June 10 th 2006 ETH Zurich Switzerland Organic










- Slides: 21

June 10 th, 2006 ETH Zurich, Switzerland Organic Molecules on Insulating Surfaces Investigated by NC-AFM Enrico Gnecco NCCR Nanoscale Science University of Basel, Switzerland

Motivations I electrodes metallic substrate molecule

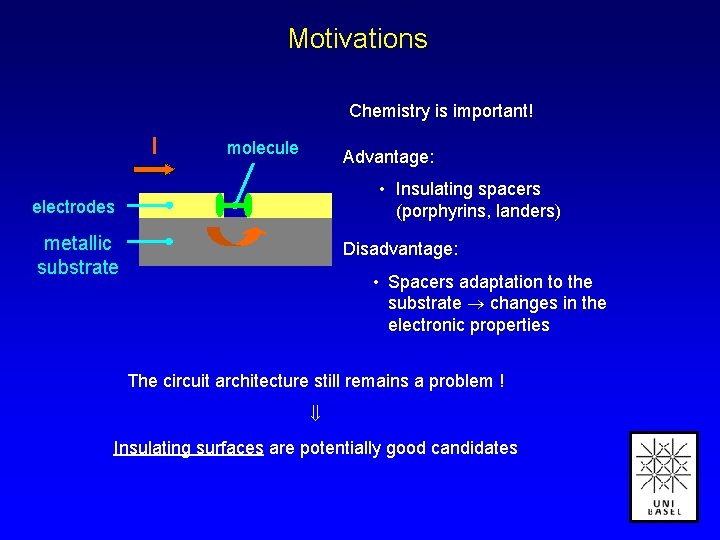
Motivations Chemistry is important! I molecule Advantage: • Insulating spacers (porphyrins, landers) electrodes metallic substrate Disadvantage: • Spacers adaptation to the substrate changes in the electronic properties The circuit architecture still remains a problem ! Insulating surfaces are potentially good candidates

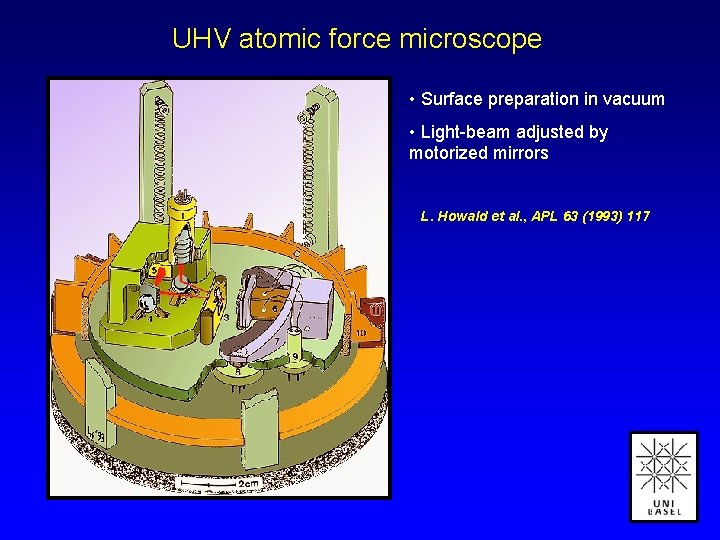
UHV atomic force microscope • Surface preparation in vacuum • Light-beam adjusted by motorized mirrors L. Howald et al. , APL 63 (1993) 117

