Minerals Composition and Physical and Chemical Properties Mineral





















- Slides: 24

Minerals Composition and Physical and Chemical Properties

Mineral Composition • Minerals are made of elements • Elements are made up of atoms • Atoms are the basic building blocks of all matter. They are invisible Atoms are organized on the Periodic Table of Elements

Atoms They contain 3 parts… Protons n n Found in the nucleus Positive charge Neutrons n n Found in the nucleus Charge of 0 Electrons n n Orbits the nucleus Negative charge

Element A substance in which all of the atoms are the same. The 6 most common elements on the earth are n n n P C H O N S Phosphorus Carbon Hydrogen Oxygen Nitrogen Sulfur A compound is a substance in which there are 2 or more types of atoms.

Periodic Table Where elements are arranged Atomic number n Number of protons Element abbreviation Atomic mass n Number of protons + number of neutrons

Symbols to know • Aluminum - Al • Calcium - Ca • Carbon - C • Chlorine - Cl • Copper - Cu • Helium - He • Hydrogen - H • Iron - Fe • Lead – Pb • Gold- Au • Argon- Ar • Magnesium - Mg • Mercury - Hg • Nitrogen - N • Oxygen - O • Potassium - K • Silicon - Si • Sodium - Na • Sulfur - S • Uranium – U Silver- Ag

Periodic Table Information Each element is made of all the same kind of atom Elements can combine together of form compounds which have a definite chemical formula Which elements combine depend on the electron configuration. Substances can combine to form mixtures.

Connection to Minerals are made up of a single element or a compound. Since compounds are combinations of elements, it is important to know how and why elements combine. The properties of minerals are due to the internal structure of the atoms. n Otherwise known as their crystal arrangement.

What are minerals? Minerals are aggregates of rocks. This means many different minerals make up rocks. Characteristics of minerals n n n Naturally occurring Inorganic Solids They have a specific chemical make-up Have a crystalline internal structure

Mineral Formation Minerals are formed in two different ways 1. Cooling of magma - This is the most common. The slower the rate of cooling the larger the crystal. 2. Evaporation of water - As it evaporates the water leaves the mineral behind.

Uses of minerals Copper is used for wiring Gold is used for jewelry Talc is used for baby powder Quartz is used for glass Limestone can be processed to make concrete Gems are brighter and more colorful than common minerals. This makes them rare and highly prized. Ores are minerals that contain useful substances that can be mined for profit. A common example is bauxite which is turned into aluminum.

Properties of Minerals Mineral Identification Physical and Chemical Over 3000 different minerals exist, but only 50 are common. These minerals are identified using 6 basic characteristics.

Hardness The measure of resistance to abrasion. Hardness is measured on a scale called Mohs Hardness scale. Talc is the softest mineral. Diamond is the hardest mineral Quartz is that hardest common mineral.

Color is not always the best characteristic to identify a mineral with. This is because a mineral can have many different colors due to irregularities.

Streak The color of the mineral in powder form. This can be determined by using a scratching plate. Metallic minerals leave a dark streak.

Luster The appearance or quality of light reflected from a minerals surface. The luster can be metallic or nonmetallic. If a mineral is nonmetallic it can have vitreous, pearly, resinous or earthy lusters.

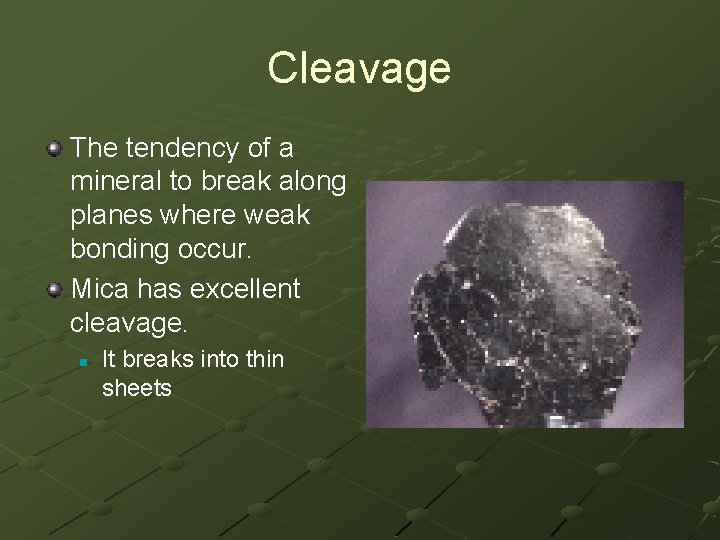
Cleavage The tendency of a mineral to break along planes where weak bonding occur. Mica has excellent cleavage. n It breaks into thin sheets

