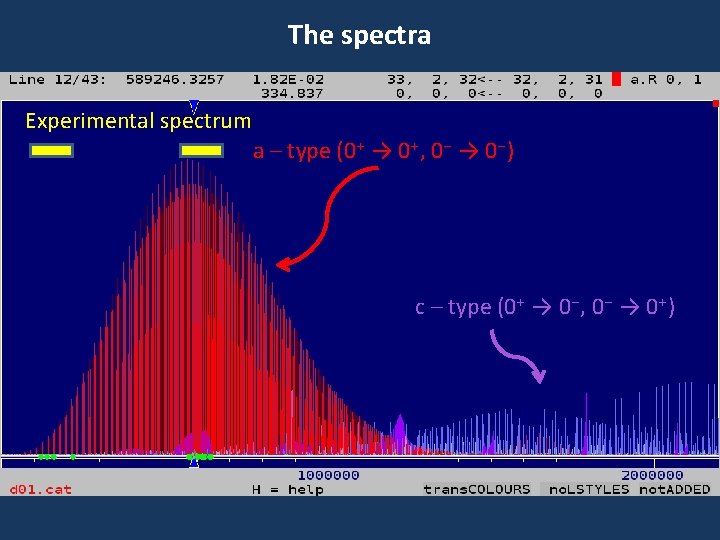
The spectra Experimental spectrum a – type (0+ → 0+, 0 - → 0 -) c – type (0+ → 0 -, 0 - → 0+)

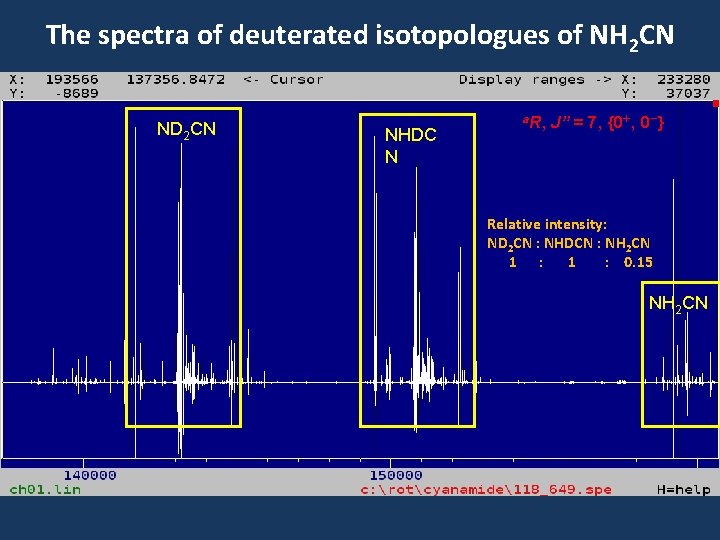
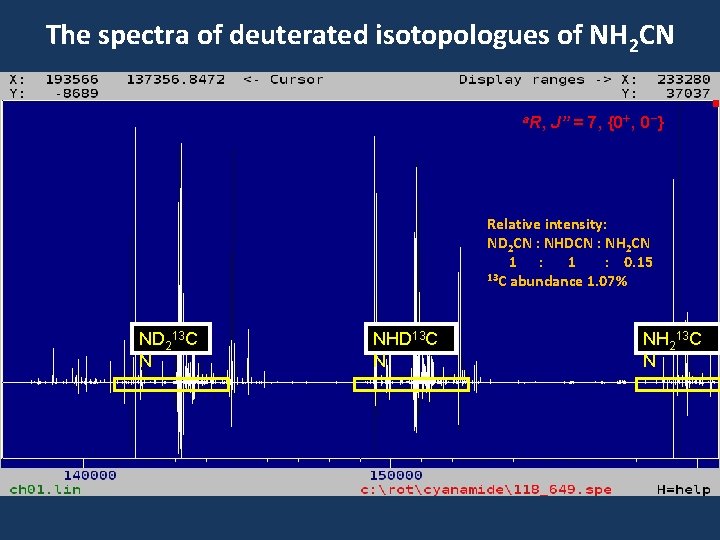
The spectra of deuterated isotopologues of NH 2 CN ND 2 CN NHDC N a. R, J” = 7, {0+, 0 -} Relative intensity: ND 2 CN : NHDCN : NH 2 CN 1 : 0. 15 NH 2 CN

The spectra of deuterated isotopologues of NH 2 CN a. R, J” = 7, {0+, 0 -} Relative intensity: ND 2 CN : NHDCN : NH 2 CN 1 : 0. 15 13 C abundance 1. 07% ND 213 C N NHD 13 C N NH 213 C N