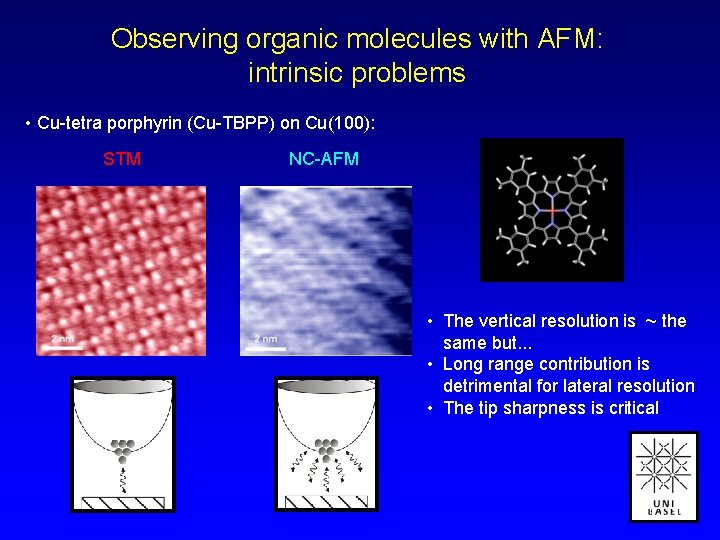
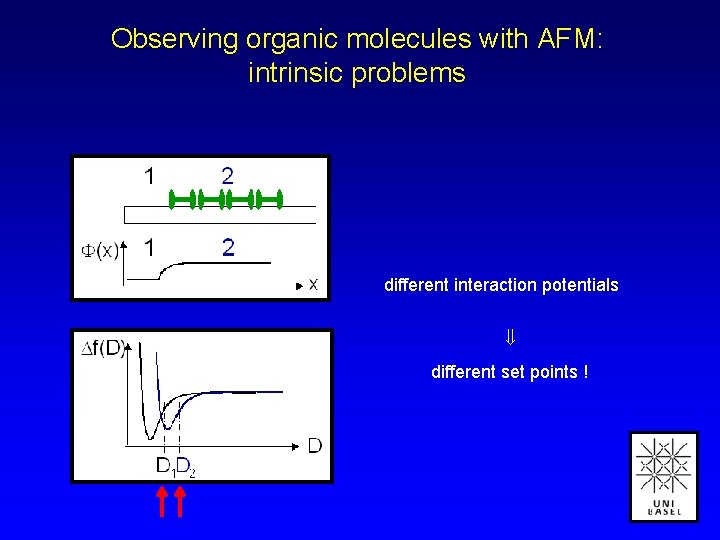
Observing organic molecules with AFM: intrinsic problems • Cu-tetra porphyrin (Cu-TBPP) on Cu(100): STM NC-AFM • The vertical resolution is ~ the same but. . . • Long range contribution is detrimental for lateral resolution • The tip sharpness is critical

Observing organic molecules with AFM: intrinsic problems different interaction potentials different set points !

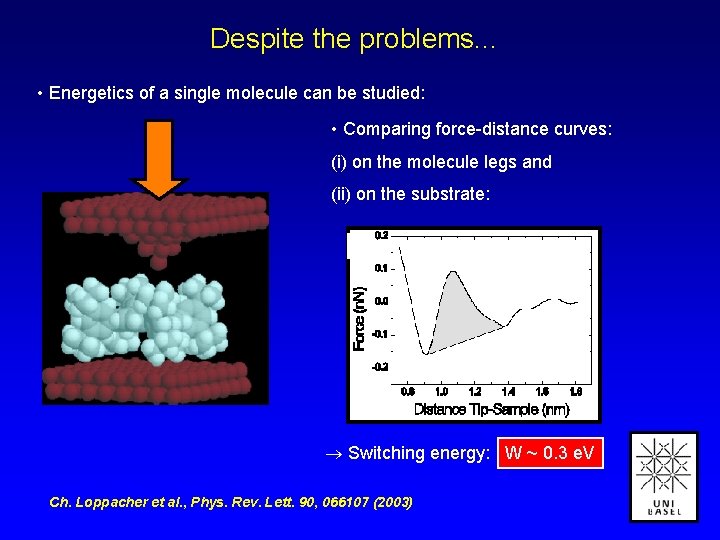
Despite the problems. . . • Energetics of a single molecule can be studied: • Comparing force-distance curves: (i) on the molecule legs and (ii) on the substrate: Switching energy: W ~ 0. 3 e. V Ch. Loppacher et al. , Phys. Rev. Lett. 90, 066107 (2003)

Switching to insulators. . . • “Atomic” resolution on KBr(100): a = 0. 66 nm b = 0. 47 nm • Stable nanopatterns can be created: 5 nm EG et al. , Phys. Rev. Lett. 88, 215501 (2002) 50 nm

Trapping the molecules. . . • How to reduce the mobility of the molecules? • Heating at 380 °C Spiral pattern Step height: 0. 35 nm K. Yamamoto et al. , J. Cryst. Growth 94 (1989) 629

