Redox Titrations Introduction 1 Redox Titration Based on
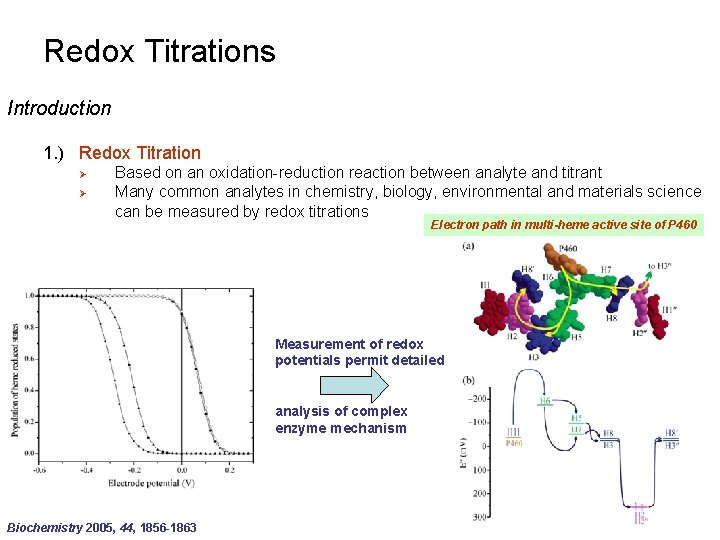
Redox Titrations Introduction 1. ) Redox Titration Ø Ø Based on an oxidation-reduction reaction between analyte and titrant Many common analytes in chemistry, biology, environmental and materials science can be measured by redox titrations Electron path in multi-heme active site of P 460 Measurement of redox potentials permit detailed analysis of complex enzyme mechanism Biochemistry 2005, 44, 1856 -1863

Redox Titrations Shape of a Redox Titration Curve 1. ) Voltage Change as a Function of Added Titrant Ø Consider the Titration Reaction (essentially goes to completion): K ≈ 1016 Ø Ø Ø Ce 4+ is added with a buret to a solution of Fe 2+ Pt electrode responds to relative concentration of Fe 3+/Fe 2+ & Ce 4+/Ce 3+ Calomel electrode used as reference Indicator half-reactions at Pt electrode: Eo = 0. 767 V Eo = 1. 70 V
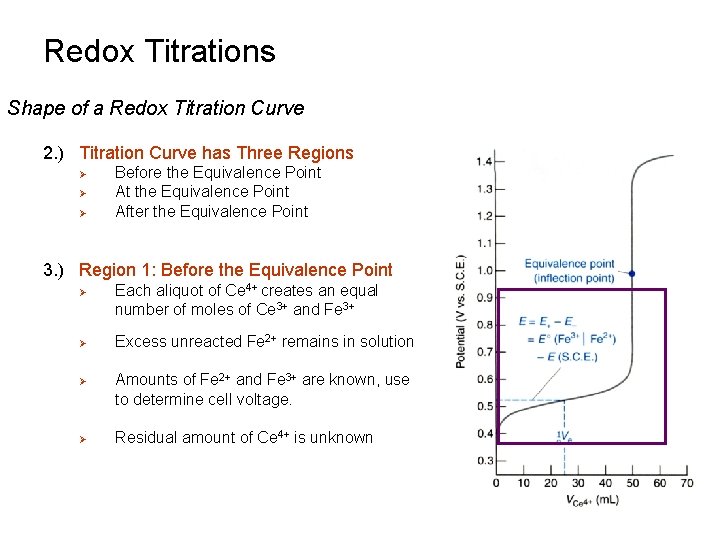
