Journal Club Ryuzo Kawamori Naoko Tajima Yasuhiko Iwamoto

Journal Club Ryuzo Kawamori, Naoko Tajima, Yasuhiko Iwamoto, Atsunori Kashiwagi, Kazuaki Shimamoto, Kohei Kaku, on behalf of the Voglibose Ph-3 Study Group Voglibose for prevention of type 2 diabetes mellitus: a randomised, double-blind trial in Japanese individuals with impaired glucose tolerance Published online April 22, 2009 DOI: 10. 1016/S 0140 -6736(09)60222 -1 Bakris GL, Toto RD, Mc. Cullough PA, Rocha R, Purkayastha D, Davis P; GUARD (Gauging Albuminuria Reduction With Lotrel in Diabetic Patients With Hypertension) Study Investigators. Effects of different ACE inhibitor combinations on albuminuria: results of the GUARD study. Kidney Int. 2008 Jun; 73(11): 1303 -9. 2009年 4月30日 8: 30 -8: 55 8階 医局 埼玉医科大学 総合医療センター 内分泌・糖尿病内科 Department of Endocrinology and Diabetes, Saitama Medical Center, Saitama Medical University 松田 昌文 Matsuda, Masafumi

Department of Medicine, Metabolism and Endocrinology, Juntendo University School of Medicine, Tokyo, Japan (Prof R Kawamori MD); Division of Diabetes, Metabolism and Endocrinology, Department of Internal Medicine, Jikei University School of Medicine, Tokyo, Japan (Prof N Tajima MD); Diabetes Centre, Department of Medicine, Tokyo Women’s Medical University, Tokyo, Japan (Prof Y Iwamoto MD); Department of Medicine, Shiga University of Medical Science, Shiga, Japan (Prof A Kashiwagi MD); Second Department of Internal Medicine, Sapporo Medical University, Sapporo, Japan (Prof K Shimamoto MD); and Diabetes and Endocrine Division, Department of Medicine, Kawasaki Medical School, Kurashiki, Japan (Prof K Kaku MD) www. thelancet. com Published online April 22, 2009 DOI: 10. 1016/S 0140 -6736(09)60222 -1

