Carbohydrates Lecture 3 Chemical Properties of Monosaccharides Reducing

Carbohydrates Lecture 3

Chemical Properties of Monosaccharides

Reducing Sugars Reducing sugars • Are monosaccharides with a carbonyl group that oxidizes to give a carboxylic acid. • Undergo reaction with Benedict’s reagent (Cu 2+) to give the corresponding carboxylic acid. • Include the monosaccharides glucose, galactose, and fructose.
![Oxidation of D-Glucose is oxidized to a carboxylic acid Benedicts reagent [O] Cu+ (reduced Oxidation of D-Glucose is oxidized to a carboxylic acid Benedicts reagent [O] Cu+ (reduced](http://slidetodoc.com/presentation_image/dc2cb2d310d08860351b814f11ac73f1/image-4.jpg)
Oxidation of D-Glucose is oxidized to a carboxylic acid Benedicts reagent [O] Cu+ (reduced form) Glucose is a reducing sugar

• A reducing sugar is any sugar that either has an aldehyde group or is capable of forming one in solution through isomerism. • Reducing sugars can be oxidized by relatively mild oxidizing agents such as cupric ion (Cu 2+). The carbonyl carbon (aldehyde group) is oxidized to a carboxylic acid, at the same time; cupric ion (Cu 2+) is reduced to cuprous ion (Cu 1+). • Sugars with ketone groups in their open chain form are capable of isomerizing via a series of tautomeric shifts to produce an aldehyde group in solution. • ketone-bearing sugars like fructose are considered reducing sugars but it is the isomer containing an aldehyde group which is reducing since ketones cannot be oxidized without decomposition of the sugar.
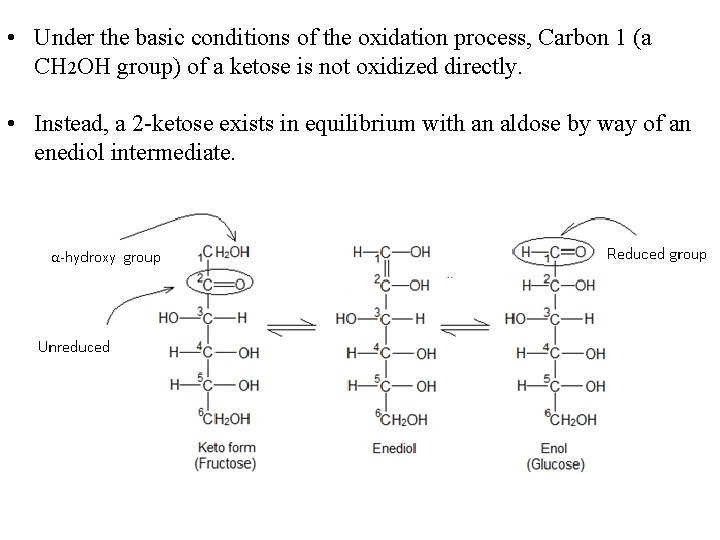
• Under the basic conditions of the oxidation process, Carbon 1 (a CH 2 OH group) of a ketose is not oxidized directly. • Instead, a 2 -ketose exists in equilibrium with an aldose by way of an enediol intermediate.