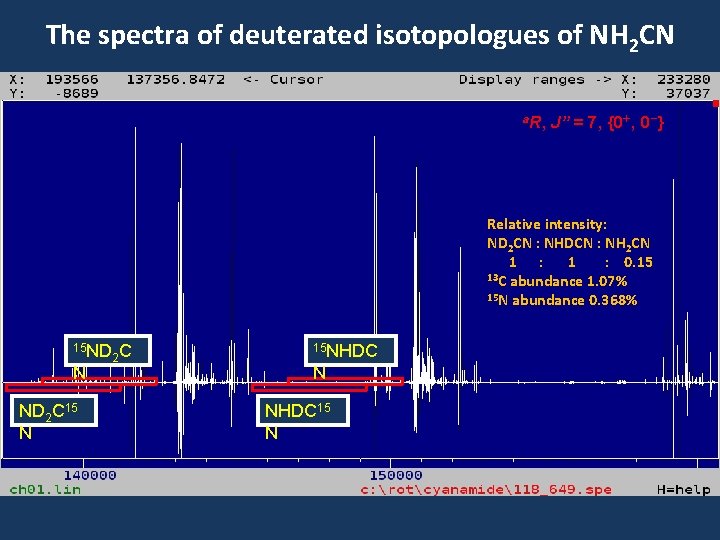
The spectra of deuterated isotopologues of NH 2 CN a. R, J” = 7, {0+, 0 -} Relative intensity: ND 2 CN : NHDCN : NH 2 CN 1 : 0. 15 13 C abundance 1. 07% 15 N abundance 0. 368% 15 ND N ND 2 C 15 N 2 C 15 NHDC N NHDC 15 N

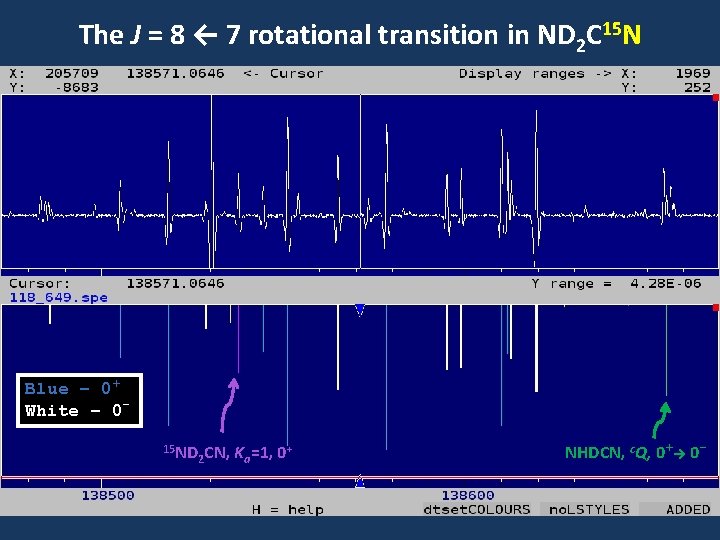
The J = 8 ← 7 rotational transition in ND 2 C 15 N Blue – 0+ White – 015 ND 2 CN, Ka=1, 0+ NHDCN, c. Q, 0+→ 0 -

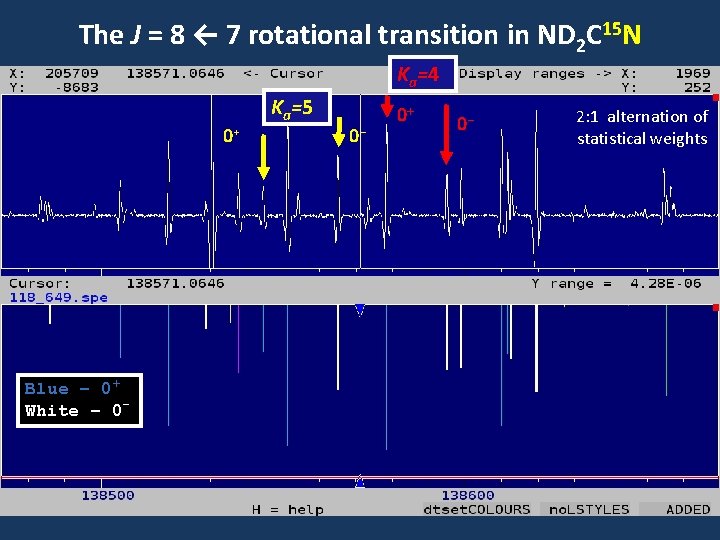
The J = 8 ← 7 rotational transition in ND 2 C 15 N Ka=4 0+ Blue – 0+ White – 0 - Ka=5 0 - 0+ 0 - 2: 1 alternation of statistical weights