Redox Titrations Shape of a Redox Titration Curve 2. ) Titration Curve has Three Regions Ø Ø Ø Before the Equivalence Point At the Equivalence Point After the Equivalence Point 3. ) Region 1: Before the Equivalence Point Ø Ø Each aliquot of Ce 4+ creates an equal number of moles of Ce 3+ and Fe 3+ Excess unreacted Fe 2+ remains in solution Amounts of Fe 2+ and Fe 3+ are known, use to determine cell voltage. Residual amount of Ce 4+ is unknown
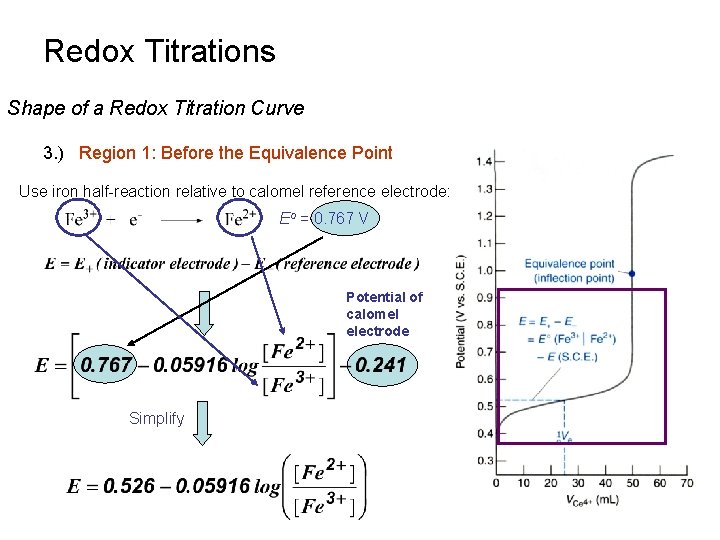
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Use iron half-reaction relative to calomel reference electrode: Eo = 0. 767 V Potential of calomel electrode Simplify
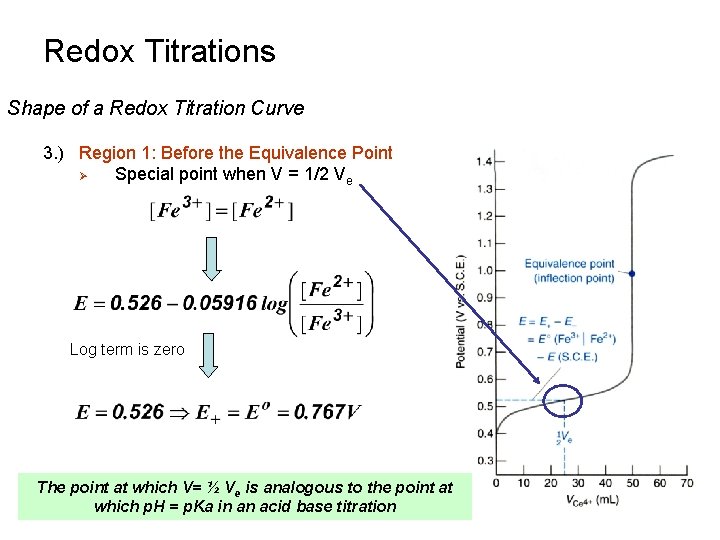
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Ø Special point when V = 1/2 Ve Log term is zero The point at which V= ½ Ve is analogous to the point at which p. H = p. Ka in an acid base titration
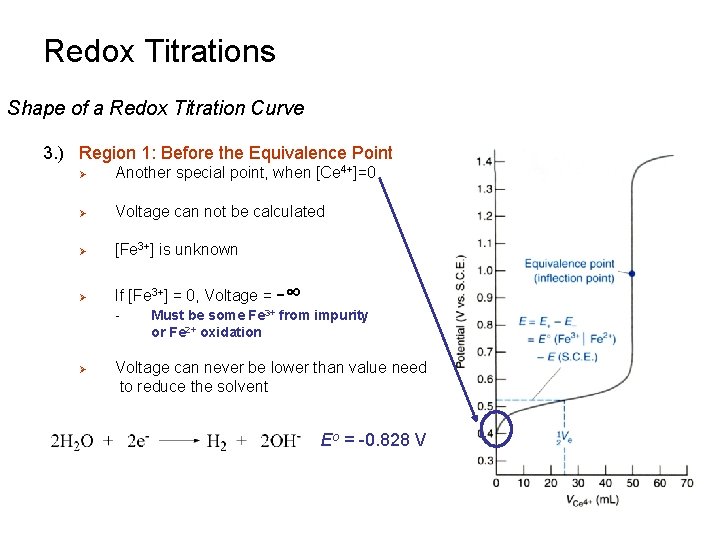
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Ø Another special point, when [Ce 4+]=0 Ø Voltage can not be calculated Ø [Fe 3+] is unknown Ø If [Fe 3+] = 0, Voltage = -∞ - Ø Must be some Fe 3+ from impurity or Fe 2+ oxidation Voltage can never be lower than value need to reduce the solvent Eo = -0. 828 V
