Physical v Chemical Physical Properties vs Chemical Properties










- Slides: 11

Physical v. Chemical

Physical Properties vs Chemical Properties ØPhysical Properties – can be observed without changing the identity of the substance Ø Examples: odor, color, taste, volume, state, hardness, density, melting and boiling points

Physical Properties vs Chemical Properties �Chemical Properties – its ability to form new substances Ø Examples: flammability, oxidation/corrosion

What is oxidation? ? � Oxidation is the interaction between oxygen molecules and all the different substances they may contact, from metal to living tissue. � Freshly cut apple turning brown � Bicycle fender rusting � Penny suddenly turning green *Prevent oxidation? Need a layer of protection � https: //www. youtube. com/watch? v=p 3 P 0 mjeb 5 UU

Physical or Chemical Properties? 1. 2. 3. 4. Boiling Point of an alcohol Diamonds are hard Sugars ferments to form alcohol A metal wire conducts an electric current

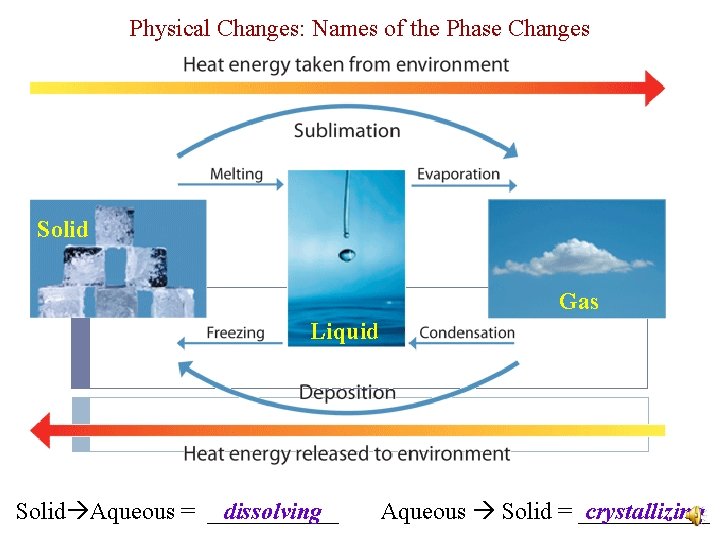
Physical Changes *Usually reversible (not always) �Physical Change – a change that does not affect the composition of a substance �Change like: � Crushing, �Most ripping, breaking or any phase change common physical (phase) changes: solid liquid gas *Chemical bonds are not broken

Physical Changes: Names of the Phase Changes Solid Gas Liquid Solid Aqueous = ______ dissolving Aqueous Solid = ______ crystallizing

Chemical Changes *Irreversible �Chemical Change – a change that a substance becomes a new, different substance. �Chemical changes are called reactions. �Changes like: � Burning, rusting, rotting or decomposing, fermenting, cooking, baking and other chemical reactions *Chemical bonds are broken and molecules rearrange to form new

Indications of a Chemical Reaction/Changes � Temperature � Gives Changes off heat (exothermic), absorbs heat (endothermic) � Produces a gas � Changes color �a new color is formed � Odor changes � Produces a solid (precipitate) https: //www. youtube. co m/watch? v=M 8 tyjw. B 42 X 4

Physical or Chemical Changes? Ice melting to water 2. Burning Paper 3. Baking a cake 4. Putting blue food color in water 1.

Exit Slip Label as physical or chemical change: 1. Making ice cubes 2. Salt dissolving in water