Reduction of Monosaccharides The reduction of monosaccharides • Involves the carbonyl group. • Produces sugar alcohols called alditols. • Such as D-glucose gives D-glucitol also called sorbitol. D-Glucitol
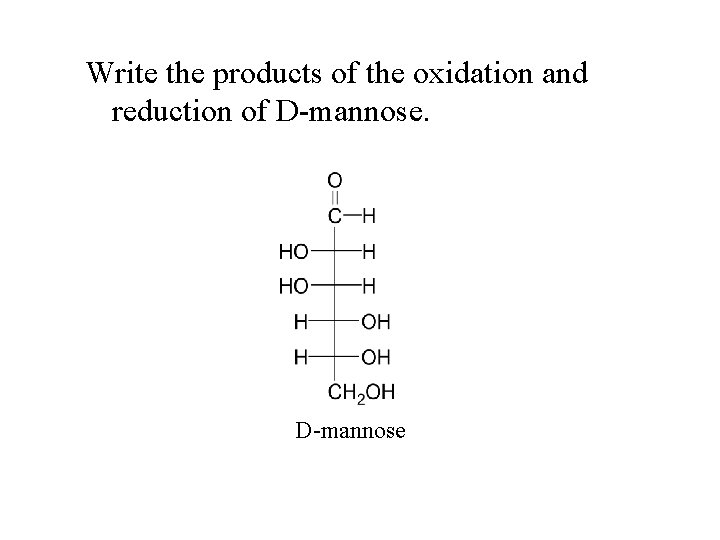
Write the products of the oxidation and reduction of D-mannose
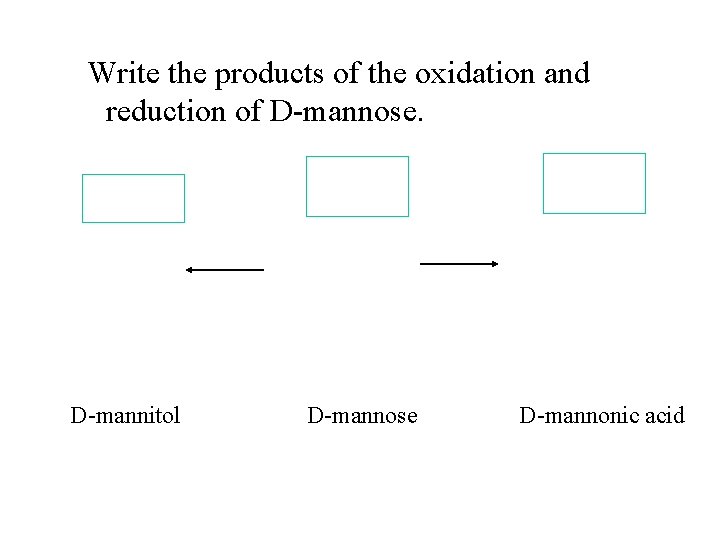
Write the products of the oxidation and reduction of D-mannose. D-mannitol D-mannose D-mannonic acid

oligosaccharides • oligosaccharides are carbohydrates formed of 2 -10 monosaccharides units covalently bonded to each other by glycosidic bonds. • according to number of sugar units they are classified into disaccharides, trisaccharides and so on. • This figure represents the chemical structure of Raffinose, which is a trisaccharide composed of galactose, glucose and fructose. • Raffinose can be found in peas, beans, cabbage, broccoli, other vegetables and whole grains.
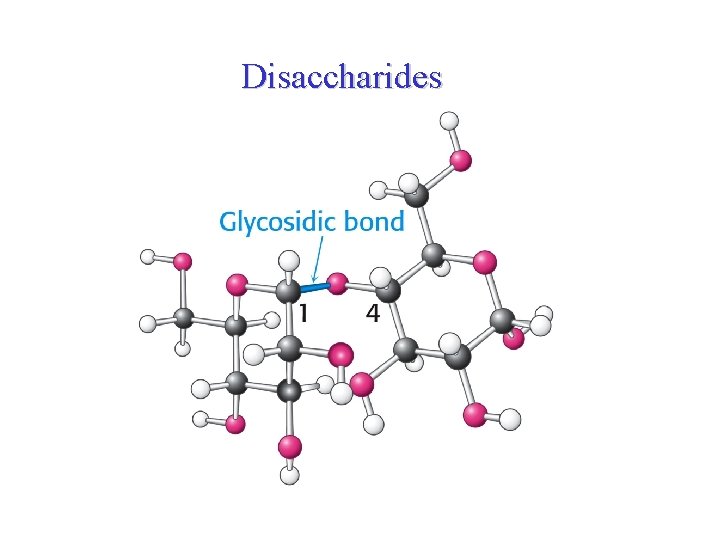
Disaccharides

Disaccharides • Disaccharides are oligosaccharides formed of 2 monosaccharides units covalently bonded by glycosidic bonds. • The most physiologically important disaccharides are maltose, lactose and sucrose. • The hydroxyl group formed as a result of hemiacetal formation is highly reactive when compared to other hydroxyl groups. • This hydroxyl group present in one monosaccharide reacts with any one of the hydroxyl groups attached to C-1, C-2, C-3, C-4, or C-6 of another monosaccharide to produce 1→ 1, 1→ 2, 1→ 3, 1→ 4, and 1→ 6 linked disaccharides.

Disaccharides • When only one anomeric carbon is involved in glycosidic bond formation, reducing disaccharides are formed. • If both anomeric carbon atoms of monosaccharides are involved in glycosidic bond formation that results in the formation of a nonreducing disaccharides such as sucrose. • In the case of reducing disaccharides, one end of the molecule having free anomeric carbon is called reducing end and the other end, where the anomeric carbon is involved in glycosidic bond, is called as non-reducing end.