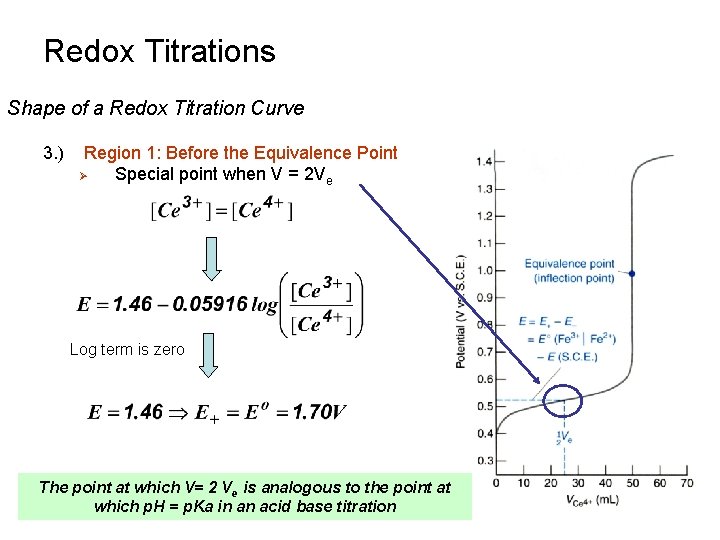
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Ø Special point when V = 2 Ve Log term is zero The point at which V= 2 Ve is analogous to the point at which p. H = p. Ka in an acid base titration
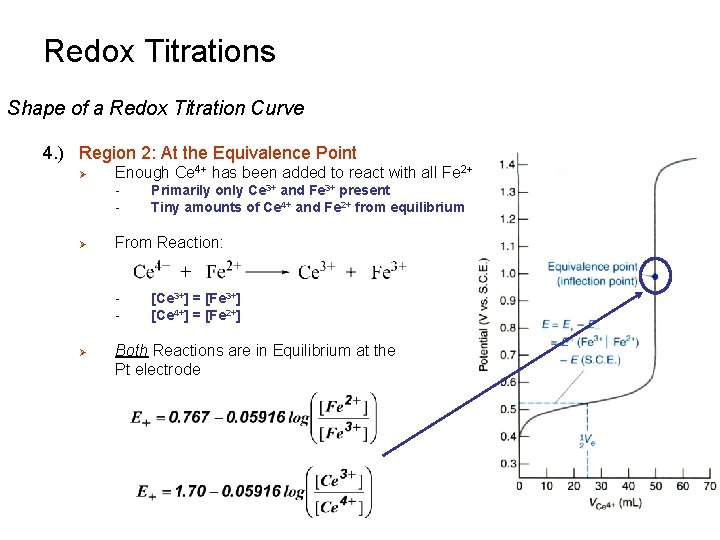
Redox Titrations Shape of a Redox Titration Curve 4. ) Region 2: At the Equivalence Point Ø Enough Ce 4+ has been added to react with all Fe 2+ - Ø From Reaction: - Ø Primarily only Ce 3+ and Fe 3+ present Tiny amounts of Ce 4+ and Fe 2+ from equilibrium [Ce 3+] = [Fe 3+] [Ce 4+] = [Fe 2+] Both Reactions are in Equilibrium at the Pt electrode

Redox Titrations Shape of a Redox Titration Curve 4. ) Region 2: At the Equivalence Point Ø Ø Ø Don’t Know the Concentration of either Fe 2+ or Ce 4+ Can’t solve either equation independently to determine E+ Instead Add both equations together Add Rearrange
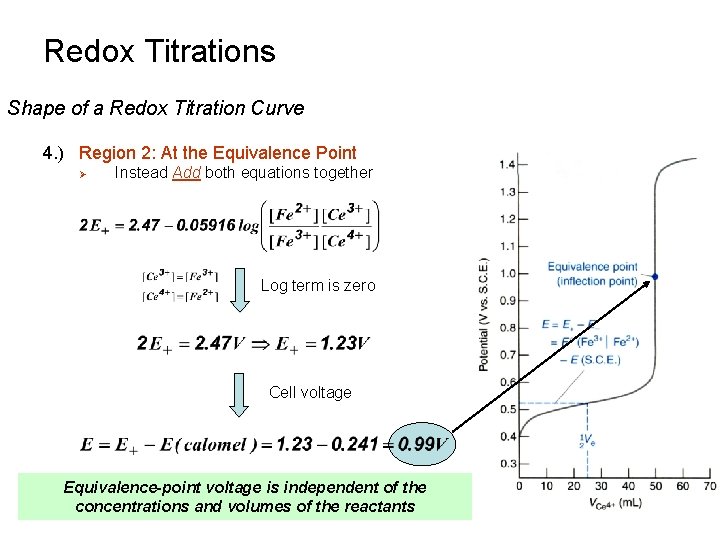
Redox Titrations Shape of a Redox Titration Curve 4. ) Region 2: At the Equivalence Point Ø Instead Add both equations together Log term is zero Cell voltage Equivalence-point voltage is independent of the concentrations and volumes of the reactants