April 25, 2009 at Granvia Hotel Okayama

Background The increased prevalence of type 2 diabetes mellitus is a major concern for health providers. We therefore assessed whether voglibose, an α-glucosidase inhibitor, could prevent the development of type 2 diabetes in high-risk Japanese individuals with impaired glucose tolerance.
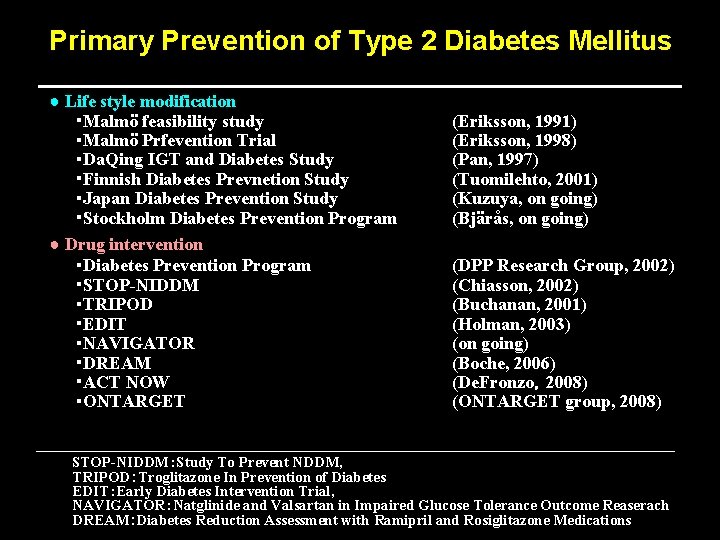
Primary Prevention of Type 2 Diabetes Mellitus ● Life style modification ・Malmö feasibility study ・Malmö Prfevention Trial ・Da. Qing IGT and Diabetes Study ・Finnish Diabetes Prevnetion Study ・Japan Diabetes Prevention Study ・Stockholm Diabetes Prevention Program ● Drug intervention ・Diabetes Prevention Program ・STOP-NIDDM ・TRIPOD ・EDIT ・NAVIGATOR ・DREAM ・ACT NOW ・ONTARGET (Eriksson, 1991) (Eriksson, 1998) (Pan, 1997) (Tuomilehto, 2001) (Kuzuya, on going) (Bjärås, on going) (DPP Research Group, 2002) (Chiasson, 2002) (Buchanan, 2001) (Holman, 2003) (on going) (Boche, 2006) (De. Fronzo,2008) (ONTARGET group, 2008) STOP-NIDDM:Study To Prevent NDDM, TRIPOD:Troglitazone In Prevention of Diabetes EDIT:Early Diabetes Intervention Trial, NAVIGATOR:Natglinide and Valsartan in Impaired Glucose Tolerance Outcome Reaserach DREAM:Diabetes Reduction Assessment with Ramipril and Rosiglitazone Medications
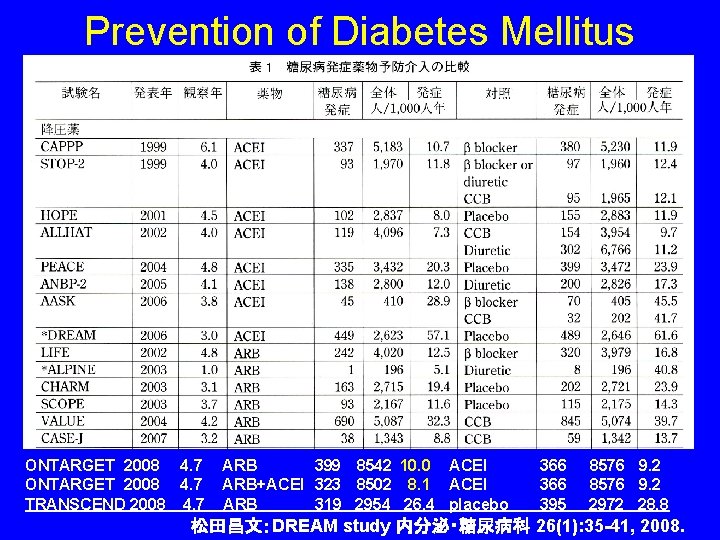
Prevention of Diabetes Mellitus ONTARGET 2008 4. 7 TRANSCEND 2008 4. 7 ARB 399 8542 10. 0 ACEI ARB+ACEI 323 8502 8. 1 ACEI ARB 319 2954 26. 4 placebo 366 395 8576 9. 2 2972 28. 8 松田昌文:DREAM study 内分泌・糖尿病科 26(1): 35 -41, 2008.
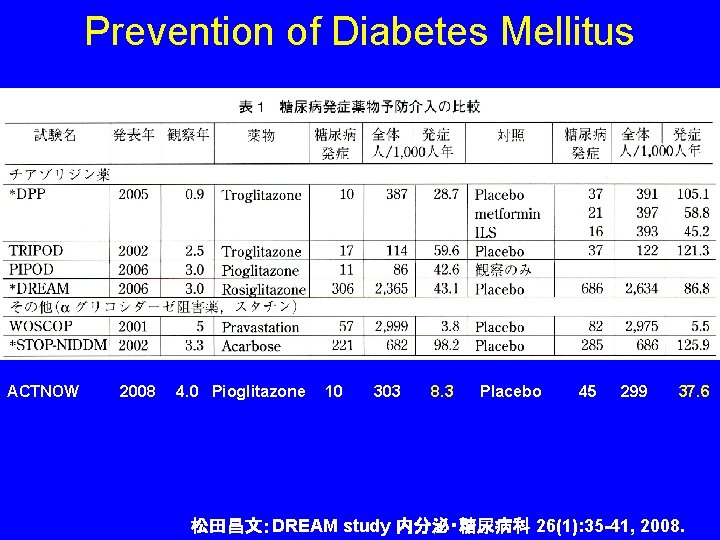
Prevention of Diabetes Mellitus ACTNOW 2008 4. 0 Pioglitazone 10 303 8. 3 Placebo 45 299 37. 6 松田昌文:DREAM study 内分泌・糖尿病科 26(1): 35 -41, 2008.