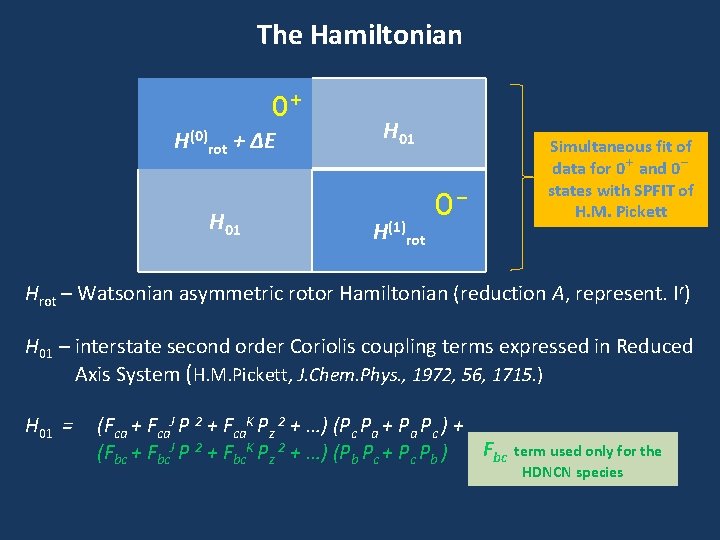
The Hamiltonian 0+ H(0)rot + ΔE H 01 H(1)rot 0 - Simultaneous fit of data for 0+ and 0 states with SPFIT of H. M. Pickett Hrot – Watsonian asymmetric rotor Hamiltonian (reduction A, represent. Ir) H 01 – interstate second order Coriolis coupling terms expressed in Reduced Axis System (H. M. Pickett, J. Chem. Phys. , 1972, 56, 1715. ) H 01 = (Fca + Fca. J P 2 + Fca. K Pz 2 + …) (Pc Pa + Pa Pc ) + Fbc (Fbc + Fbc. J P 2 + Fbc. K Pz 2 + …) (Pb Pc + Pc Pb ) term used only for the HDNCN species

The fitted constants for ND 2 C 15 N + similar results for NH 213 CN, NHD 13 CN, ND 213 CN, NHDC 15 N, 15 ND CN, 15 NHDCN 2 For ND 2 CN E = 494551. 901(27) MHz Fca = 267. 6102(19) MHz

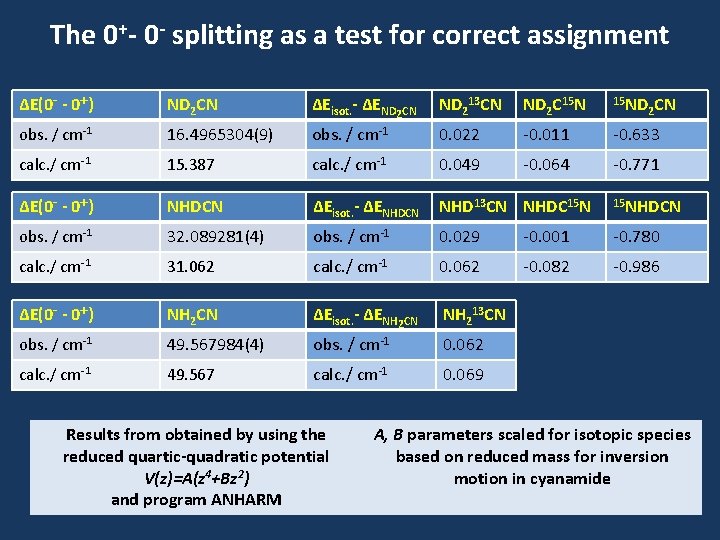
The 0+- 0 - splitting as a test for correct assignment ΔE(0 - - 0+) ND 2 CN ΔEisot. - ΔEND 2 CN ND 213 CN ND 2 C 15 ND obs. / cm-1 16. 4965304(9) obs. / cm-1 0. 022 -0. 011 -0. 633 calc. / cm-1 15. 387 calc. / cm-1 0. 049 -0. 064 -0. 771 ΔE(0 - - 0+) NHDCN ΔEisot. - ΔENHDCN NHD 13 CN NHDC 15 NHDCN obs. / cm-1 32. 089281(4) obs. / cm-1 0. 029 -0. 001 -0. 780 calc. / cm-1 31. 062 calc. / cm-1 0. 062 -0. 082 -0. 986 ΔE(0 - - 0+) NH 2 CN ΔEisot. - ΔENH 2 CN NH 213 CN obs. / cm-1 49. 567984(4) obs. / cm-1 0. 062 calc. / cm-1 49. 567 calc. / cm-1 0. 069 Results from obtained by using the reduced quartic-quadratic potential V(z)=A(z 4+Bz 2) and program ANHARM 2 CN A, B parameters scaled for isotopic species based on reduced mass for inversion motion in cyanamide

