Chapter 5 The Periodic Table History u Russian

Chapter 5 The Periodic Table

History u Russian scientist Dmitri Mendeleev taught chemistry in terms of properties. u Mid 1800 – only atomic masse of elements were known. u Wrote down the elements in order of increasing mass. u Found a pattern of repeating properties.

u Grouped Mendeleev’s Table elements in columns by similar properties in order of increasing atomic mass. u Problems - felt that the properties were more important than the mass, so switched order. u Found some gaps - Must be undiscovered elements. u Predicted found. their properties before they were

u Elements The modern table are still grouped by properties similar properties are in the same column. u Order is in increasing atomic number. u Added a column of elements Mendeleev didn’t know about – Noble Gases. u The noble gases weren’t found because they didn’t react with anything.
u Horizontal rows are called periods u There are 7 periods

u Vertical columns are called groups or families. u Elements are placed in columns by similar properties.

The elements in the A groups are called the representative elements or main group 1 A 8 A elements 2 A 3 A 4 A 5 A 6 A 7 A u
The group B are called the transition elements u These are called the inner transition elements and they belong here
u Group 1 A are the alkali metals - most reactive u Group 2 A are the alkaline earth metals
u Group 7 A is called the Halogens u Group 8 A are the noble gases

s 1 S- block s 2 u Alkali metals all end in s 1 u Alkaline earth metals all end in s 2 u really have to include He but it fits better later. u Helium has the properties of the noble gases.

Transition Metals -d block d 1 d 2 d 3 s 1 d 5 d 6 d 7 d 8 d 10
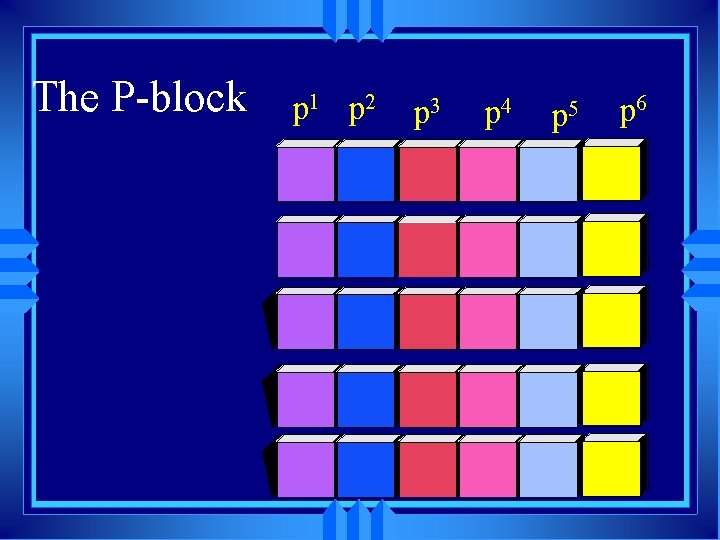
The P-block p 1 p 2 p 3 p 4 p 5 p 6
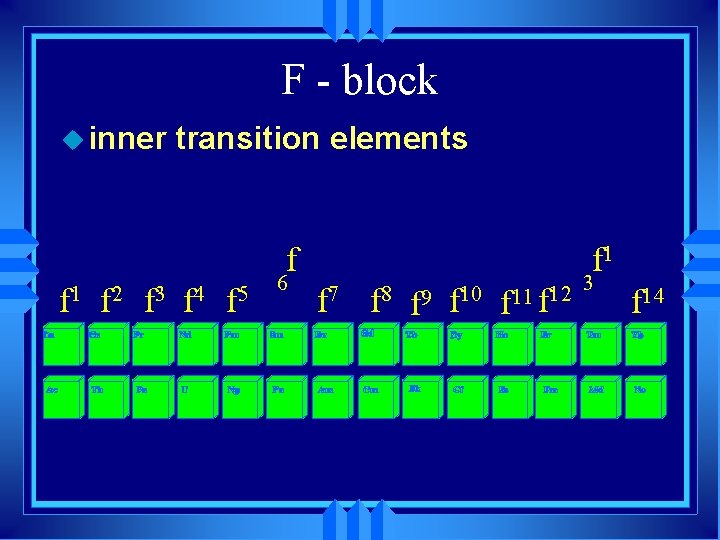
F - block u inner transition elements f f 1 f 2 f 3 f 4 f 5 6 f 1 f 7 f 8 f 9 f 10 f 11 f 12 3 f 14

Atomic Size u First problem - where do you start measuring. u The electron cloud doesn’t have a definite edge. u They get around this by measuring more than 1 atom at a time.