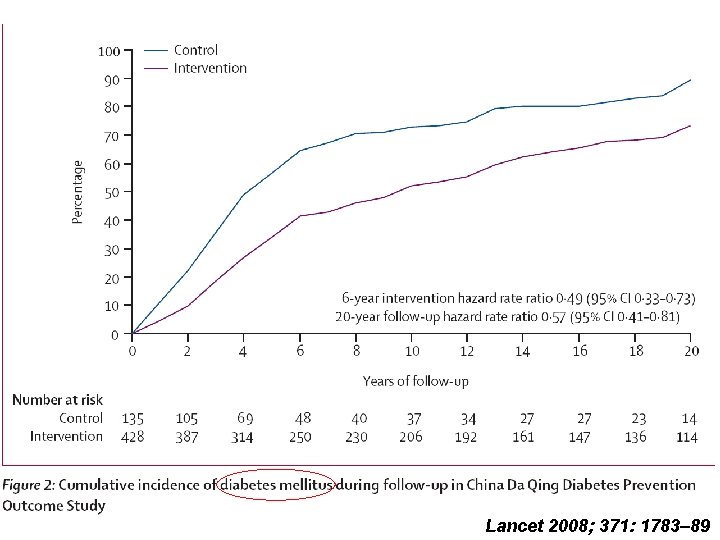
Lancet 2008; 371: 1783– 89





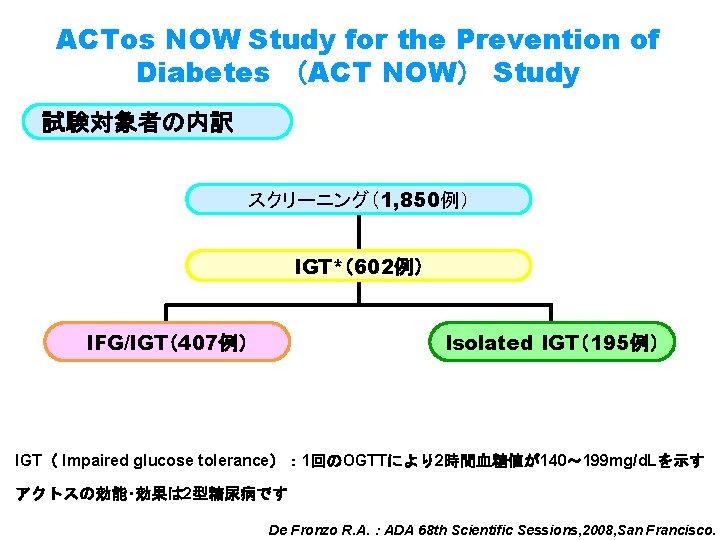
ACTos NOW Study for the Prevention of Diabetes (ACT NOW) Study 試験対象者の内訳 スクリーニング(1, 850例) IGT*(602例) IFG/IGT(407例) Isolated IGT(195例) IGT( Impaired glucose tolerance): 1回のOGTTにより2時間血糖値が140~ 199 mg/d. Lを示す アクトスの効能・効果は 2型糖尿病です De Fronzo R. A. :ADA 68 th Scientific Sessions, 2008, San Francisco.