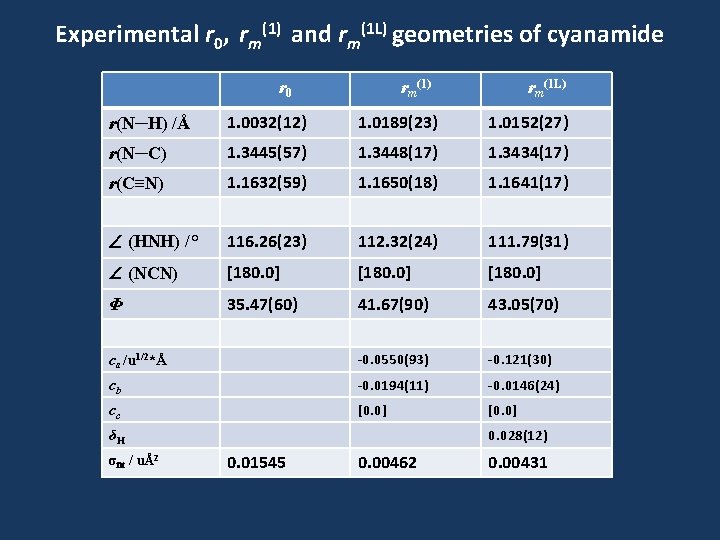
Experimental r 0, rm(1) and rm(1 L) geometries of cyanamide r 0 rm(1) rm(1 L) r(N─H) /Å 1. 0032(12) 1. 0189(23) 1. 0152(27) r(N─C) 1. 3445(57) 1. 3448(17) 1. 3434(17) r(C≡N) 1. 1632(59) 1. 1650(18) 1. 1641(17) (HNH) /° 116. 26(23) 112. 32(24) 111. 79(31) (NCN) [180. 0] Φ 35. 47(60) 41. 67(90) 43. 05(70) ca /u 1/2*Å -0. 0550(93) -0. 121(30) cb -0. 0194(11) -0. 0146(24) cc [0. 0] 0. 028(12) δH σfit / uÅ2 0. 01545 0. 00462 0. 00431

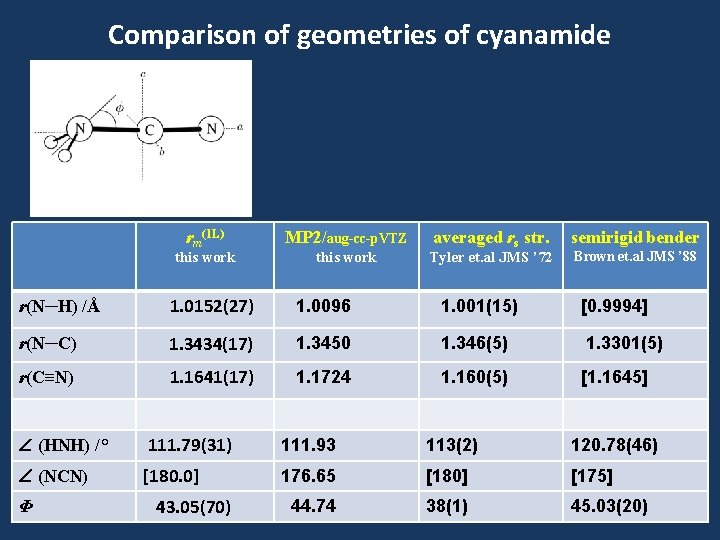
Comparison of geometries of cyanamide rm(1 L) this work MP 2/aug-cc-p. VTZ this work averaged rs str. Tyler et. al JMS ’ 72 semirigid bender Brown et. al JMS ’ 88 r(N─H) /Å 1. 0152(27) 1. 0096 1. 001(15) [0. 9994] r(N─C) 1. 3434(17) 1. 3450 1. 346(5) 1. 3301(5) r(C≡N) 1. 1641(17) 1. 1724 1. 160(5) [1. 1645] (HNH) /° 111. 79(31) 111. 93 113(2) 120. 78(46) (NCN) [180. 0] 176. 65 [180] [175] 44. 74 38(1) 45. 03(20) Φ 43. 05(70)

Conclusions • The mmw and smm rotational spectra of 7 rare isotopic species of cyanamide have been assigned , up to Ka=7 and J”=34 (ca 200 lines for each species). • Spectroscopic information on 15 ND 2 CN, ND 2 C 15 N has been considerably improved. • The spectra of NH 213 CN, NHD 13 CN, ND 213 CN, 15 NHDCN, NHDC 15 N have been assigned for the first time. • The structure of cyanamide has been derived. • We hope that further progress in understanding of the cyanamide geometry will come from semi-experimental equilibrium structure.

Acknowledgments We are indebted to Wolfgang Jabs (Giessen) who recorded all of the spectra used in this work. We are grateful to Ewa Bialkowska-Jaworska (Warszawa) for help with ab initio calculations.