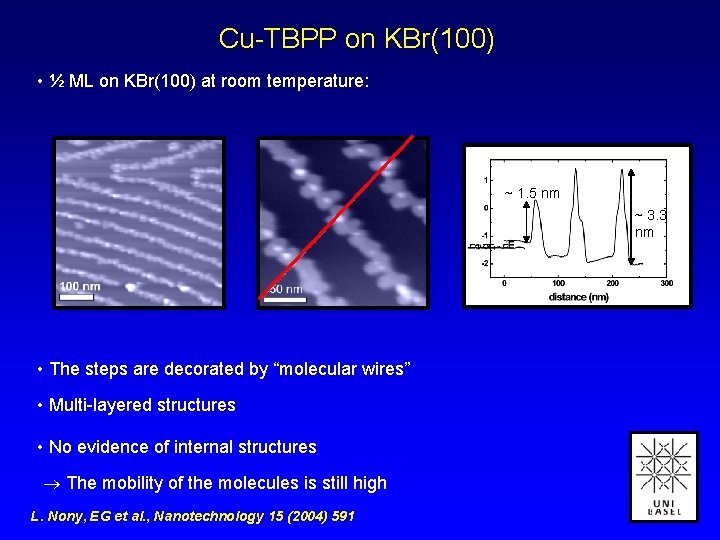
Cu-TBPP on KBr(100) • ½ ML on KBr(100) at room temperature: ~ 1. 5 nm ~ 3. 3 nm • The steps are decorated by “molecular wires” • Multi-layered structures • No evidence of internal structures The mobility of the molecules is still high L. Nony, EG et al. , Nanotechnology 15 (2004) 591

Lowering the mobility. . . • KBr(100) irradiated with 1 k. V e at 120 °C: • Rectangular holes (~10 nm wide) • Mono-layer depth (0. 33 nm) Holes as molecular traps? R. Bennewitz et al. , Surf. Sci. 474, L 197 (2001)

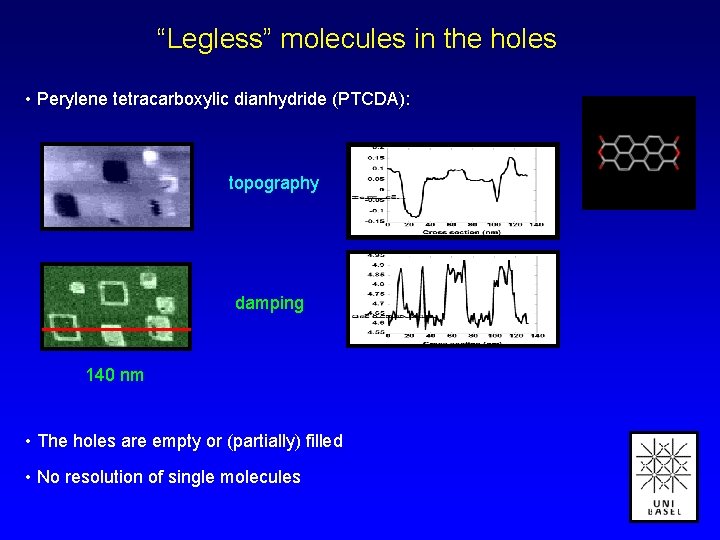
“Legless” molecules in the holes • Perylene tetracarboxylic dianhydride (PTCDA): topography damping 140 nm • The holes are empty or (partially) filled • No resolution of single molecules

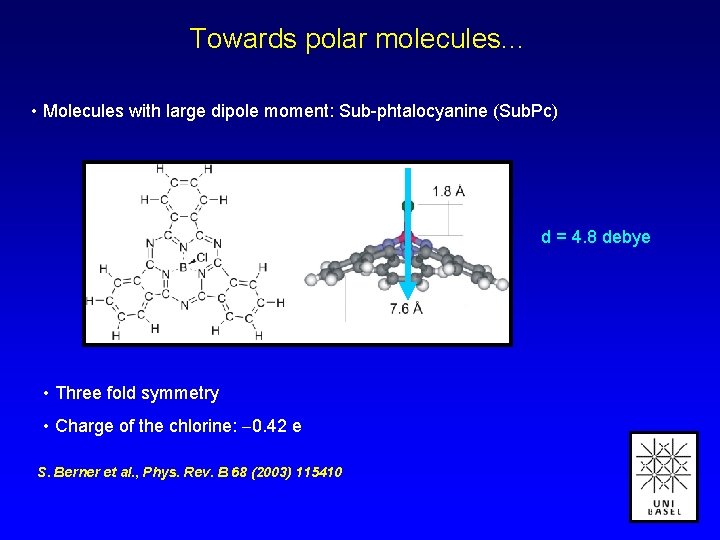
Towards polar molecules. . . • Molecules with large dipole moment: Sub-phtalocyanine (Sub. Pc) d = 4. 8 debye • Three fold symmetry • Charge of the chlorine: 0. 42 e S. Berner et al. , Phys. Rev. B 68 (2003) 115410