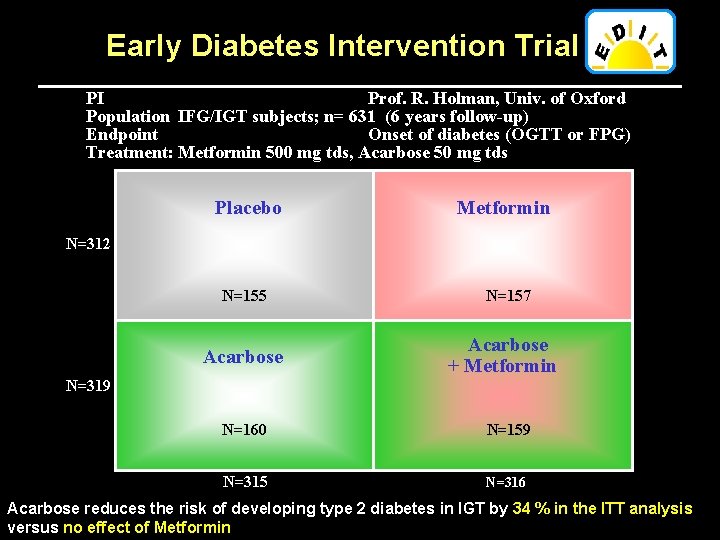
Early Diabetes Intervention Trial PI Prof. R. Holman, Univ. of Oxford Population IFG/IGT subjects; n= 631 (6 years follow-up) Endpoint Onset of diabetes (OGTT or FPG) Treatment: Metformin 500 mg tds, Acarbose 50 mg tds Placebo Metformin N=155 N=157 N=312 Acarbose + Metformin N=319 N=160 N=159 N=315 N=316 Acarbose reduces the risk of developing type 2 diabetes in IGT by 34 % in the ITT analysis versus no effect of Metformin


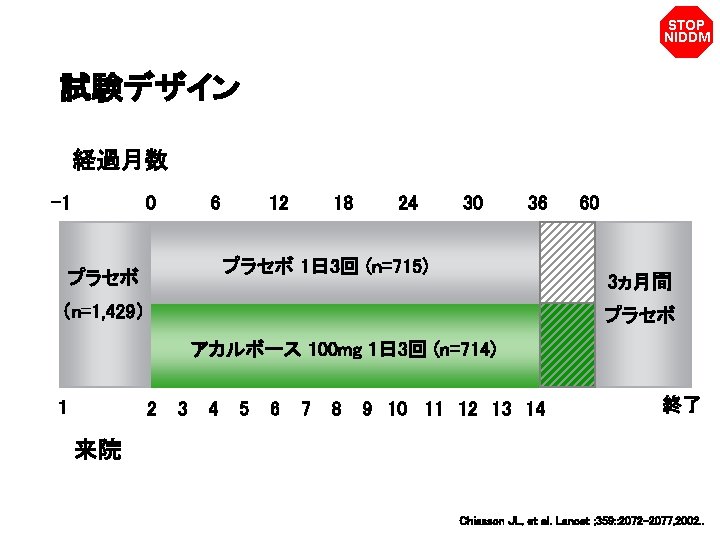
STOP NIDDM 試験デザイン 経過月数 0 6 12 18 24 30 36 プラセボ 1日 3回 (n=715) プラセボ (n=1, 429) アカルボース 100 mg 1日 3回 (n=714) 1 来院 2 3 4 5 6 7 8 9 10 11 12 13 14 60 Placebo – 1 3ヵ月間 プラセボ 終了 Chiasson JL, et al. Lancet ; 359: : 2072– 2077, 2002. .


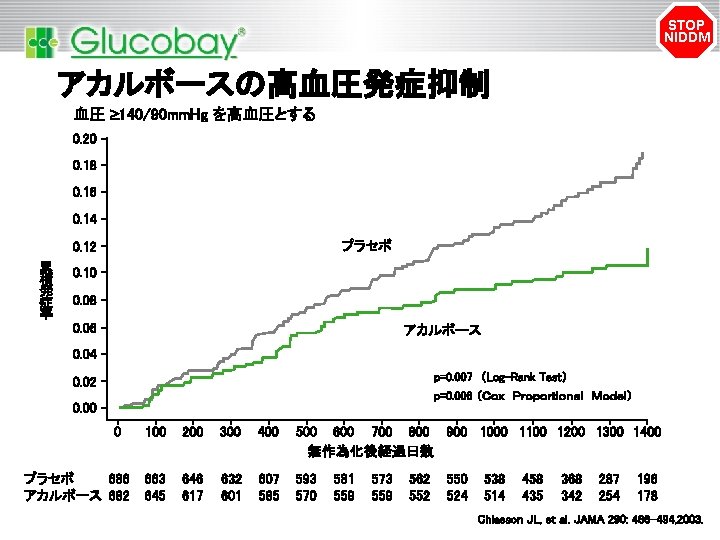
STOP NIDDM アカルボースの高血圧発症抑制 血圧 ³ 140/90 mm. Hg を高血圧とする 0. 20 0. 18 0. 16 0. 14 プラセボ 0. 12 累 積 発 症 率 0. 10 0. 08 0. 06 アカルボース 0. 04 p=0. 007 (Log-Rank Test) 0. 02 p=0. 006 (Cox Proportional Model) 0. 00 0 プラセボ 686 アカルボース 682 100 200 300 400 500 600 700 800 900 1000 1100 1200 1300 1400 無作為化後経過日数 663 645 646 617 632 601 607 585 593 570 581 559 573 559 562 550 524 538 514 458 435 368 342 287 254 196 178 Chiasson JL, et al. JAMA 290: 486– 494, 2003.
