Understanding back titrations In an ordinary titration we

Understanding back titrations

In an ordinary titration we react a known volume of a standard solution (one whose concentration is known) with a known volume or mass of a solution/substance whose concentration is unknown. Usually the unknown solution is in the conical flask and we add standard solution from a burette until a colour change indicates that all the unknown has reacted. For example, 25. 0 m. L of HCl solution (in the conical flask) is reacted with 1. 00 mol L– 1 Na. OH solution (in the burette). Phenolphthalein indicator changes from colourless to pink after the addition of 20. 0 m. L of Na. OH. The concentration of the HCl is therefore 0. 800 mol L– 1.

Many reactions are not suitable for analysis by a standard titration, perhaps because the reaction is very slow, or because other reaction products form which will get in the way of an exact result. Chemists often avoid these problems by doing a back titration. Calcium carbonate is a common ingredient in indigestion tablets. It reacts with acid, but the reaction with solid Ca. CO 3 takes time, and the CO 2 produced is acidic. These factors make a direct titration difficult. Instead, we react the crushed tablets with a known volume of excess of standard HCl, heat the mixture to remove all CO 2, and then titrate in standard Na. OH until the rest of the acid has reacted.

In this method the Ca. CO 3 in the tablet reacts with an excess of HCl. The unreacted HCl reacts with Na. OH from the burette.

To calculate the mass of calcium carbonate in the tablet: • Calculate the amount of HCl added to the flask. • Calculate the amount of Na. OH used in the titres. • Use n(Na. OH) to calculate the amount of HCl reacting with the Na. OH. This is the HCl left over after the reaction with the tablet. • Calculate the amount of HCl which reacted with the tablet. • Calculate the amount of Ca. CO 3 in the tablet, and hence the mass of Ca. CO 3 in the tablet.

Data C(HCl) = 1. 073 mol L-1 c(Na. OH) = 0. 484 mol L-1 V(HCl) = 20. 0 m. L V(Na. OH) = 18. 43 m. L Ca. CO 3 + 2 HCl → Ca. Cl 2 + CO 2 + H 2 O HCl + Na. OH → Na. Cl + H 2 O 1 Calculate the amount of HCl added to the flask.

2 Calculate the amount of Na. OH used in the titration. 3 Use n(Na. OH) to calculate the amount of HCl reacting with the Na. OH. This is the HCl left over after the reaction with the tablet. HCl + Na. OH → Na. Cl + H 2 O

4 Calculate the amount of HCl which reacted with the tablet.

5 Calculate the amount of Ca. CO 3 in the tablet, and hence the mass of Ca. CO 3 in the tablet. Ca. CO 3 + 2 HCl → Ca. Cl 2 + CO 2 + H 2 O
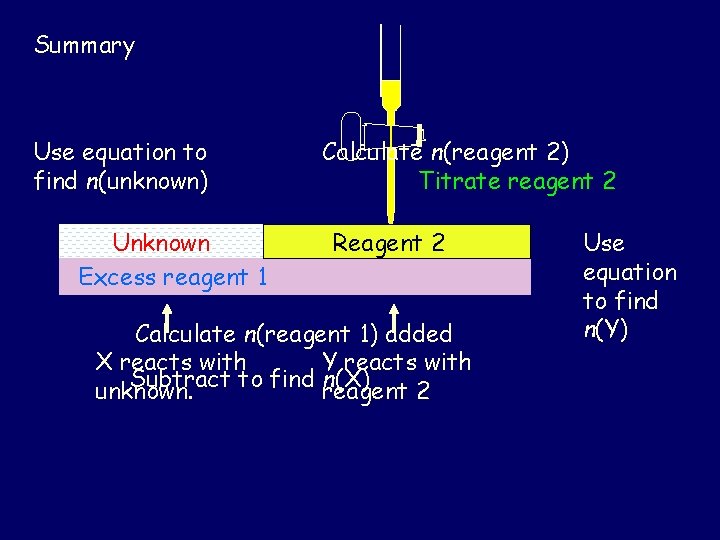
Summary Use equation to find n(unknown) Unknown Excess reagent 1 Calculate n(reagent 2) Titrate reagent 2 Reagent 2 Calculate n(reagent 1) added X reacts with Y reacts with Subtract to find reagent n(X) unknown. 2 Use equation to find n(Y)
- Slides: 10