Maltose • Maltose is a disaccharide made up of two glucose residue joined by a glycosidic linkage between C-1 of one glucose residue and C-4 of the other. • The configuration of the anomeric carbon of glucose involved in the linkage is alpha and hence the glycosidic linkage is (1→ 4).
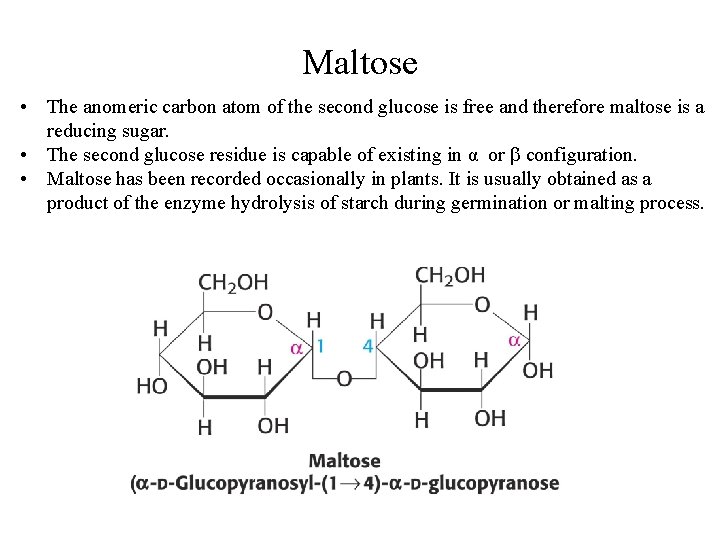
Maltose • The anomeric carbon atom of the second glucose is free and therefore maltose is a reducing sugar. • The second glucose residue is capable of existing in α or configuration. • Maltose has been recorded occasionally in plants. It is usually obtained as a product of the enzyme hydrolysis of starch during germination or malting process.
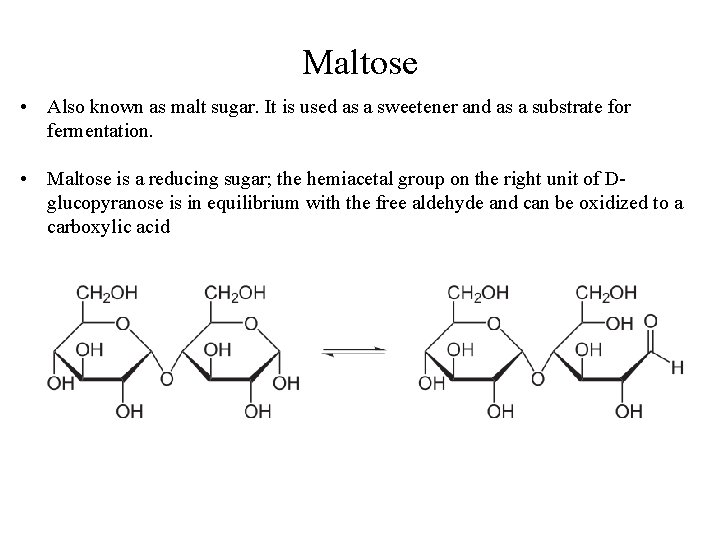
Maltose • Also known as malt sugar. It is used as a sweetener and as a substrate for fermentation. • Maltose is a reducing sugar; the hemiacetal group on the right unit of Dglucopyranose is in equilibrium with the free aldehyde and can be oxidized to a carboxylic acid
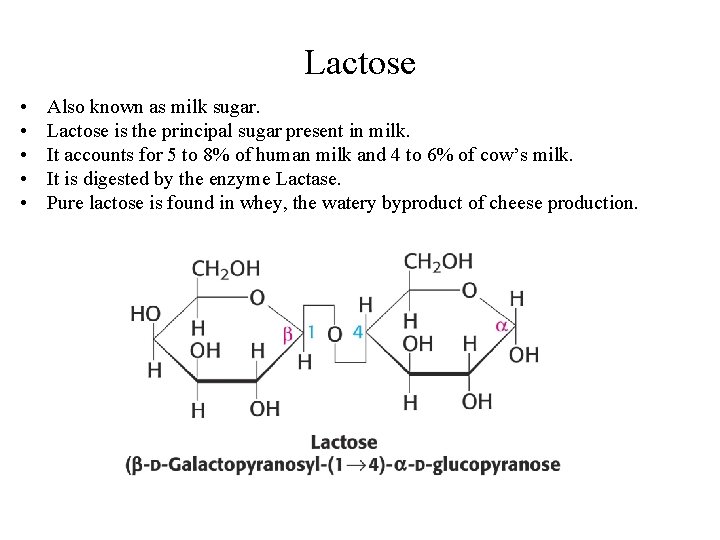
Lactose • • • Also known as milk sugar. Lactose is the principal sugar present in milk. It accounts for 5 to 8% of human milk and 4 to 6% of cow’s milk. It is digested by the enzyme Lactase. Pure lactose is found in whey, the watery byproduct of cheese production.
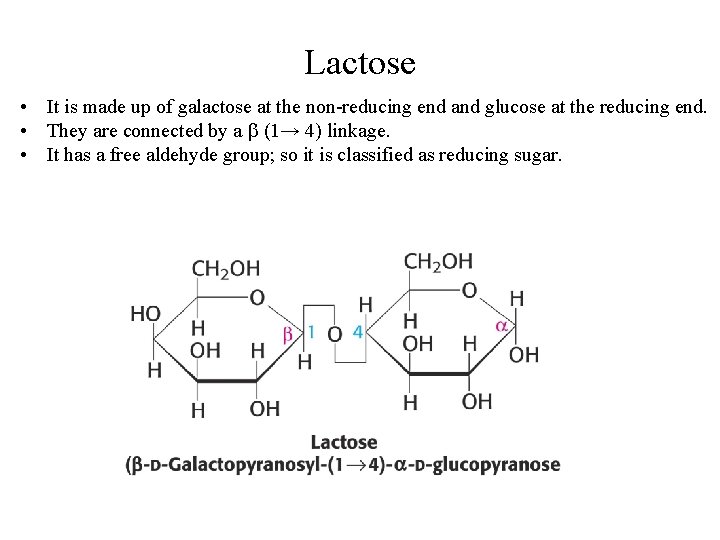
Lactose • It is made up of galactose at the non-reducing end and glucose at the reducing end. • They are connected by a (1→ 4) linkage. • It has a free aldehyde group; so it is classified as reducing sugar.