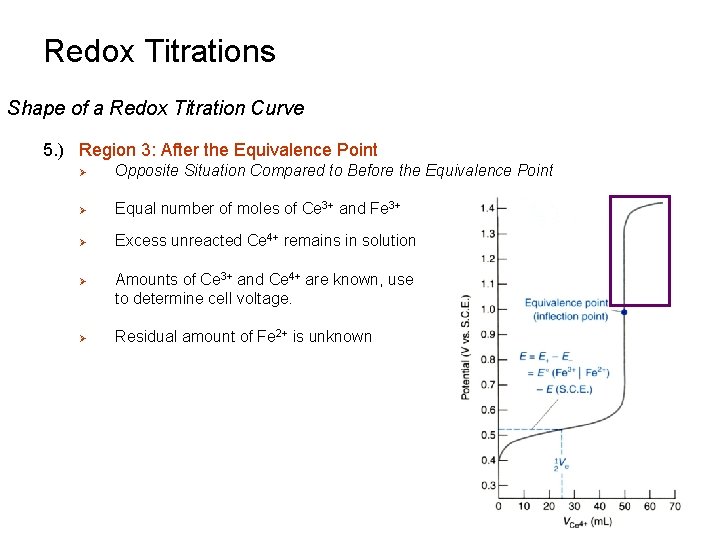
Redox Titrations Shape of a Redox Titration Curve 5. ) Region 3: After the Equivalence Point Ø Opposite Situation Compared to Before the Equivalence Point Ø Equal number of moles of Ce 3+ and Fe 3+ Ø Excess unreacted Ce 4+ remains in solution Ø Ø Amounts of Ce 3+ and Ce 4+ are known, use to determine cell voltage. Residual amount of Fe 2+ is unknown
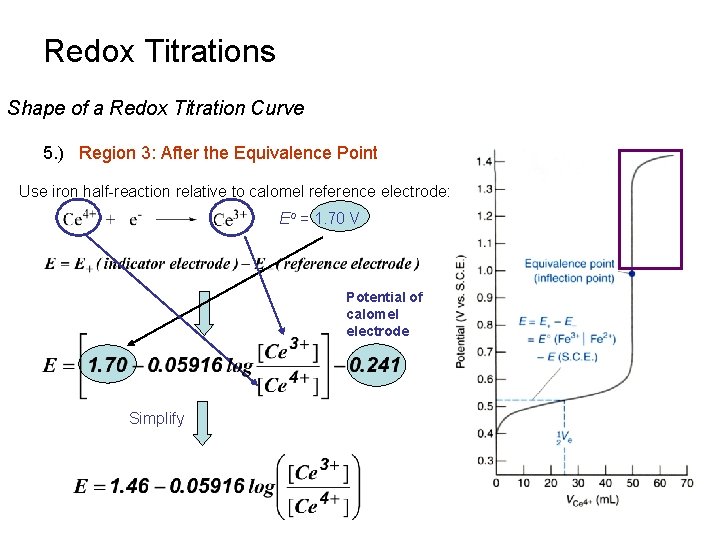
Redox Titrations Shape of a Redox Titration Curve 5. ) Region 3: After the Equivalence Point Use iron half-reaction relative to calomel reference electrode: Eo = 1. 70 V Potential of calomel electrode Simplify
Redox Titrations Shape of a Redox Titration Curve 6. ) Titration Only Depends on the Ratio of Reactants Ø Independent on concentration and/or volume Ø Same curve if diluted or concentrated by a factor of 10
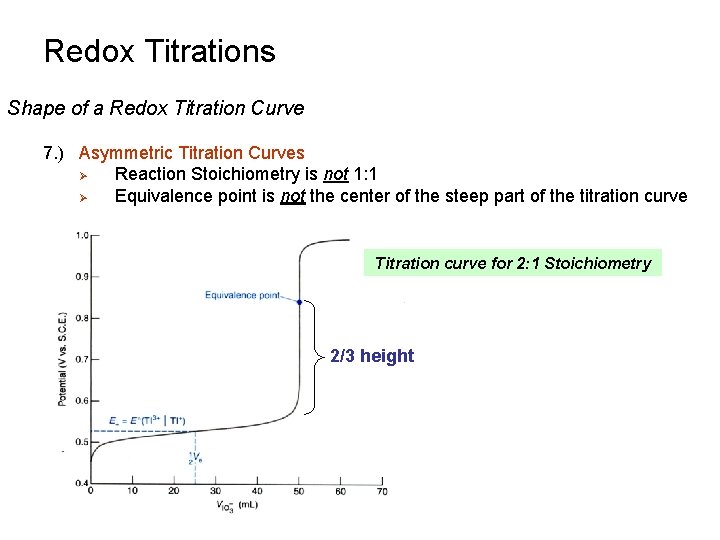
