Exothermic Endothermic Labs Makeup Exothermic Lab Exothermic means

Exothermic. Endothermic Labs Make-up

Exothermic Lab • Exothermic means energy is being released from the reaction into the surrounding air • Heat turns from chemical energy into heat energy. • Because there is now more heat than you started with, the temperature goes up! • A temperature change can be an indicator of a chemical reaction! • Take temperature readings every minute until the reaction is done.

Now watch the temperature as we allow the Steelwe wool react withthe thecontrol oxygentest, in thewe air. While aretowatching It may like much happening but it will soaknot thelook steel wool in is vinegar to remove Keep a close eye on as This well as the. Is!protective coating onthe thetemp metal. The steelit wool. Youwith will the see oxygen changes!in the air. will allow to react Keep taking temperature readings every min. Steel Wool Bundle 250 200 150 100 50 50 On thermometer, there Are three types of lines: 1) Short black: 2. 5 degrees 2) Long black: 5 degrees 3) Long blue: lower one is 0 degrees, middle one is 50 degrees, upper one is 100 degrees To start out, we will do a “control temperature test” to determine that the temp in the beaker is not already changing on it’s own. We do this by putting thermometer in the beaker by itself for 1 minute. We watch this now: Vinegar Success! You that the hasalready risen aschanging a result of Now that wecan cansee confirm thattemperature the temp is not onthe its Reaction! This is an Exothermic Reaction!!! Also, you should “rust” own in the beaker we know that any change in temp will besee because Has on the steel. Place wool itself! Rustwool is an of the reaction. of formed the reaction alone. the steel onindicator thermometer.

Endothermic Lab • Exothermic means energy is being released from the reaction into the surrounding air • Heat turns from chemical energy into heat energy. • Because there is now more heat than you started with, the temperature goes up! • A temperature change can be an indicator of a chemical reaction! • Take temperature readings every minute until the reaction is done.
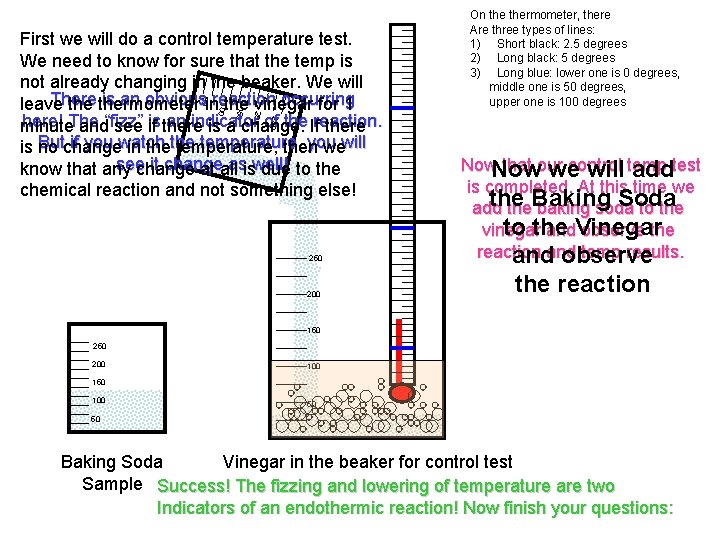
250 200 150 100 50 First we will do a control temperature test. We need to know for sure that the temp is not already changing in the beaker. We will There is an obviousinreaction occurring leave thermometer the vinegar for 1 here! The an indicator of the Ifreaction. minute and“fizz” see is if there is a change. there Butchange if you watch temperature, you is no in thethe temperature, then wewill see it change asiswell! know that any change at all due to the chemical reaction and not something else! On thermometer, there Are three types of lines: 1) Short black: 2. 5 degrees 2) Long black: 5 degrees 3) Long blue: lower one is 0 degrees, middle one is 50 degrees, upper one is 100 degrees 250 200 Now that our control we will temp addtest is completed. At this time we the Soda add the. Baking baking soda to the vinegar and. Vinegar observe the reaction temp results. andand observe the reaction 150 200 150 100 50 50 Baking Soda Vinegar in the beaker for control test Sample Success! The fizzing and lowering of temperature are two Indicators of an endothermic reaction! Now finish your questions:
- Slides: 5