Physical and Chemical Changes and Exothermic and Endothermic


















- Slides: 25

Physical and Chemical Changes and Exothermic and Endothermic Reactions

Physical Change ● Changes in a substance that do not alter the chemical properties of the substance ● Things about the substance change, but the substance remains the same ● For instance, an ice cube melts into water and you can boil that into a gas, but it remains H 2 O the whole time ● One or more of its physical properties may be changed ● Ex: change in size, shape, dissolving. possibly color change (as long as a new substance is not created)

Chemical Change ● Changes that alter the chemical properties of a substance ● A change that creates a different substance ● Clues: bubbles (indicates the production of a gas), getting hot or cold, color change ● Ex: iron rusting, souring of milk, burning paper

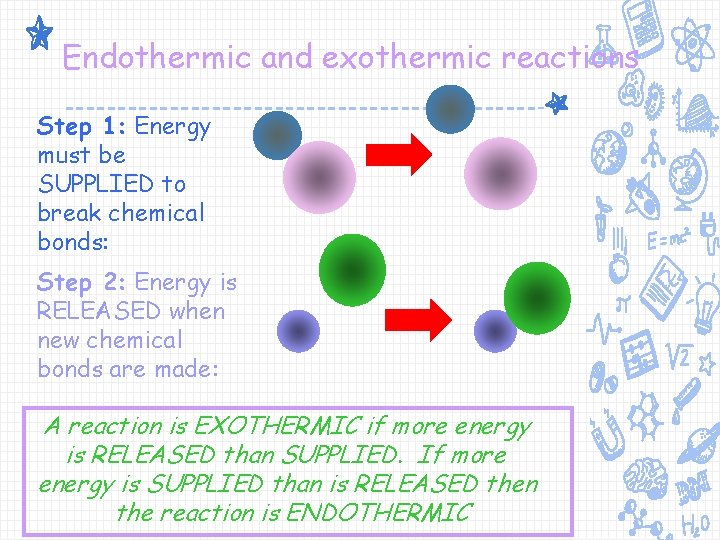
Endothermic and exothermic reactions Step 1: Energy must be SUPPLIED to break chemical bonds: Step 2: Energy is RELEASED when new chemical bonds are made: A reaction is EXOTHERMIC if more energy is RELEASED than SUPPLIED. If more energy is SUPPLIED than is RELEASED then the reaction is ENDOTHERMIC


Endothermic Reactions: a change (e. g. a chemical reaction) that requires (or absorbs) heat. Photosynthesis is an endothermic reaction (requires energy input from sun) Forming Na + and Cl - ions from Na. Cl is an endothermic process

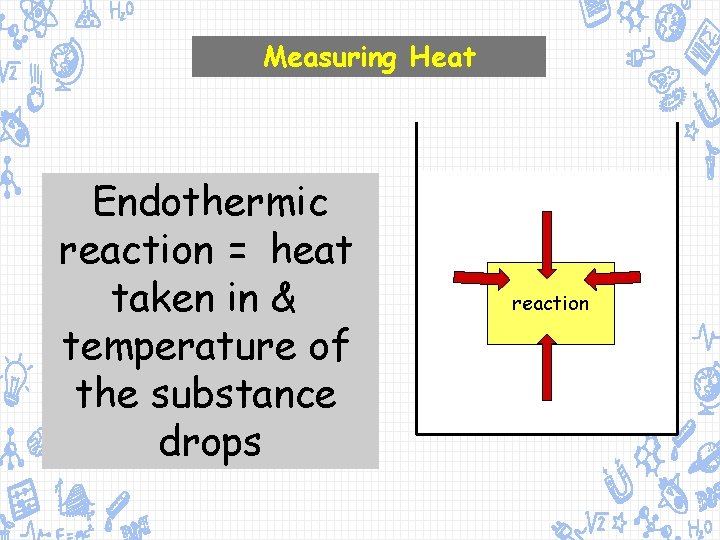
Measuring Heat Endothermic reaction = heat taken in & temperature of the substance drops reaction

Ammonium nitrate + water

Starting temperature 20⁰ C

Add ammonium nitrate to water

ending temperature of 12⁰ C a temperature decrease of 8⁰ C


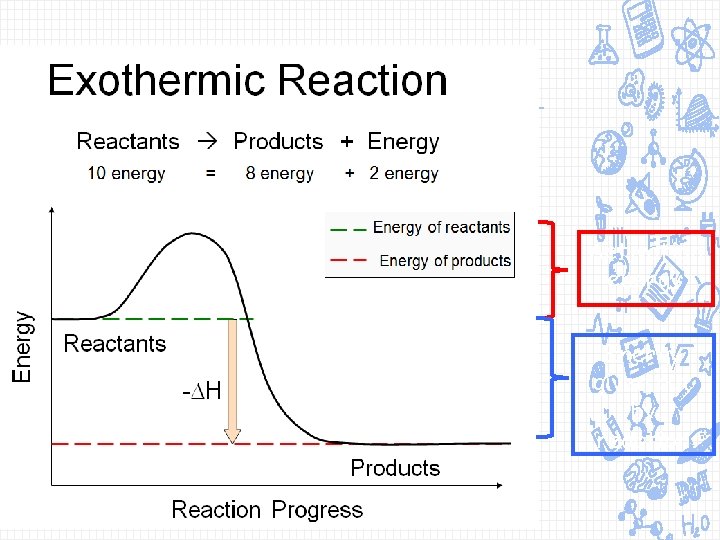
Activation energy Energy given out by reaction