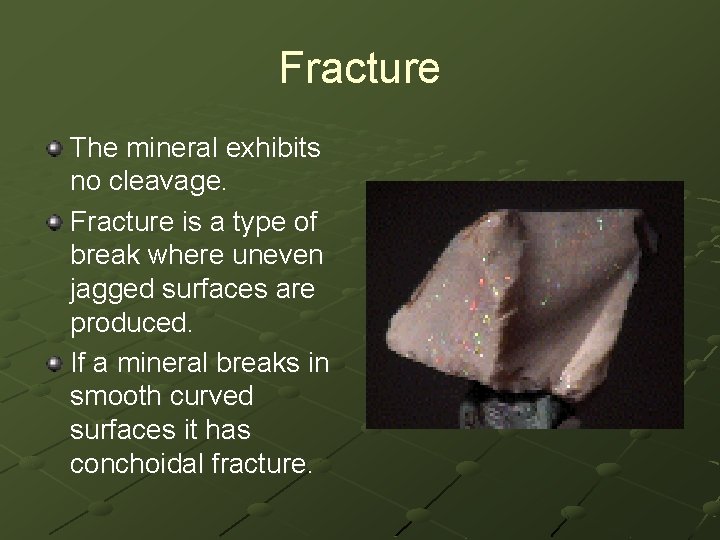
Fracture The mineral exhibits no cleavage. Fracture is a type of break where uneven jagged surfaces are produced. If a mineral breaks in smooth curved surfaces it has conchoidal fracture.

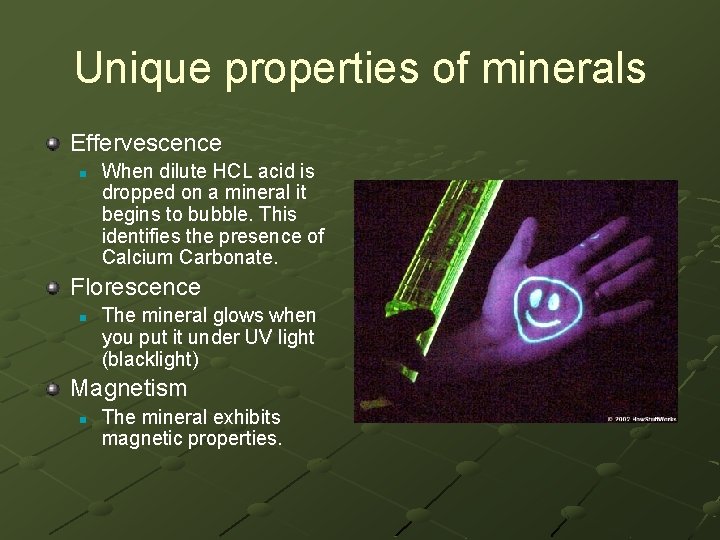
Unique properties of minerals Effervescence n When dilute HCL acid is dropped on a mineral it begins to bubble. This identifies the presence of Calcium Carbonate. Florescence n The mineral glows when you put it under UV light (blacklight) Magnetism n The mineral exhibits magnetic properties.

Halite tastes like salt.

Graphite has a greasy feel.

Magnetite is magnetic.

Calcite exhibits double refraction. This means if you put calcite on your writing you can see it twice.

Mineral Groups o 4000 minerals make up the Earth’s crust, but 98% of the crust is made up of only 8 elements. o Most rock forming minerals are silicates. o Silicates are minerals that contain silicon and oxygen and usually one or two more elements. o Other minerals are classified by their composition. Examples include carbonates, oxides, and halides.