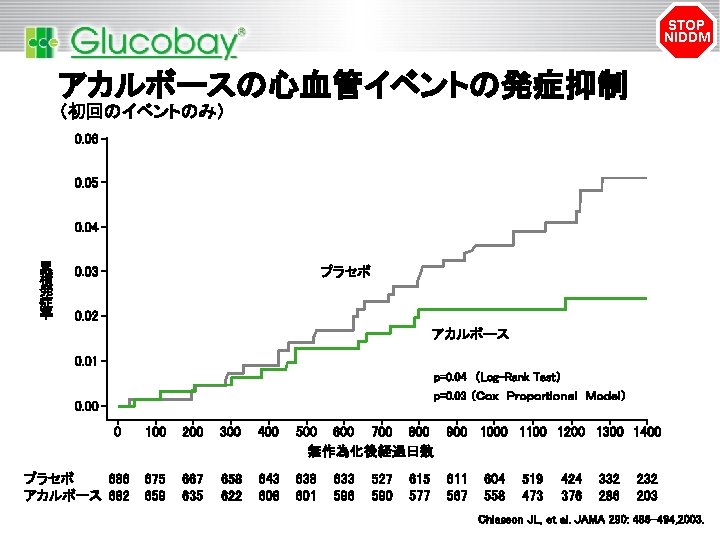
STOP NIDDM アカルボースの心血管イベントの発症抑制 (初回のイベントのみ) 0. 06 0. 05 0. 04 累 積 発 症 率 0. 03 プラセボ 0. 02 アカルボース 0. 01 p=0. 04 (Log-Rank Test) p=0. 03 (Cox Proportional Model) 0. 00 0 プラセボ 686 アカルボース 682 100 200 300 400 500 600 700 800 900 1000 1100 1200 1300 1400 無作為化後経過日数 675 659 667 635 658 622 643 608 638 601 633 596 527 590 615 577 611 567 604 558 519 473 424 376 332 286 232 203 Chiasson JL, et al. JAMA 290: 486– 494, 2003.



Methods 1780 eligible patients on a standard diet and taking regular exercise with impaired glucose tolerance were randomly assigned to oral voglibose 0・ 2 mg three times a day (n=897) or placebo (n=883) in a multicentre, doubleblind, parallel group trial. Treatment was continued until participants developed type 2 diabetes (primary endpoint) or normoglycaemia (secondary endpoint), or for a minimum of 3 years, subject to the fi ndings of an interim analysis. Analysis was by full analysis set. This trial is registered with the University Hospital Medical Information Network (UMIN) clinical trials registry, number UMIN 000001109.
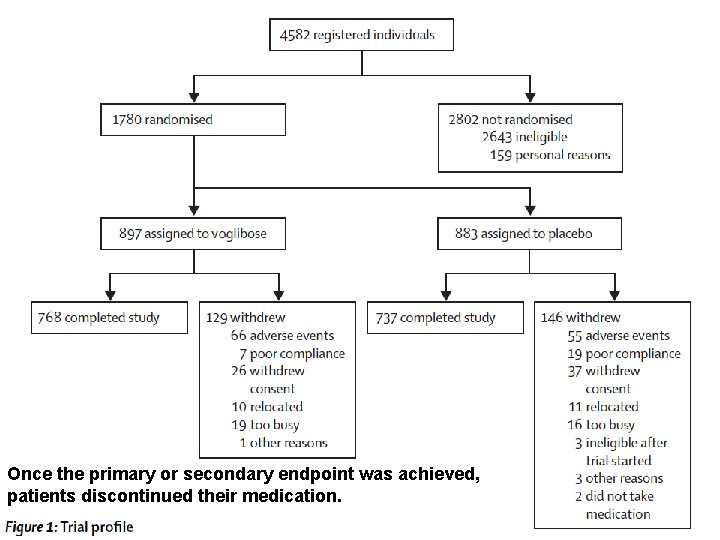
Once the primary or secondary endpoint was achieved, patients discontinued their medication.
