O18 Evaluation of the Prognostic and Predictive Significance









- Slides: 21

O-18 Evaluation of the Prognostic and Predictive Significance of Hepatocellular Carcinoma Circulating Tumor Cells Expressing Programmed Death-Ligand 1 (PD-L 1) Pin Jun Chen, Paul Winograd, Shuang Hou, Colin Court, Saeed Sadeghi, Richard Finn, Yazhen Zhu, Fady Kaldas, Ronald Busuttil, James Tomlinson, Hsian-Rong Tseng, Vatche G. Agopian 13 th Annual Conference 13 th 20 ► 22 September 2019 Chicago, USA ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Nothing to Disclose 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Hepatocellular Carcinoma: Epidemiology • HCC is the 6 th most common cancer and 3 rd leading cause of cancer death worldwide • HCC incidence and mortality continues to rise in the United States • Majority of patients present with surgically unresectable, incurable disease. Forner, A. , M. Reig, and J. Bruix, Lancet, 2018. Petrick JL, et al. , J Clin Oncol. 2016 May 20. 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA 3

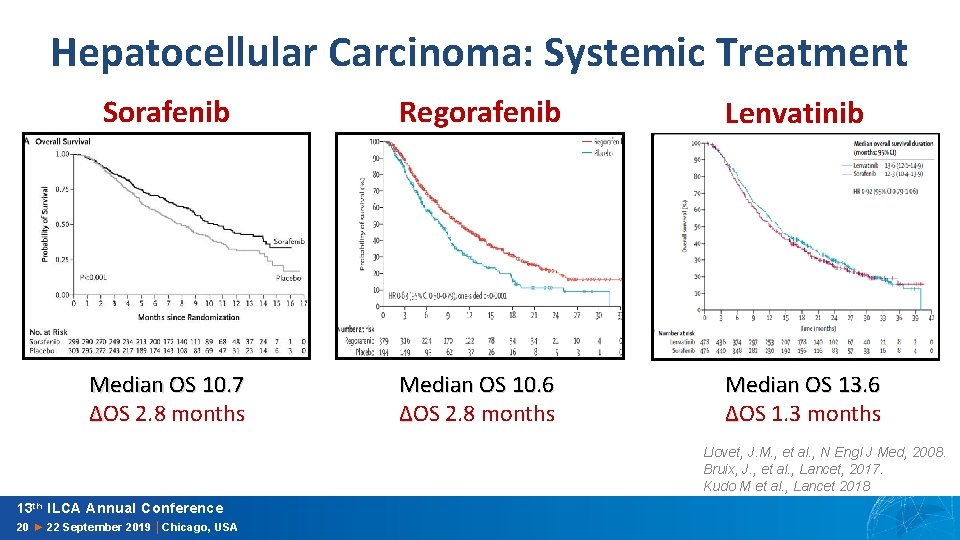
Hepatocellular Carcinoma: Systemic Treatment Sorafenib Regorafenib Lenvatinib Median OS 10. 7 ∆OS 2. 8 months Median OS 10. 6 ∆OS 2. 8 months Median OS 13. 6 ∆OS 1. 3 months Llovet, J. M. , et al. , N Engl J Med, 2008. Bruix, J. , et al. , Lancet, 2017. Kudo M et al. , Lancet 2018 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Immunotherapy: PD-1/PD-L 1 Inhibitors Retrieved from: www. cancer. gov 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