Redox Titrations Shape of a Redox Titration Curve 7. ) Asymmetric Titration Curves Ø Reaction Stoichiometry is not 1: 1 Ø Equivalence point is not the center of the steep part of the titration curve Titration curve for 2: 1 Stoichiometry 2/3 height
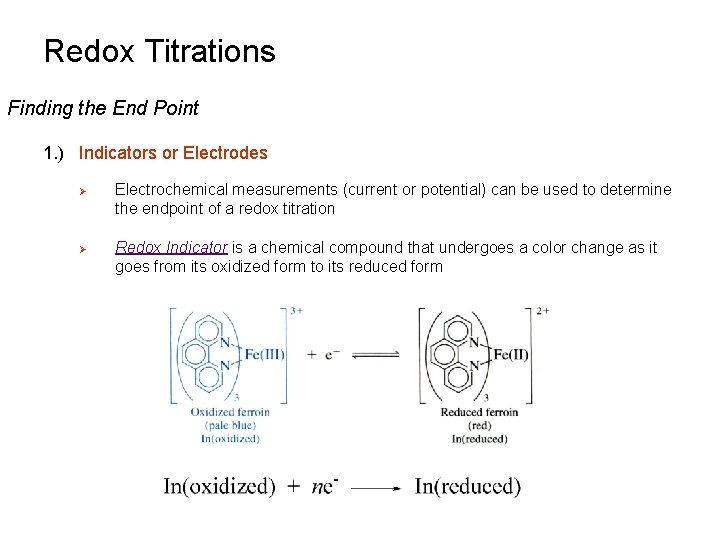
Redox Titrations Finding the End Point 1. ) Indicators or Electrodes Ø Ø Electrochemical measurements (current or potential) can be used to determine the endpoint of a redox titration Redox Indicator is a chemical compound that undergoes a color change as it goes from its oxidized form to its reduced form

Redox Titrations Finding the End Point 2. ) Redox Indicators Ø Color Change for a Redox Indicator occurs mostly over the range: where Eo is the standard reduction potential for the indicator and n is the number of electrons involved in the reduction For Ferroin with Eo = 1. 147 V, the range of color change relative to SHE: Relative to SCE is:
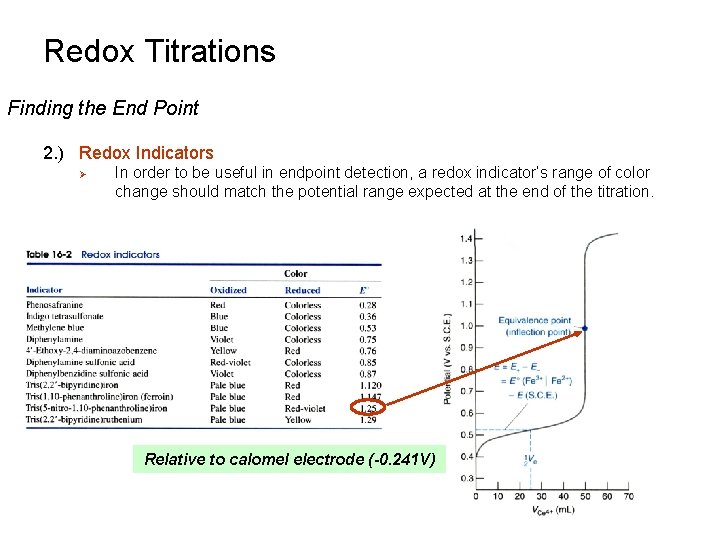
Redox Titrations Finding the End Point 2. ) Redox Indicators Ø In order to be useful in endpoint detection, a redox indicator’s range of color change should match the potential range expected at the end of the titration. Relative to calomel electrode (-0. 241 V)

Redox Titrations Common Redox Reagents 1. ) Adjustment of Analyte Oxidation State Ø Before many compounds can be determined by Redox Titrations, must be converted into a known oxidation state - Ø This step in the procedure is known as prereduction or preoxidation Reagents for prereduction or preoxidation must: - Ø Totally convert analyte into desired form Be easy to remove from the reaction mixture Avoid interfering in the titration Potassium Permanganate (KMn. O 4) - Strong oxidant Own indicator Titration of VO 2+ with KMn. O 4 p. H ≤ 1 Eo = 1. 507 V Violet colorless p. H neutral or alkaline Eo = 1. 692 V Violet brown p. H strolngly alkaline Eo = 0. 56 V Violet green Before Near After Equivalence point

Redox Titrations Common Redox Reagents 2. ) Example A 50. 00 m. L sample containing La 3+ was titrated with sodium oxalate to precipitate La 2(C 2 O 4)3, which washed, dissolved in acid, and titrated with 18. 0 m. L of 0. 006363 M KMn. O 4. Calculate the molarity of La 3+ in the unknown.
- Slides: 19