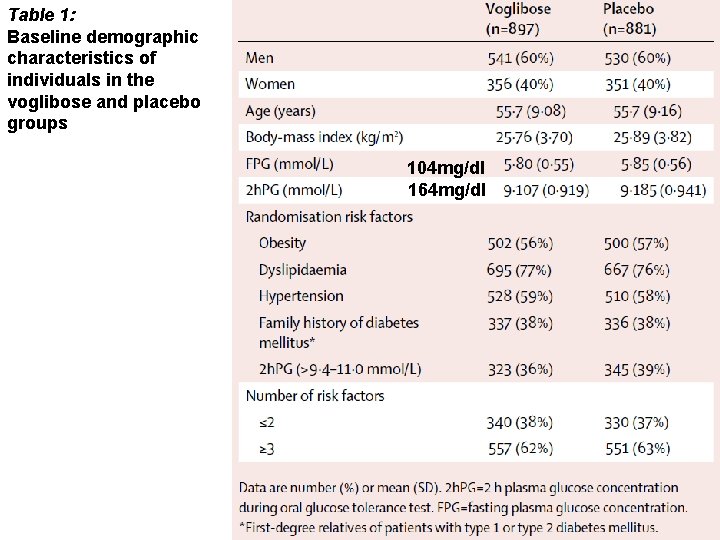
Table 1: Baseline demographic characteristics of individuals in the voglibose and placebo groups 104 mg/dl 164 mg/dl
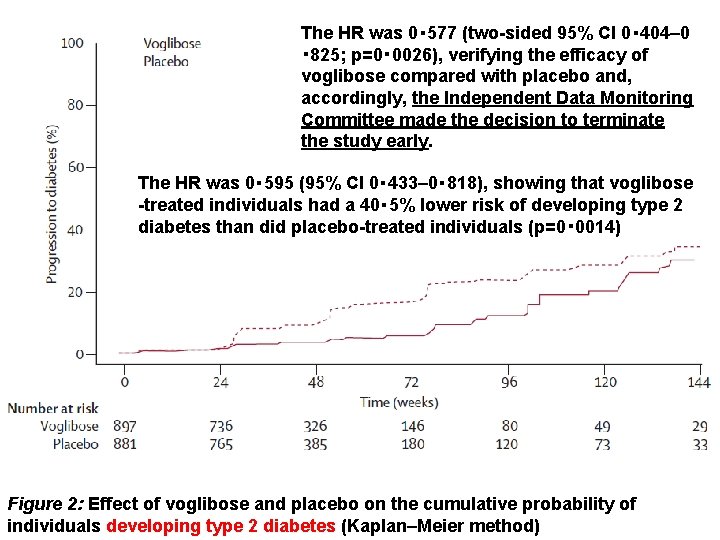
The HR was 0・ 577 (two-sided 95% CI 0・ 404– 0 ・ 825; p=0・ 0026), verifying the efficacy of voglibose compared with placebo and, accordingly, the Independent Data Monitoring Committee made the decision to terminate the study early. The HR was 0・ 595 (95% CI 0・ 433– 0・ 818), showing that voglibose -treated individuals had a 40・ 5% lower risk of developing type 2 diabetes than did placebo-treated individuals (p=0・ 0014) Figure 2: Effect of voglibose and placebo on the cumulative probability of individuals developing type 2 diabetes (Kaplan–Meier method)
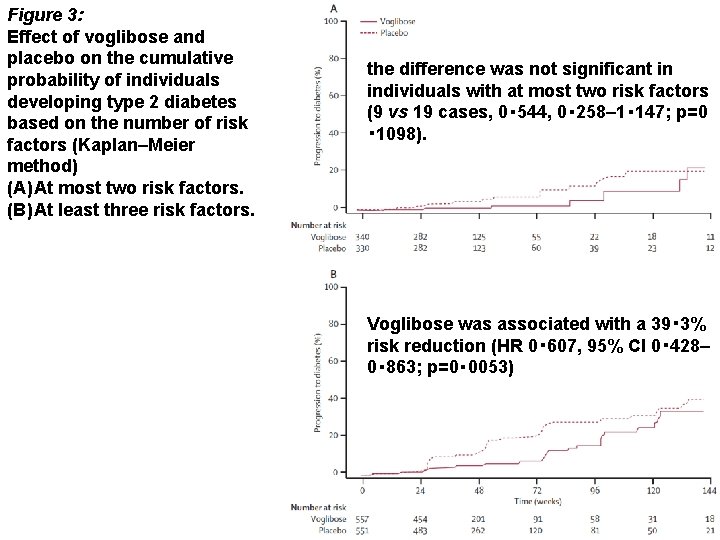
Figure 3: Effect of voglibose and placebo on the cumulative probability of individuals developing type 2 diabetes based on the number of risk factors (Kaplan–Meier method) (A) At most two risk factors. (B) At least three risk factors. the difference was not significant in individuals with at most two risk factors (9 vs 19 cases, 0・ 544, 0・ 258– 1・ 147; p=0 ・ 1098). Voglibose was associated with a 39・ 3% risk reduction (HR 0・ 607, 95% CI 0・ 428– 0・ 863; p=0・ 0053)