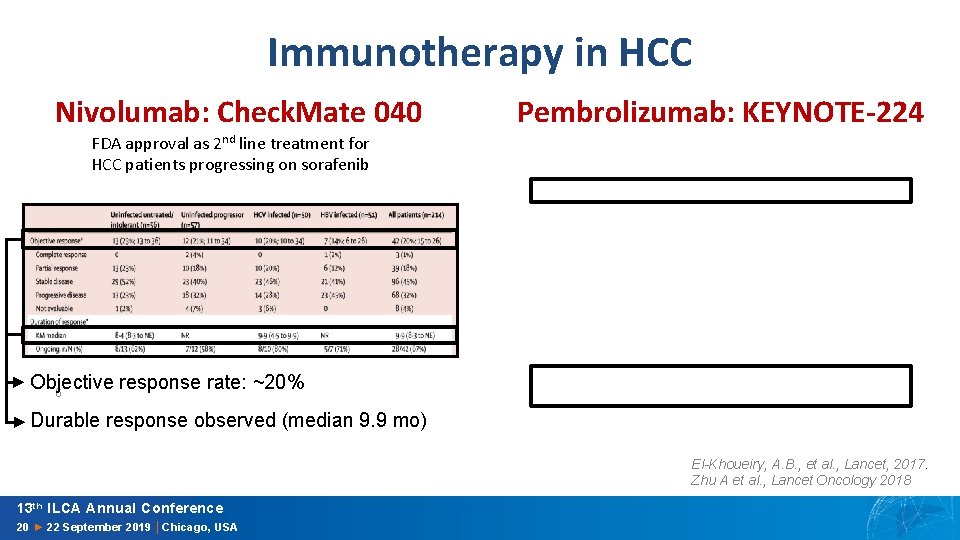
Immunotherapy in HCC Nivolumab: Check. Mate 040 Pembrolizumab: KEYNOTE-224 FDA approval as 2 nd line treatment for HCC patients progressing on sorafenib Objective response rate: ~20% 6 Durable response observed (median 9. 9 mo) El-Khoueiry, A. B. , et al. , Lancet, 2017. Zhu A et al. , Lancet Oncology 2018 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Biomarkers for Immunotherapy Treatment Response Nivolumab: Check. Mate 040 • Tumor PD-L 1 status by IHC • Check. Mate 040 trial: no association between treatment response and PD-L 1 status Pembrolizumab: KEYNOTE-224 • Tumor Proportion Score(43% vs 22% ORR; not significant) • Tumor/Immune Combined Score (32% vs 20% ORR, p=0. 021) Tumor PD-L 1 expression in HCC El-Khoueiry, A. B. , et al. , Lancet, 2017. Zhu A et al. , Lancet Oncology 2018 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

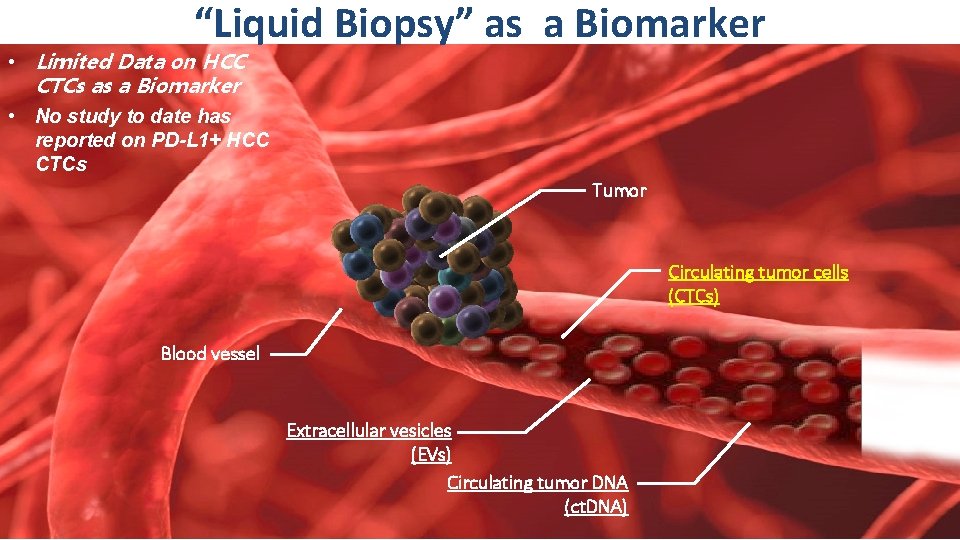
“Liquid Biopsy” as a Biomarker • Limited Data on HCC CTCs as a Biomarker • No study to date has reported on PD-L 1+ HCC CTCs Tumor Circulating tumor cells (CTCs) Blood vessel 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA Extracellular vesicles (EVs) Circulating tumor DNA (ct. DNA)