Atomic Size } Radius u. Atomic Radius = half the distance between two nuclei of a diatomic molecule.

Trends in Atomic Size u. Influenced by two factors. • Energy Level - Higher energy level is further away from nucleus. • Charge on nucleus - More charge (more protons) pulls electrons in closer.
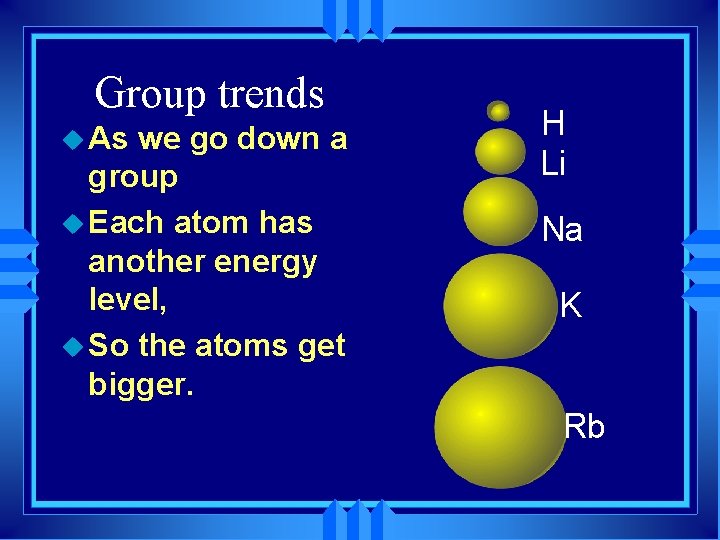
Group trends u As we go down a group u Each atom has another energy level, u So the atoms get bigger. H Li Na K Rb

Periodic Trends u As you go across a period the radius gets smaller. u Same energy level. u More nuclear charge. u Outermost electrons are pulled closer to nucleus. Na Mg Al Si P S Cl Ar

Ionization Energy u The amount of energy required to completely remove an electron from a gaseous atom. u Removing one electron makes a +1 ion. u The energy required is called the first ionization energy.

Ionization Energy u The second ionization energy is the energy required to remove the second electron. u Always greater than first IE. u The third IE is the energy required to remove a third electron. u Greater than 1 st or 2 nd IE.

What determines IE u The greater the nuclear charge the greater IE. u The smaller the atom the higher the IE. u Filled and half filled orbitals have lower energy, so achieving them is easier, lower IE. u Shielding
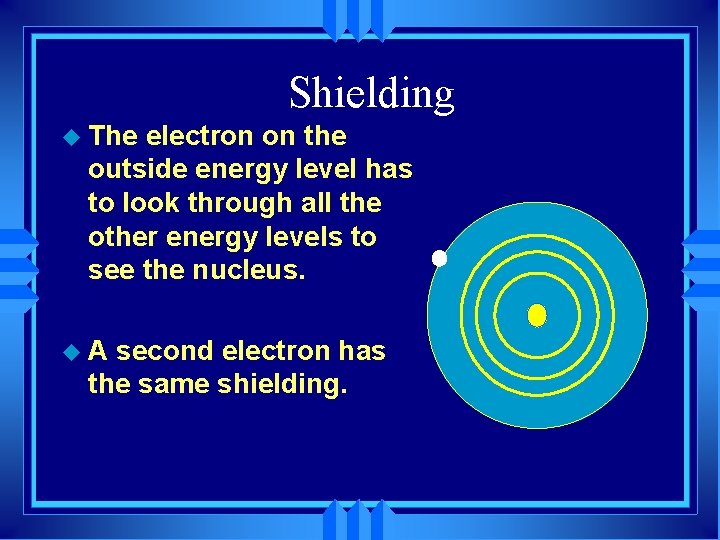
Shielding u The electron on the outside energy level has to look through all the other energy levels to see the nucleus. u. A second electron has the same shielding.