Sub. PC molecules on e -irradiated KBr • 1 ML on KBr(100) at 80 °C: 18 nm • Single molecules are resolved ! L. Nony, EG et al. , Nano Lett. 4 (2004) 2185

Molecular confinement • Some details: 1. 4 nm • The molecules are aligned in rows oriented 45° • Along some edges the molecules are mismatched • Height of the islands ~ 0. 6 nm (+ hole depth = 1 nm)

Matching the substrate. . . Potential arrangement of the molecules : • Apparent size ~1 nm • Alignment along the [110] axis • Regular rows: 3 b ~ 1. 4 nm • Distance between molecules in a row: 2 b ~ 0. 95 nm

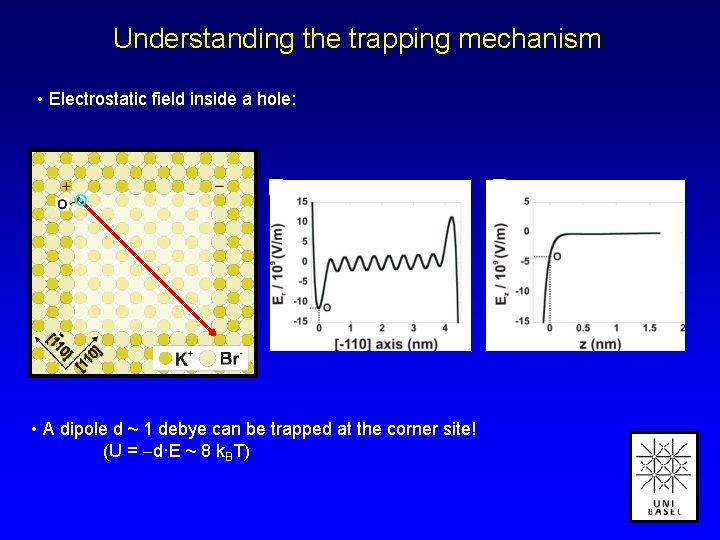
Understanding the trapping mechanism • Electrostatic field inside a hole: • A dipole d ~ 1 debye can be trapped at the corner site! (U = d·E ~ 8 k. BT)

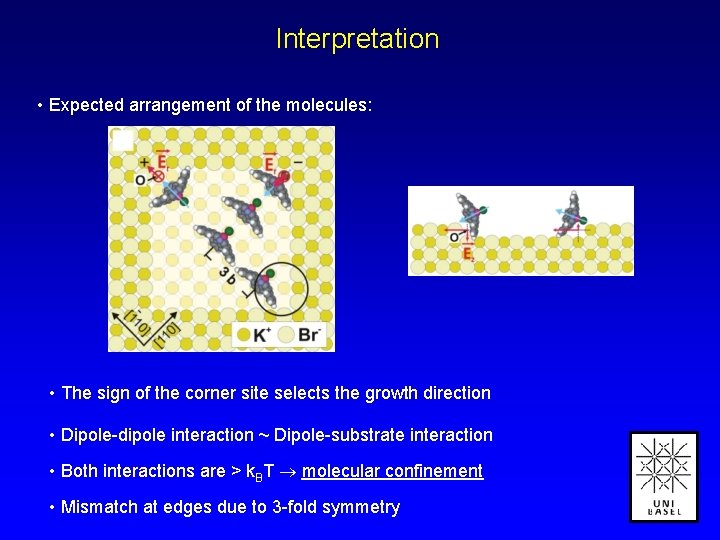
Interpretation • Expected arrangement of the molecules: • The sign of the corner site selects the growth direction • Dipole-dipole interaction ~ Dipole-substrate interaction • Both interactions are > k. BT molecular confinement • Mismatch at edges due to 3 -fold symmetry

Empty vs filled holes • On larger scale. . . 150 nm • Only the holes < 15 nm in size are filled !

Conclusions • Holes created by e irradiation on KBr act as molecular traps • Single organic molecules on insulators have been resolved by AFM • The size of the holes is critical Outlook • Molecules with 4 -fold symmetry • How to contact electrodes? • Theory of molecular confinement?

Acknowledgments UNI Basel University of Tokyo Ernst Meyer T. Eguchi Christoph Gerber Laurent Nony (*) CNRS Toulouse Alexis Baratoff A. Gourdon Roland Bennewitz (**) C. Joachim Oliver Pfeiffer Thomas Young (*) Now at Univ. Aix-Marseille III, France (**) Now at Mc. Gill Univ. , Montreal, Canada This work was supported by • The Swiss National Science Fundation • The Swiss National Center of Competence in Research on Nanoscale Science