Specific Aims 1. Identify and enumerate HCC CTCs, and evaluate feasibility of phenotyping CTCs expressing PD-L 1 2. To evaluate the potential of PD-L 1+ CTCs to serve as a prognostic biomarker in discriminating early/advanced stage diseases and survival 3. Assess ability of PD-L 1+ CTCs as a predictive biomarker in a subset of patients undergoing anti-PD-1 therapy 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Our HCC CTC Approach: Nano. Velcro Blood Draw & Shipment (<72 h) 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA Depletion of RBCs And PBMC Banking (1. 5 hr) CTCs Capture in Nano. Velcro Chips (1 hr) Immunostaining 1 st Ab (Overnight) 2 nd Ab (45 min) Microscopy Imaging + Image Analysis /Archiving (10 min)

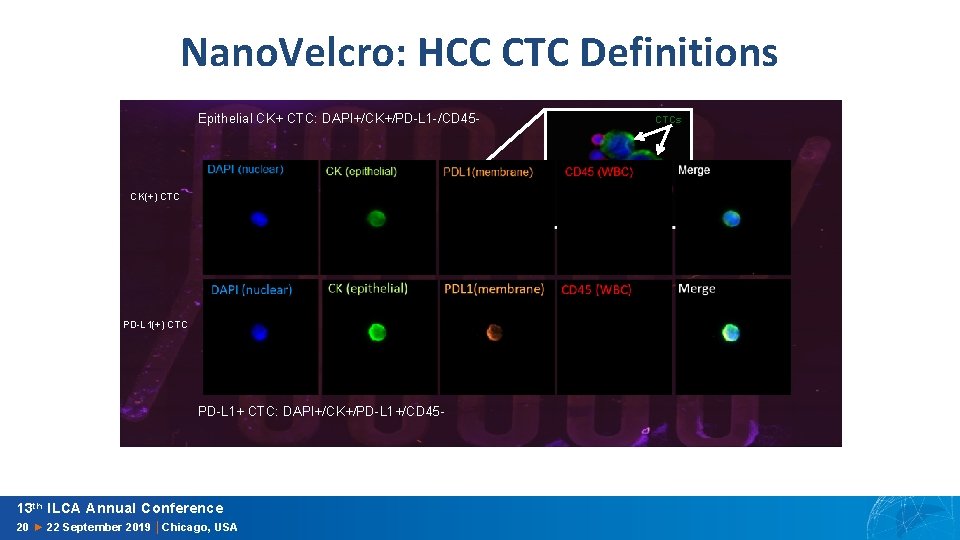
Nano. Velcro: HCC CTC Definitions Epithelial CK+ CTC: DAPI+/CK+/PD-L 1 -/CD 45 - CTCs CK(+) CTC WBCs PD-L 1(+) CTC PD-L 1+ CTC: DAPI+/CK+/PD-L 1+/CD 45 - 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

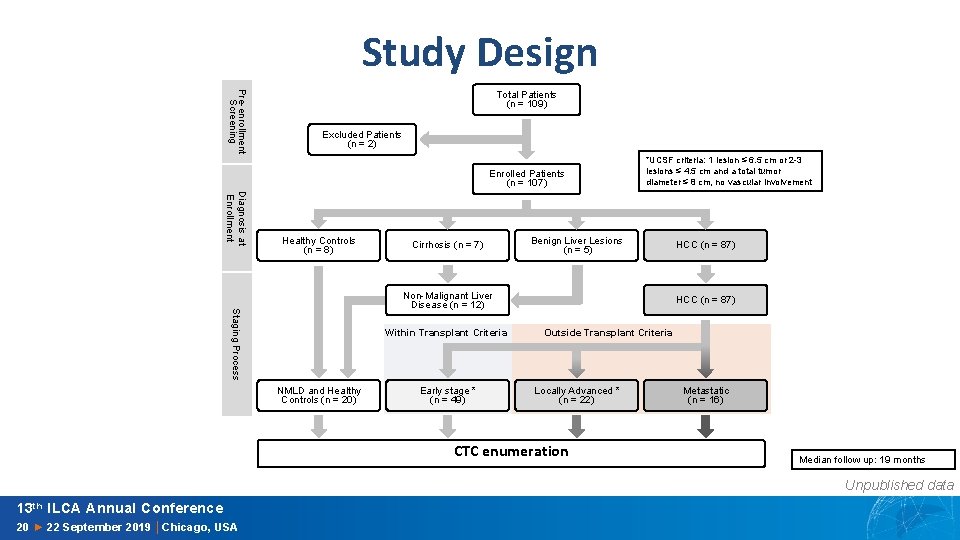
Study Design Pre-enrollment Screening Total Patients (n = 109) Excluded Patients (n = 2) Enrolled Patients (n = 107) Diagnosis at Enrollment Healthy Controls (n = 8) Cirrhosis (n = 7) *UCSF criteria: 1 lesion ≤ 6. 5 cm or 2 -3 lesions ≤ 4. 5 cm and a total tumor diameter ≤ 8 cm, no vascular involvement Benign Liver Lesions (n = 5) Staging Process Non-Malignant Liver Disease (n = 12) Within Transplant Criteria NMLD and Healthy Controls (n = 20) Early stage * (n = 49) HCC (n = 87) Outside Transplant Criteria Locally Advanced * (n = 22) CTC enumeration Metastatic (n = 16) Median follow up: 19 months Unpublished data 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