Exothermic Reaction: a change (e. g. a chemical reaction) that releases heat. Burning fossil fuels is an exothermic reaction

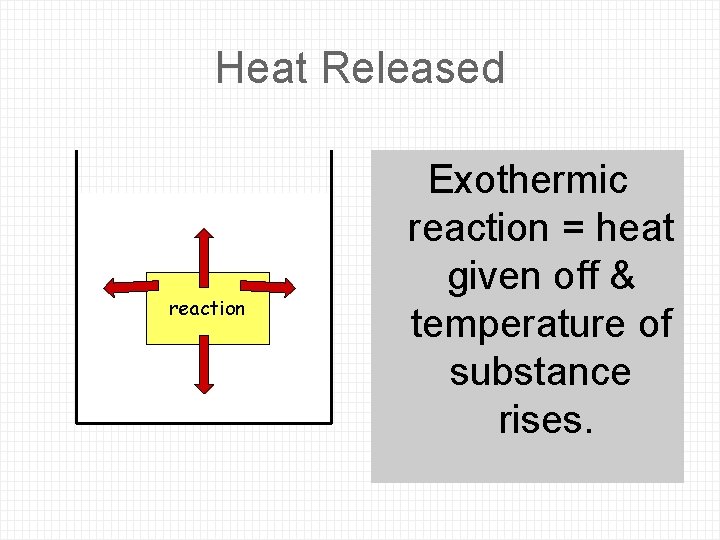
Heat Released reaction Exothermic reaction = heat given off & temperature of substance rises.

Exothermic vs endothermic: EXOTHERMIC – more energy is given out than is taken in (e. g. burning, respiration) © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910 ENDOTHERMIC – energy is taken in but not necessarily given out (e. g. photosynthesis)


Examples Exothermic • Combustion of fuels • Evaporation of Water • Yeast & Hydrogen Peroxide • Epsom salts & water © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910 Endothermic • • Photosynthesis Acetic Acid & Sodium Bicarbonate Cold Packs Melting Ice

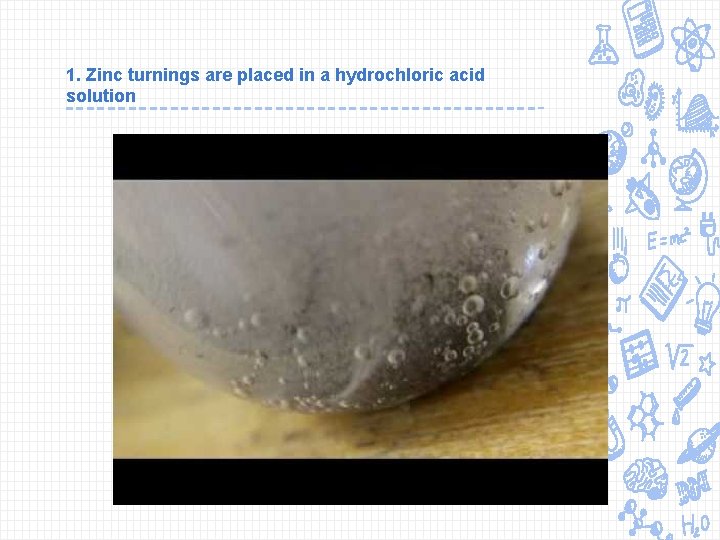
1. Zinc turnings are placed in a hydrochloric acid solution

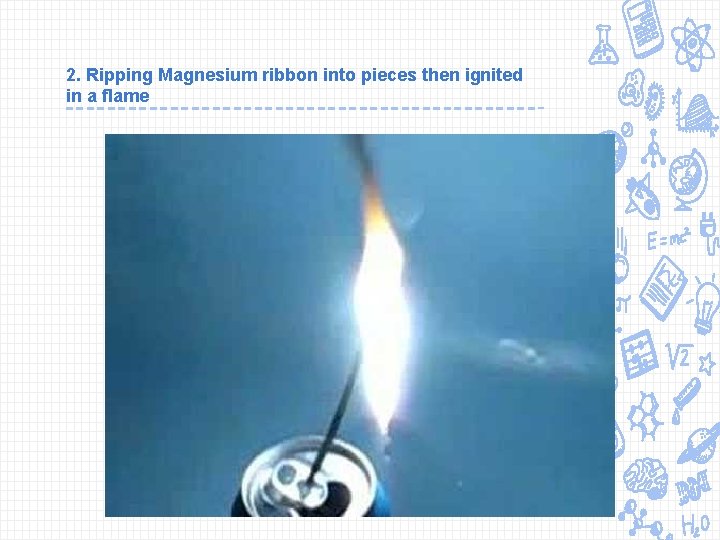
2. Ripping Magnesium ribbon into pieces then ignited in a flame

3. A penny is coated with zinc then heated Normal Penny Coated with Zinc After being heated

4. Polymers A and B are mixed

5. Hexane is poured down ramp and ignited

6. A styrofoam cup in acetone

7. Gas bubbles are made, then ignited