KF Coulometry Volumetric Coulometric Titration Volumetric Karl Fischer















- Slides: 25

KF Coulometry

Volumetric / Coulometric Titration Volumetric Karl Fischer Iodine is added by burette during titration. Water as a major component: 100 ppm 100 % - + Coulometric Karl Fischer Iodine is generated electrochemically during titration. Water in trace amounts: 1 ppm - 5 % 2021/9/16 AA 2

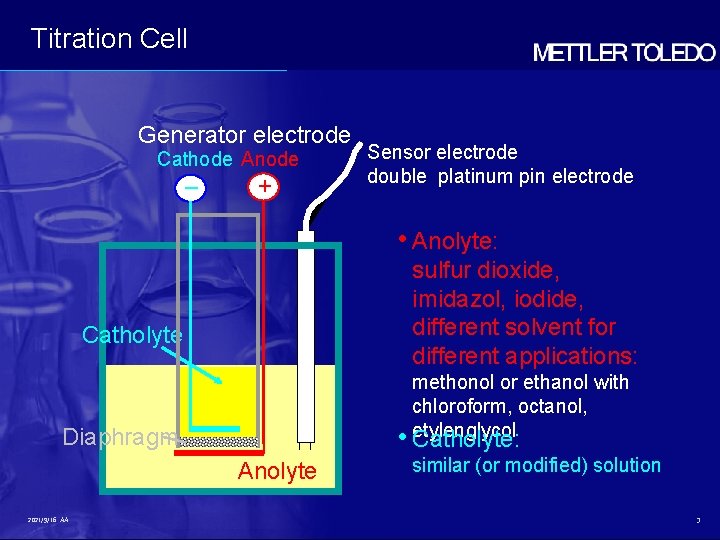
Titration Cell Generator electrode Cathode Anode – + Sensor electrode double platinum pin electrode • Anolyte: sulfur dioxide, imidazol, iodide, different solvent for different applications: Catholyte methonol or ethanol with chloroform, octanol, • etylenglycol Catholyte: Diaphragm Anolyte 2021/9/16 AA similar (or modified) solution 3

Coulometry • Based on the same reaction as volumetric Karl Fischer Titration: H 2 O + 2 I + SO 2 + RN + ROHÙ (RNH)SO 4 R + 2(RNH)I - Iodine reacts with water 1: 1 - The solvent methanol is involved in the reaction. - A suitable base keeps the p. H 5 - 7 • But iodine will be produced just in time from iodide: 2 I Anodic Oxidation I 2 + 2 e- 2021/9/16 AA 4

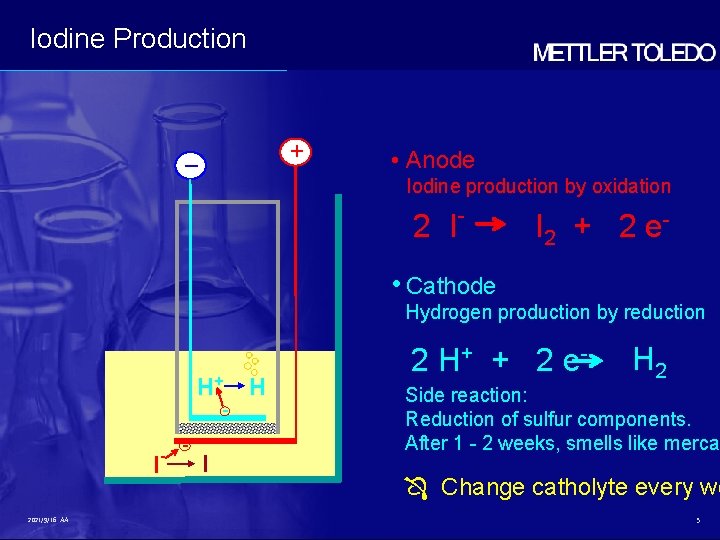
Iodine Production + – • Anode Iodine production by oxidation 2 I - I 2 + 2 e - • Cathode Hydrogen production by reduction H+ - I 2021/9/16 AA - - I H 2 H+ + 2 e - H 2 Side reaction: Reduction of sulfur components. After 1 - 2 weeks, smells like mercap Change catholyte every we 5