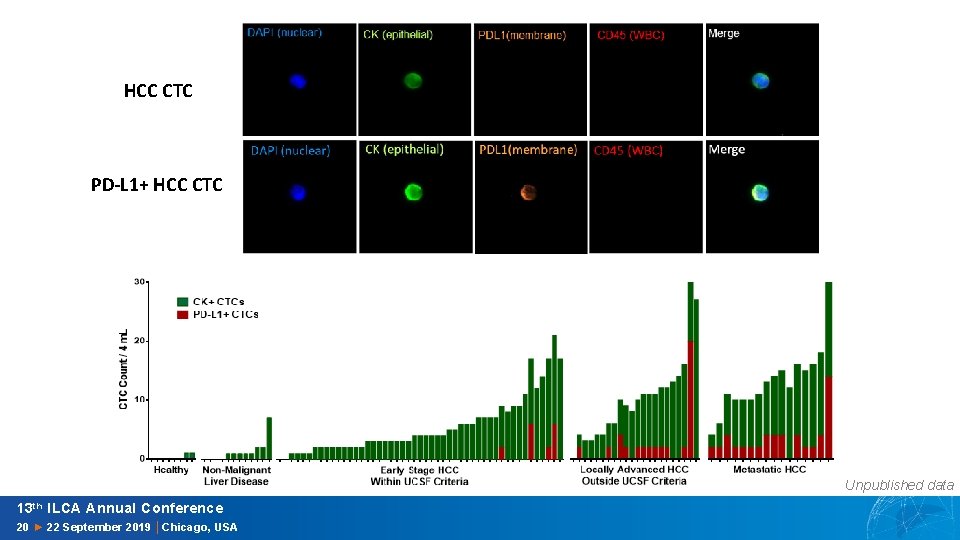
HCC CTC PD-L 1+ HCC CTC Unpublished data 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

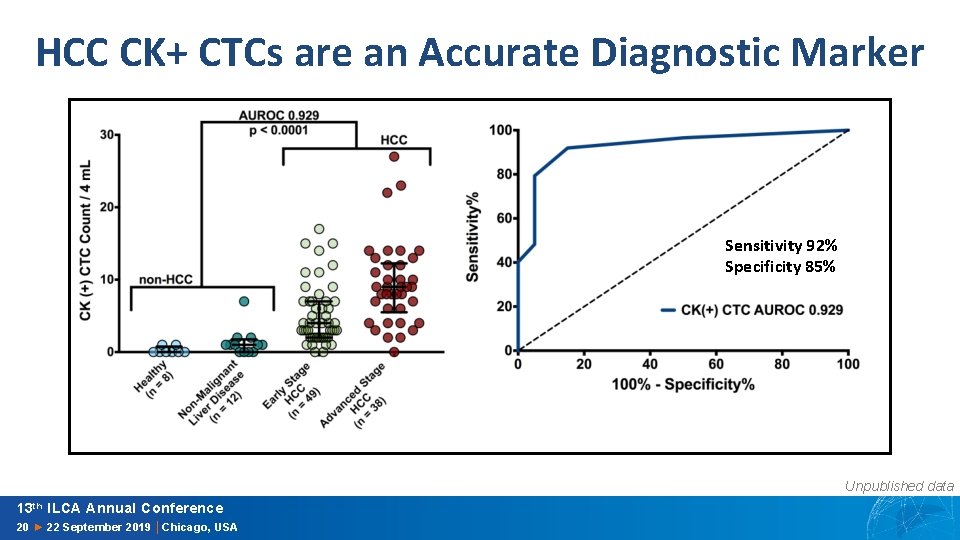
HCC CK+ CTCs are an Accurate Diagnostic Marker Sensitivity 92% Specificity 85% Unpublished data 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