Sucrose • Sucrose (also known table sugar) is the most abundant disaccharide in the biological world. • It is found in fruits, sugar cane, and sugar beets. • Caramel is the solid residue formed from heating sucrose. • A flavoring agent called invert sugar is produced by the hydrolysis of sucrose under acidic conditions, which breaks it apart into glucose and fructose; invert sugar is sweeter than sucrose because of the fructose. • Some of the sugar found in honey is formed in this fashion; invert sugar is also produced in jams and jellies prepared from acid-containing fruits.
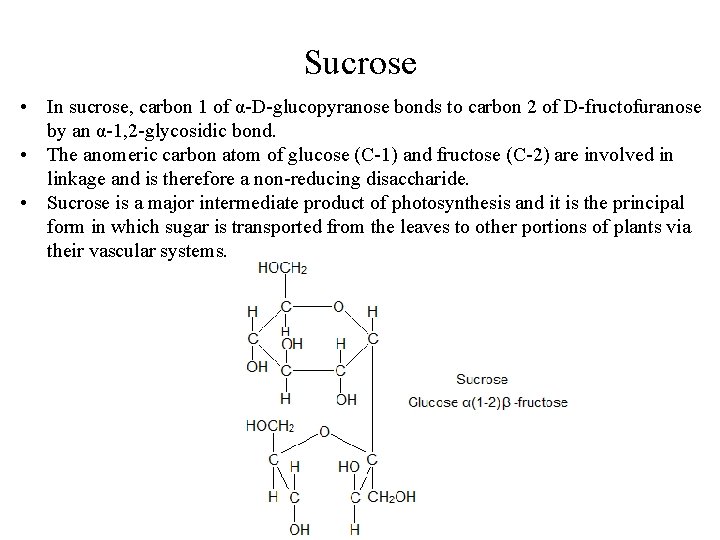
Sucrose • In sucrose, carbon 1 of α-D-glucopyranose bonds to carbon 2 of D-fructofuranose by an α-1, 2 -glycosidic bond. • The anomeric carbon atom of glucose (C-1) and fructose (C-2) are involved in linkage and is therefore a non-reducing disaccharide. • Sucrose is a major intermediate product of photosynthesis and it is the principal form in which sugar is transported from the leaves to other portions of plants via their vascular systems.

Disaccharides • • Lactose hydrolysed by lactase Maltose hydrolysed by maltase Sucrose hydrolysed by sucrase All these enzymes found on surface of epithelial cells lining small intestine
- Slides: 21