Group trends u. As you go down a group first IE decreases because the electron is further away from the nucleus. u. More shielding.

Periodic trends u All the atoms in the same period have the same energy level. • Same shielding. • Increasing nuclear charge • So IE generally increases from left to right. u Exceptions at full and 1/2 fill orbitals.

What Causes the Trends u Full Energy Levels are very low energy. u Noble Gases have full orbitals. u Atoms behave in ways to achieve noble gas configuration.

Electron Affinity u The energy change associated with adding an electron to a gaseous atom. u Easiest to add to group 7 A - gets them to full energy level. u Increase from left to right atoms become smaller, with greater nuclear charge. u Decrease as we go down a group.

Ionic Size u Cations form by losing electrons. u Cations are smaller than the atom they come from. u Metals form cations. u Cations of representative elements have noble gas configuration.

Ionic size u Anions form by gaining electrons. u Anions are bigger than the atom they come from. u Nonmetals u Anions form anions. of representative elements have noble gas configuration.

Group trends u Adding energy level u Ions get bigger as you go down. Li+1 Na+1 K+1 Rb+1 Cs+1

Periodic Trends u Across the period nuclear charge increases so they get smaller. u Energy level changes between anions and cations. Li+1 B+3 Be+2 C+4 N-3 O-2 F-1

Electronegativity

Electronegativity u The tendency for an atom to attract electrons to itself when it is chemically combined with another element. u How fair it shares - large electronegativity means it pulls the electron toward it. u Atoms with large negative electron affinity have larger electronegativity.

Group Trend u The further down a group the farther the electron is away from nucleus and the more electrons an atom has which increases shielding. u More u Low willing to share. electronegativity.

Periodic Trend u Metals (left side) - let their electrons go easily • Low electronegativity u Nonmetals (right side) - they want more electrons. • High electronegativity.
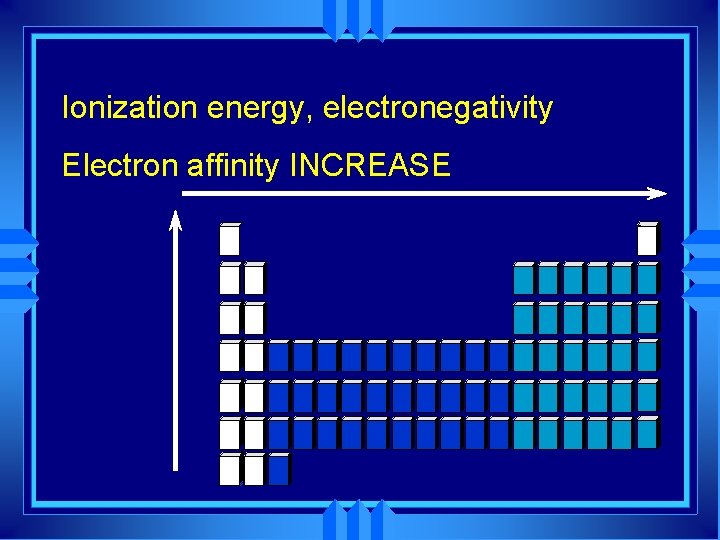
Ionization energy, electronegativity Electron affinity INCREASE
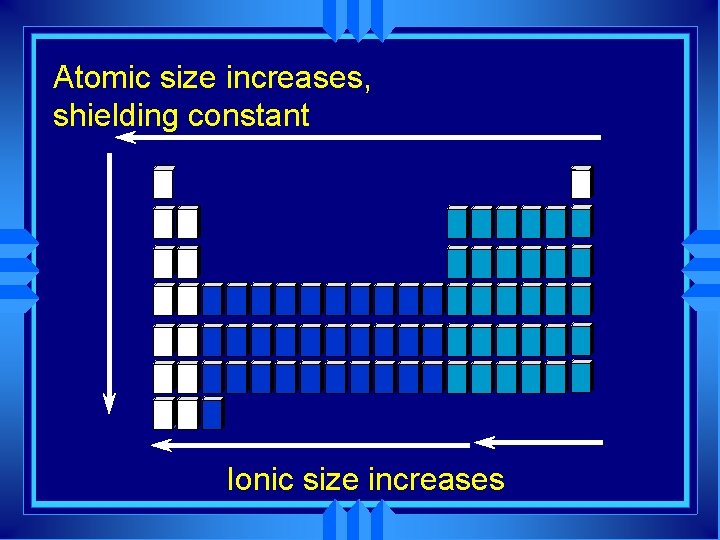
Atomic size increases, shielding constant Ionic size increases