PD-L 1+ CTCs are a Prognostic Biomarker Sensitivity 71% Specificity 92% 15 Unpublished data 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

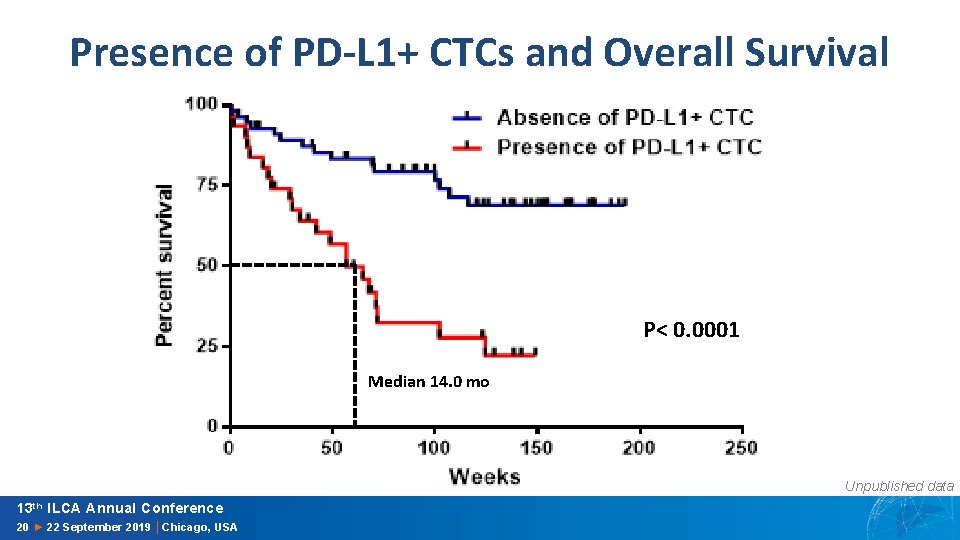
Presence of PD-L 1+ CTCs and Overall Survival P< 0. 0001 Median 14. 0 mo Unpublished data 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

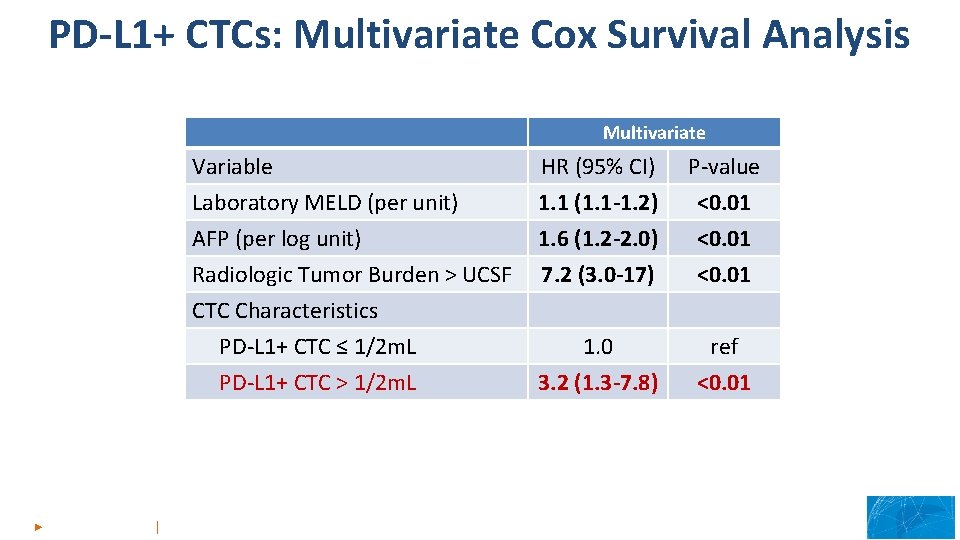
PD-L 1+ CTCs: Multivariate Cox Survival Analysis Multivariate Variable HR (95% CI) P-value Laboratory MELD (per unit) AFP (per log unit) Radiologic Tumor Burden > UCSF CTC Characteristics 1. 1 (1. 1 -1. 2) 1. 6 (1. 2 -2. 0) 7. 2 (3. 0 -17) <0. 01 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