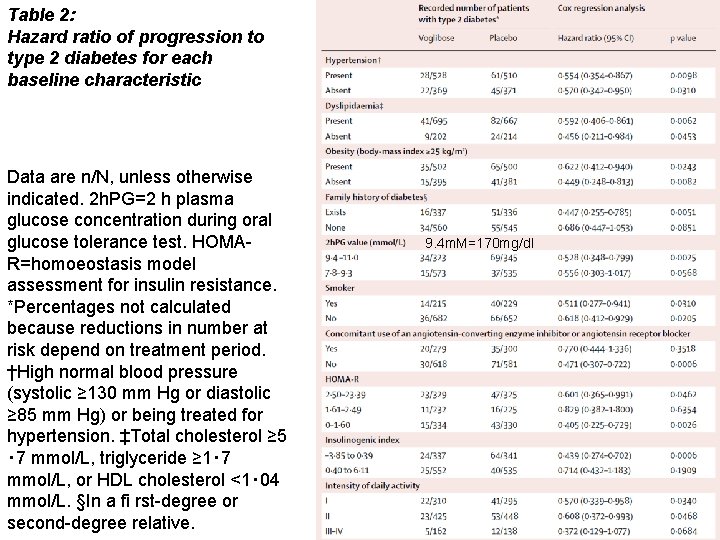
Table 2: Hazard ratio of progression to type 2 diabetes for each baseline characteristic Data are n/N, unless otherwise indicated. 2 h. PG=2 h plasma glucose concentration during oral glucose tolerance test. HOMAR=homoeostasis model assessment for insulin resistance. *Percentages not calculated because reductions in number at risk depend on treatment period. †High normal blood pressure (systolic ≥ 130 mm Hg or diastolic ≥ 85 mm Hg) or being treated for hypertension. ‡Total cholesterol ≥ 5 ・ 7 mmol/L, triglyceride ≥ 1・ 7 mmol/L, or HDL cholesterol <1・ 04 mmol/L. §In a fi rst-degree or second-degree relative. 9. 4 m. M=170 mg/dl

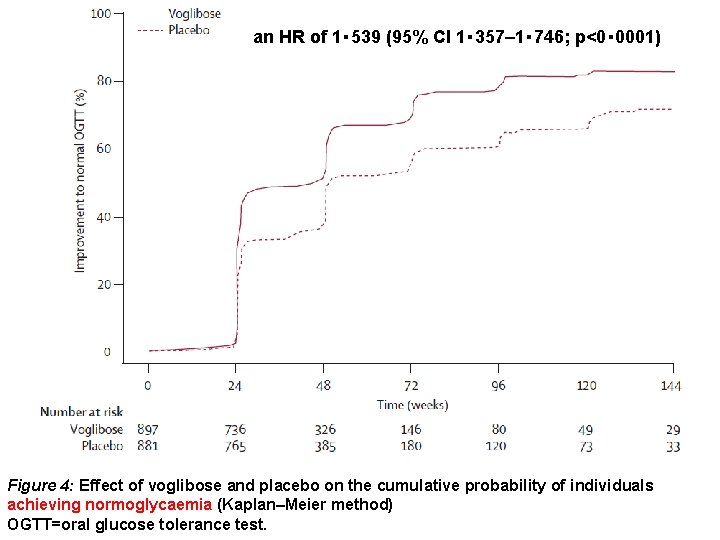
an HR of 1・ 539 (95% CI 1・ 357– 1・ 746; p<0・ 0001) Figure 4: Effect of voglibose and placebo on the cumulative probability of individuals achieving normoglycaemia (Kaplan–Meier method) OGTT=oral glucose tolerance test.