Coulometry Theory Definition One Coulomb C is the quantity of charge transported by an electric current of one Ampere (A) during one second (s). 1 C = 1 A • 1 s To produce one mol of a chemical compound, using one electron, 96484 C are required. 2021/9/16 AA Charles Augustin de Coulomb 14. 6. 1736 - 23. 8. 1806 6

Coulometry Theory Definition One Coulomb C is the quantity of charge transported by an electric current of one Ampere (A) during one second (s). 1 C = 1 A • 1 s To produce one mol of a chemical compound, using one electron, 96484 C are required. Two iodide ions react to iodine, which in turn reacts with water: Therefore water (18 2 I– 1 I 2 mol with H 2 O g) is equivalent to 2 x 96484 C or 10. 72 C / mg water. Absolute method, no standardization! 2021/9/16 AA 7

Iodine Production Speed – + H+ H I - - + I Normal conductivity Very low conductivity 2021/9/16 AA The iodine production speed • surface on: of the electrode depends • voltage at the generator electrode • the conductivity of the electrolyte Influence to conductivity: Samples and additional solvent as chloroform, etc. Warning: Low conductivity high current = 400 m. A 2100 µ low current = 200 m. A 1050 µg 8

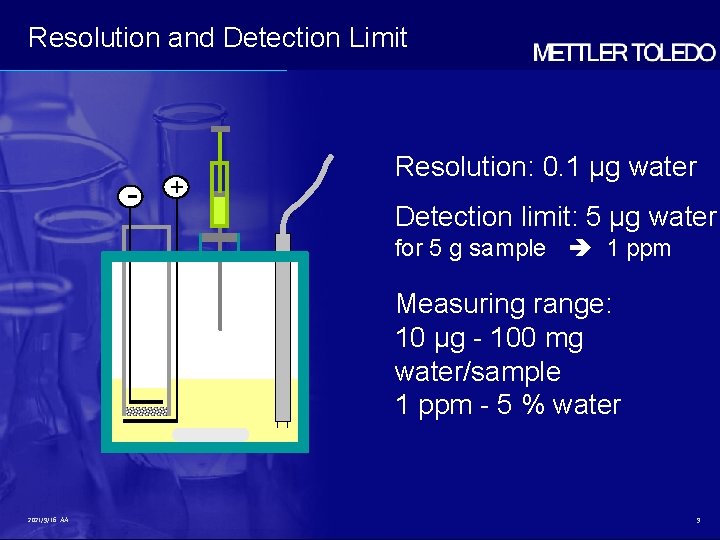
Resolution and Detection Limit - + Resolution: 0. 1 µg water Detection limit: 5 µg water for 5 g sample 1 ppm Measuring range: 10 µg - 100 mg water/sample 1 ppm - 5 % water 2021/9/16 AA 9

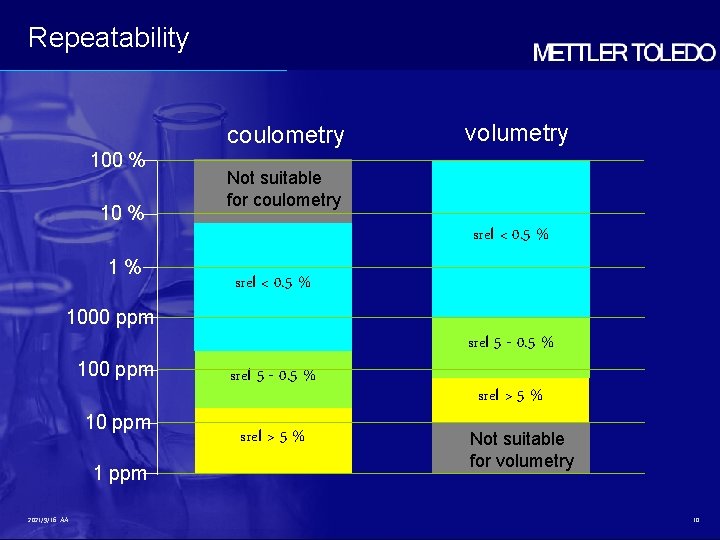
Repeatability coulometry 100 % 10 % volumetry Not suitable for coulometry srel < 0. 5 % 1% srel < 0. 5 % 1000 ppm 10 ppm 1 ppm 2021/9/16 AA srel 5 - 0. 5 % srel > 5 % Not suitable for volumetry 10

