Minerals Properties of Minerals What is a Mineral



























- Slides: 28

Minerals Properties of Minerals

What is a Mineral? A mineral must have the following characteristics: • Naturally Occurring A substance must be formed by processes in the natural world and not created my man in a laboratory. Which ice is considered a mineral an ice cube or ice formed on a mountain? Why?

Inorganic Is salt a mineral? • The mineral cannot form from materials that were once part of a living thing. Is sugar a mineral? Is coal a mineral?

Solid • A mineral is always a solid, with a a definite volume and shape.

Crystal Structure • The particles of a mineral line up in a pattern that repeats over and over again. • This repeating pattern of a mineral’s particles forms a solid called a crystal

Groups of crystals • • • Cubic (magnetite) Hexagonal (quartz) Tetragonal (rutile) Orthorhombic (sulfur) Monoclinic (azuirite Triclinic (feldspar)

Definite Chemical Composition • A mineral always contains certain elements in definite proportions. • Most minerals are chemical compounds made of two or more elements. • Almost all pure, solid elements are metals. Copper Silver Gold

Identifying Minerals • There about 3, 800 minerals. • Each mineral has characteristic properties that can be used to identify it.

Color Gold • Easily observed physical property. • Color alone is NOT a good way to make an identification. Pyrite Chalcopyrite

Streak • The streak of a mineral is the color of its powder. • Rub a mineral against a piece of unglazed porcelain tile. • Color of minerals may vary but streak is always the same.

Luster • Luster describes how light is reflected from a mineral’s surface. • Luster terms: metallic; glassy; submetallic or dull; silky, earthy; waxy, greasy or pearly

Luster of Minerals Metallic Glassy Galena Topaz Waxy, greasy or pearly Talc Submetallic or Dull Silky Earthy Graphite Malachite Hermatite

Density • Density is a property of how much matter there is in a given amount of space. • No matter the size of a mineral sample, the density of that mineral always remains the same. Density Formula Density= Mass Volume

Cleavage • The way a mineral breaks • apart can help to identify it. • Cleavage is the tendency of a mineral to cleave, or break, along flat, even surfaces. • The arrangement of atoms in the mineral causes it to break apart more easily in one direction than another. • Some minerals have cleavage in more than one direction.

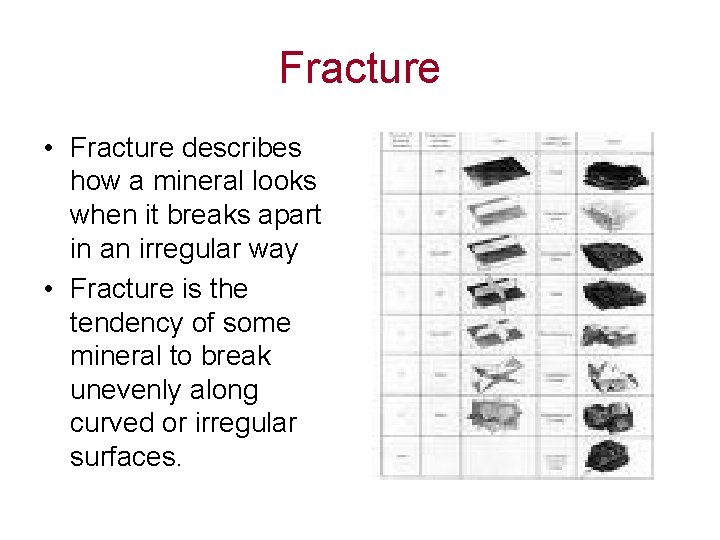
Fracture • Fracture describes how a mineral looks when it breaks apart in an irregular way • Fracture is the tendency of some mineral to break unevenly along curved or irregular surfaces.

Hardness • Hardness is a measure of the resistance of a mineral to being scratched. • Friedrich Mohs (1812) created a test to describe the hardness of minerals. • Moh’s Hardness Scale consists of 10 minerals arranged from 10 (hardest) to 1 (softest). • A mineral can scratch any mineral softer than itself, but can be scratched by any mineral that is harder.

Moh’s Hardness Scale 1. TALC The softest known mineral, talc flakes when scratched by a fingernail.

Moh’s Hardness Scale 2. Gypsum A fingernail can easily scratch it.

Moh’s Hardness Scale 3. Calcite A fingernail cannot scratch it, but a copper penny can.

Moh’s Hardness Scale 4. Fluorite A steel knife can easily scratch it.

Moh’s Hardness Scale 5. Apatite A steel knife can scratch it.

Moh’s Hardness Scale 6. Feldspar It can’t be scratched by a steel knife, but it can scratch window glass.

Moh’s Hardness Scale 7. Quartz It can scratch steel and hard glass easily.

Moh’s Hardness Scale 8. Topaz It can scratch quartz.

Moh’s Hardness Scale 9. Corundum It can scratch topaz.

Moh’s Hardness Scale 10. Diamond The hardest known mineral, diamond can scratch all other substances.

Special Properties • Some properties are particular to only a few types of minerals. • These properties include: – Fluorescence: Does it glow under ultraviolet light? – Chemical Reaction: Will a mineral effervesce or “fizz” when a drop of weak acid is placed on it? – Optical properties: Will the mineral bend light to produce a double image? – Taste: Does the mineral taste salty? – Magnetism: Will the mineral attract iron?

Fluorescence: Scheelite glows in ultraviolet light. Magnetism: Magnetite attracts these iron metals Optical Properties: Calcite bends light to produce a double image Reactivity: Aragonite reacts chemically to acids.