Chemical and Physical Properties

Descriptive Chemistry u The study of elements and the compounds they form.

Hydrogen u Lightest Element u Most Abundant in the Entire Universe u Henry Cavendish – First to systematically collect and study. u Hydrogen – “water-former” because it produces water when combusted in air.

Hydrogen u Physical • • Colorless Odorless Tasteless Least dense High speed Diffuse quickly Little attraction u Chemical • Diatomic • If split – highly flammable • Acts like 1 A and 7 A • NH 3 – ammonia • HCl – Hydrochloric acid • Metallic hydrides

u Commercial Sources • Byproduct of fuel processes • Electrolysis • Passing steam over heated coke (impure carbon obtained from coal) u Uses • Mostly ammonia • Rocket fuel • Hydrogenated oil

Alkali Metals u Most reactive metals. u Most isolated by Sir Humphrey Davy in 1807. u Form cations with +1 charge by losing one electron; 1 valence electron; s-block u Sodium – • most abundant alkali metal • 6 th most common element in the earth’s crust. u Never found as pure metals, only combined in nature.

Alkali Metals u Physical • • • Good conductors Bright luster Low densities Soft Easily cut u Chemical • Very reactive • React violently with water to produce bases • Stored in oil to prevent reaction with oxygen or water in air

u Uses • Sodium vapor street lights • Na/K pump • Baking soda • Soap • Rayon • Paper • Salt substitutes Lithium batteries Lithium makes lubricants water resistant and extreme temperature resistant Medication

Alkaline Earth Metals u Mostly metal-oxygen compounds which are mostly geologic minerals that dissolve slightly in water. u More dense, harder, and have higher melting points than alkali metals. u Form +2 cations by losing 2 electrons; 2 valence electrons u s-block element

Alkaline Earth Metals-Elements u Beryllium – important in minerals; emerald u Magnesium – 8 th most common in earth’s crust; important component in chlorophyll u Calcium – 5 th most abundant in earth’s crust; major component of bones and teeth u Radium – naturally radioactive and luminesces

Alkaline Earth Metals u Physical • Dull exterior with shiny interior • Harder metal • Low density • Malleable • Good conductors • Hard water u Chemical • Reactivity increases as atom gets larger u Uses • X-ray tubes • Lightweight alloys • Flares • Plaster • Medicine • Fireworks

Transition Metals u d-block elements u Form varying ions both positive and negative. u All have 2 valence electrons

Transition Metals u Physical • • Typical metals High density Shiny luster Good conductors Malleable Ductile Solids at room temperature • Ex. Mercury – liq. u Chemical • Nonreactive can resist corrosion • Copper and iron corrode quickly • Iron, copper, and nickel produce magnetic fields u Uses • Construction • Medical

Inner Transition Metals u Lanthanide and Actinide Series u f-block elements u 2 valence electrons

Inner Transition Metals u Physical • Paramagnetic • Many radioactive actinides • Many isotopes of actinides u Uses • Lathanides – Catalysts & glass and TV tube industries u Chemical • Emit tissue destroying and cancer producing rays • > 1/1, 000 g kills u Uses • Actinides – fission; nonexplosive fuel in nuclear reactors

Metalloids boron, silicon, arsenic, antimony, germanium, tellurium, astatine u Physical • Semi-conductors • More metallic as you go down the group u Chemical • Found in ores • As oxidation occurs Al forms an impenetrable coating of protection • 5 A-most dramatic change in properties