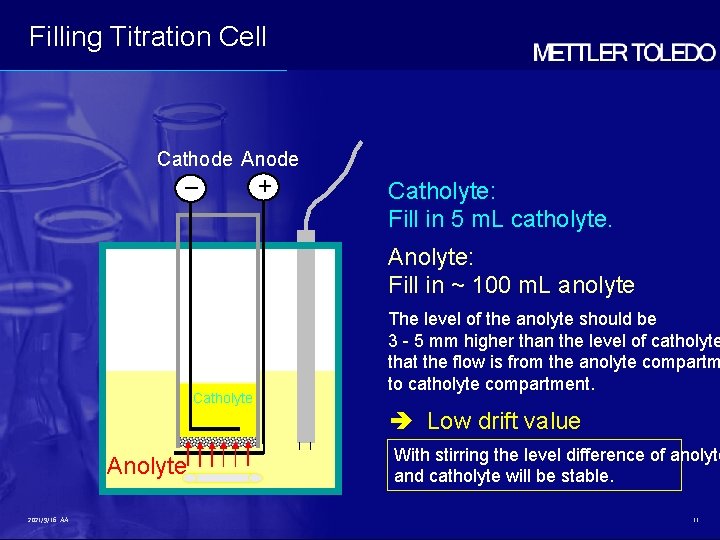
Filling Titration Cell Cathode Anode – + Catholyte: Fill in 5 m. L catholyte. Anolyte: Fill in ~ 100 m. L anolyte Catholyte The level of the anolyte should be 3 - 5 mm higher than the level of catholyte that the flow is from the anolyte compartm to catholyte compartment. Low drift value Anolyte 2021/9/16 AA With stirring the level difference of anolyte and catholyte will be stable. 11

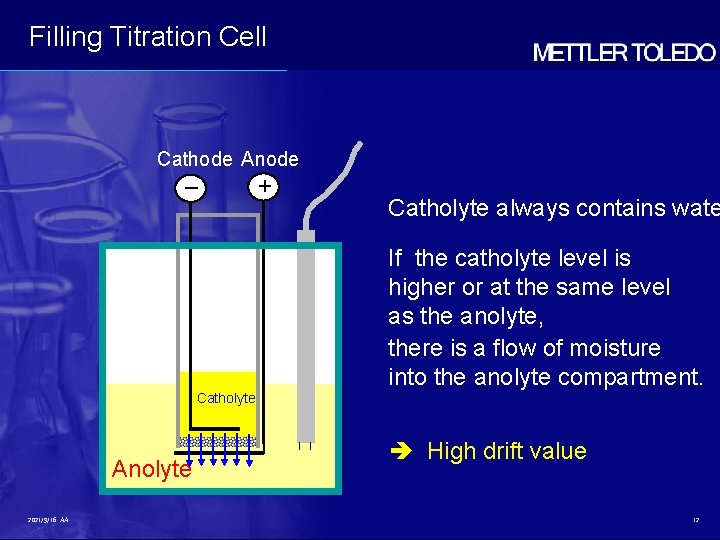
Filling Titration Cell Cathode Anode – + Catholyte always contains wate If the catholyte level is higher or at the same level as the anolyte, there is a flow of moisture into the anolyte compartment. Catholyte Anolyte 2021/9/16 AA High drift value 12