PD-L 1+ CTCs: Multivariate Cox Survival Analysis Multivariate Variable HR (95% CI) P-value Laboratory MELD (per unit) AFP (per log unit) Radiologic Tumor Burden > UCSF CTC Characteristics 1. 1 (1. 1 -1. 2) 1. 6 (1. 2 -2. 0) 7. 2 (3. 0 -17) <0. 01 1. 0 3. 2 (1. 3 -7. 8) ref <0. 01 PD-L 1+ CTC ≤ 1/2 m. L PD-L 1+ CTC > 1/2 m. L 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

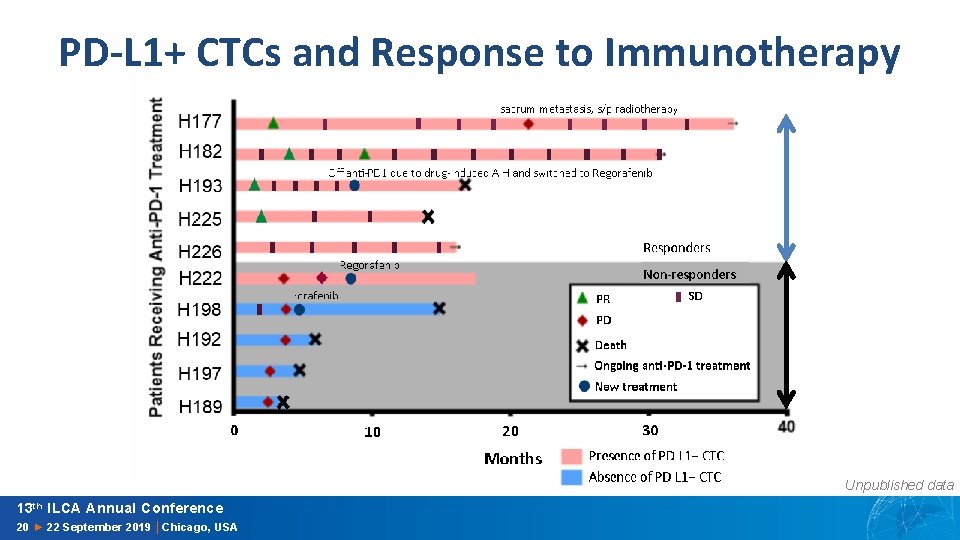
PD-L 1+ CTCs and Response to Immunotherapy Unpublished data 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Summary • First study to characterize PD-L 1+ CTC phenotyping in HCC • PD-L 1+ CTCs are prognostic: – Discriminate early stage/curable and advanced stage/incurable HCC – Independently portend poor survival after controlling for MELD, AFP, and tumor stage • PD-L 1+ CTCs are potentially predictive: – Associated with treatment response to anti-PD 1 treatment 20 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA

Agopian LAB: Collaborators: Amy Chen Shuang Hou Colin Court Paul Winograd Daniela Markovic Thank You UCLA/Dumont LCC: UCLA Dept of Surgery: Ronald Busuttil, MD Ph. D Douglas Farmer, MD Hsian-Rong Tseng UCLA Division of Medical and Molecular Pharmacology Richard Finn UCLA Division of Hematology-Oncology Saeed Sadeghi UCLA Division of Hematology -Oncology Funding: American Surgical Association Foundation NIH/NCI R 21 CA 216807 (Agopian -PI) NIH/NCI R 21 CA 235340 (Agopian- co. PI) NIH/NCI R 01 CA 218486 (Agopian – site PI) NIH/NCI R 01 CA 204145 (Agopian – site PI) 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA Fady Kaldas, MD Mia Camcam, NP LCC Coordinator Joseph Di. Norcia, MD