The Boron Family – Group 13 u A. K. A – Post-Transition metals (except Boron – metalloid) u More metallic as you go down the group. u Boron and Aluminum most important economically. u Aluminum is most common element in earth’s crust

The Boron Family u u Physical - Boron • crumbly black solid • Transmits portions of infrared spectrum • Conducts electricity at high temps. Physical – Aluminum • High strength • Low density • Lustrous • Highly conductive u Chemical • Found in ores • As oxidation occurs Al forms an impenetrable coating of protection • 5 A-most dramatic change in properties • Boron – behaves like silicon also

The Boron Family u Uses – Boron • Oxide compound used to make fiberglass. • Borax – softens water and cleans clothes • Minimizes thermal expansion in glass u Uses – Aluminum • Aircraft, kitchen utensils, lawn chairs, pop cans • Corrosion proof reflective coating when combined with oxygen • Aluminum oxide used in sandpaper

The Carbon Family – Group 14 u Carbon – only true nonmetal in group u Carbon the most important element in this group; basis for life u More metallic as you go down the group

The Carbon Family u Physical • Solids at room temp • Carbon has four forms: carbon, graphite, diamond u Chemical • Four valence electrons • Multiple bonding • Low reactivity • Combusts in oxygen • Less reactive as you go down the group

The Carbon Family u Uses • • - Carbon Amorphous carbon – electrodes Graphite – lubricant, binder in paints, writing Diamond – gems; cutting Charcoal - filters u Uses – Silicon & Others • Computer industry • Semi-conductors • Tin + Copper makes bronze

The Nitrogen Family – Group 15 u Nitrogen – gas at room temp; the rest are solids u More metallic as you go down; nitrogen and phosphorus only non metals in group u 5 valence electrons

The Nitrogen Family u Physical • Nitrogen is diatomic • 78% volume in earth’s atmosphere • Occur as four different forms identifiable by color • White phosphorus is phosphorescent u Chemical • Nitrogen – inert; Triple bonded • White phosphorus – very reactive; burns spontaneously in oxygen; grenades in Vietnam; very poisonous

The Nitrogen Family u Uses - Nitrogen • Inert gas used to create unreactive environments • Used to produce ammonia u Uses – phosphorus • Used for matches, fireworks, and signal flares • Fertilizer • Basis for phosphoric acid

The Chalcogens – Group 16 u AKA - The Oxygen Family u 2 non-metals; 2 metalloids; 2 metals(? ) u 6 valence electrons u Oxygen means “acid producer” u Polonium – first element discovered by Marie Curie in her research (1898)

The Chalcogens u Physical • • - Oxygen 21% of earth’s atmosphere Slightly soluble in water Three bonded oxygen – Ozone (layer in atmosphere that protects) Unique properties from rest of family u Chemical • Oxygen one of the most reactive; 2 nd highest electronegativity • Forms oxides • Sulfur forms sulfides

The Chalcogens u Uses – Oxygen • Sustains life for all except anaerobic bacteria • Supports all forms of combustion u Uses • Sulfur is used to make sulfuric acid Selenium – treating dandruff; photocopying • Polonium – radioactive uses; power source used in space probes and satellites

The Halogens – Group 17 u Most reactive non-metals (astatine considered a metalloid) u 7 valence electrons u Bromine liquid at room temperature all others gases u Fluorine – highest electronegativity u Very poisonous u Astatine - radioactive

The Halogens u Physical • Density, MP, BP increase as you go down group • Color darkens as you go down group u Chemical • Form salts when they react with active metals • Diatomic elements (ex. At) • Accept electrons to become stable

The Halogens u Uses • Fluorine – reinforces tooth enamel • Swimming pools • Medical • Lamps • Photographic solutions

The Noble Gases – Group 18 u Do not react except under unusual circumstances of pressure and temp u Hard to discover due to the lack of reactivity

Noble Gases u Physical • • • Colorless Odorless Tasteless Low BP and FP Little attraction Low density u Chemical • STABLE • Low reactivity u Uses • Balloons • Lighted signs • Lights
- Slides: 70