With or Without Diaphragm – + What are the differences? 2021/9/16 AA 13

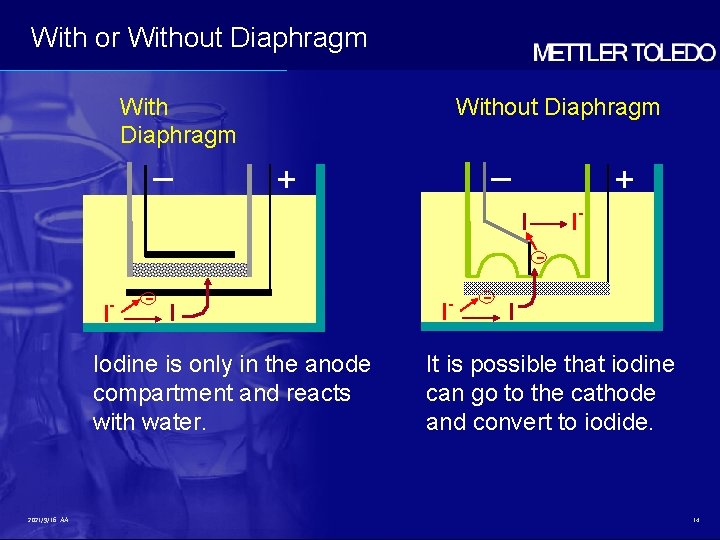
With or Without Diaphragm With Diaphragm – Without Diaphragm – + + I- I I - - I Iodine is only in the anode compartment and reacts with water. 2021/9/16 AA I - - I It is possible that iodine can go to the cathode and convert to iodide. 14

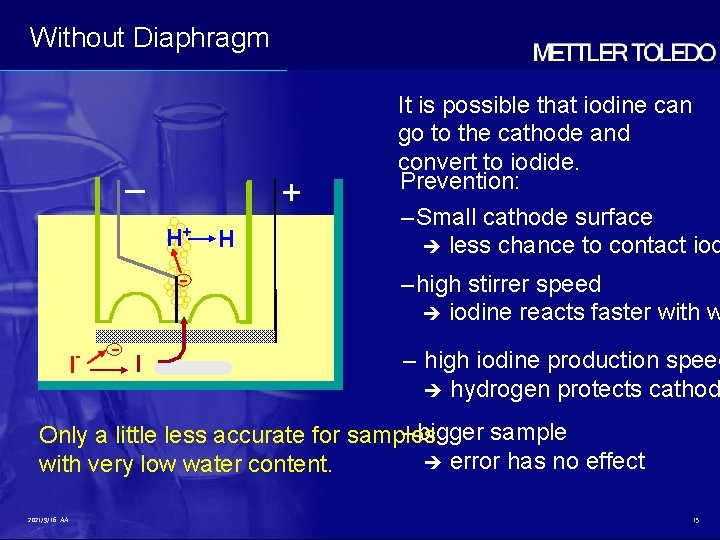
Without Diaphragm – + H+ - I - - I H It is possible that iodine can go to the cathode and convert to iodide. Prevention: – Small cathode surface less chance to contact iod – high stirrer speed iodine reacts faster with w – high iodine production speed hydrogen protects cathod – bigger sample Only a little less accurate for samples error has no effect with very low water content. 2021/9/16 AA 15

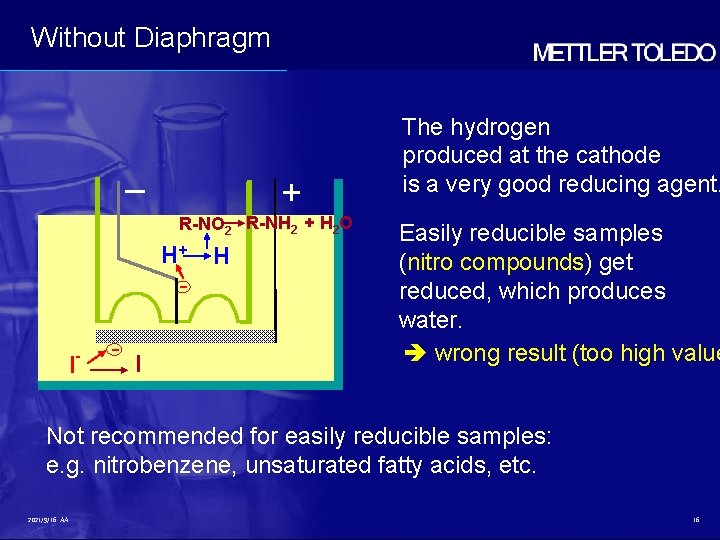
Without Diaphragm – + R-NO 2 R-NH 2 + H 2 O H+ I - - I H The hydrogen produced at the cathode is a very good reducing agent. Easily reducible samples (nitro compounds) get reduced, which produces water. wrong result (too high value Not recommended for easily reducible samples: e. g. nitrobenzene, unsaturated fatty acids, etc. 2021/9/16 AA 16

Without Diaphragm + + Titration cell easier to clean. Long-term drift value more stable. Only one reagent. Automation of emptying and refilling electrolyte. – A little bit less accuracy for very small water content (< 50 µg/sample) – Not recommended: • for easily reducible samples: nitro compounds, unsaturated fatty acids, etc. 2021/9/16 AA 17

Application With and Without Diaphragm With out diaphragm: a little bit less accuracy for very small water content (< 50 µg/sample) Examples: Transformer oil Mean n diaphragm srel 16. 3 ppm 6 19. 6 ppm 6 1. 5 % 5. 7 % 2021/9/16 AA µg water /sample with or without 34 - 40 39 - 43 with diaphragm without diaphragm 18

Without Diaphragm Titration cell without diaphragm is ideal for: • • • Hydrocarbons Halogenated hydrocarbons – + Alcohols Esters Ethers Acetamides Mineral oils Edible oils For this applications the titration Ethereal oils cell without diaphragm is recommended. 2021/9/16 AA 19

Analyte - + For a complete water determination the sample must be completely dissolved in the anolyte. Sample not dissolved, emulsion: Too low result Anolyte Different anolyte for different applications 2021/9/16 AA 20

Analyte for samples easy to dissolve - + alcohols, ethers, esters, hydrocarbons, halogenated hydrocarbons, nitro components, etc. For cell with diaphragm with methanol (HYDRANAL Coulomat AG) (apura - combi. Coulomat frit) Anolyte with ethanol (HYDARANAL Coulomat E) For cell without diaphragm with methanol (HYDRANAL Coulomat AD) (apura - combi. Coulomat fritless) 2021/9/16 AA 21

Analyte for samples not easy to dissolve edible oils, ethereal oils, ointments, etc. with methanol and octanol - + For cell with diaphragm (HYDRANAL Coulomat AG-H) (with 20 % hexanol) (apura - combi. Coulomat fritless) Anolyte add up to 40 % octanol or For cell without diaphragm decanol (HYDRANAL Coulomat AD) (apura - combi. Coulomat fritless) add up to 20 % octanol or decanol 2021/9/16 AA 22

Conductivity influences • Generation of iodine • Indication of the endpoint Conductivity of electrolyte decreases during determination ð long chained alcohols (hexanol, octanol, decanol), xylene or chloroform can be added. without diaphragm with diaphragm addition of max. 20 % to addition of max. 40 % Combi. Coulomat fritless to Combi. Coulomat frit high current at generator electrode higher robustness limit 5 - 6 m. S/cm before current breaks down 2021/9/16 AA 23

Analyte for samples difficult to dissolve mineral oils, transformer oil, silicon oils, etc with methanol and chloroform - + For cell with diaphragm (HYDRANAL Coulomat A) (with 20 % chloroform) (HYDRANAL Coulomat AG) (without chloroform) (apura - combi. Coulomat frit) (without chloroform) Anolyte add chloroform (maximum 50 %) For cell without diaphragm (HYDRANAL Coulomat AD) (apura - combi. Coulomat fritless) add up to 30 % chloroform 2021/9/16 AA 24

Analyte for Ketons and Aldehydes - + ketones and aldehydes react with methanol ketal and acetal formation + 1 H 2 O special reagent for ketones For cell with diaphragm Anolyte (HYDRANAL Coulomat AK and CG-K) with a long chain alcohol instead of methanol For cell without diaphragm (HYDRANAL Coulomat AK) Caution with aldehydes! Short chain aldehydes (for example acetaldehyde) will be oxidized at the anode. + 1 H 2 O Long chain aldehydes (for example benzaldehyde) are n 2021/9/16 AA 25