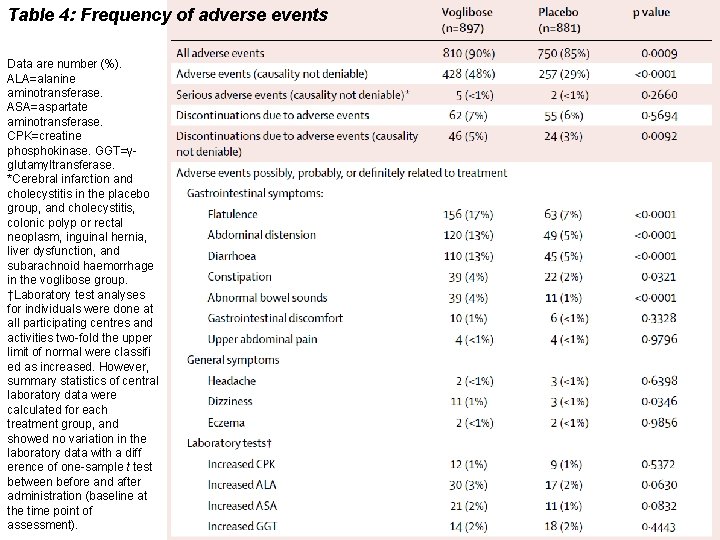
Table 4: Frequency of adverse events Data are number (%). ALA=alanine aminotransferase. ASA=aspartate aminotransferase. CPK=creatine phosphokinase. GGT=γglutamyltransferase. *Cerebral infarction and cholecystitis in the placebo group, and cholecystitis, colonic polyp or rectal neoplasm, inguinal hernia, liver dysfunction, and subarachnoid haemorrhage in the voglibose group. †Laboratory test analyses for individuals were done at all participating centres and activities two-fold the upper limit of normal were classifi ed as increased. However, summary statistics of central laboratory data were calculated for each treatment group, and showed no variation in the laboratory data with a diff erence of one-sample t test between before and after administration (baseline at the time point of assessment).

Findings In the interim analysis, voglibose was better than placebo (p=0・ 0026) in individuals treated for an average of 48・ 1 weeks (SD 36・ 3). Patients treated with voglibose had a lower risk of progression to type 2 diabetes than did those on placebo (50 of 897 vs 106 of 881; hazard ratio 0・ 595, 95% CI 0・ 433– 0・ 818; p=0・ 0014). More people in the voglibose group achieved normoglycaemia than did those in the placebo group (599 of 897 vs 454 of 881; 1・ 539, 1・ 357– 1・ 746; p<0・ 0001). 810 (90%) of 897 patients in the voglibose group had adverse events versus 750 (85%) of 881 in the placebo group. Serious adverse events (all one each) in the voglibose group were cholecystitis, colonic polyp, rectal neoplasm, inguinal hernia, liver dysfunction, and subarachnoid haemorrhage, and in the placebo group were cerebral infarction and cholecystitis.

Interpretation Voglibose, in addition to lifestyle modification, can reduce the development of type 2 diabetes in high-risk Japanese individuals with impaired glucose tolerance. Funding Takeda.





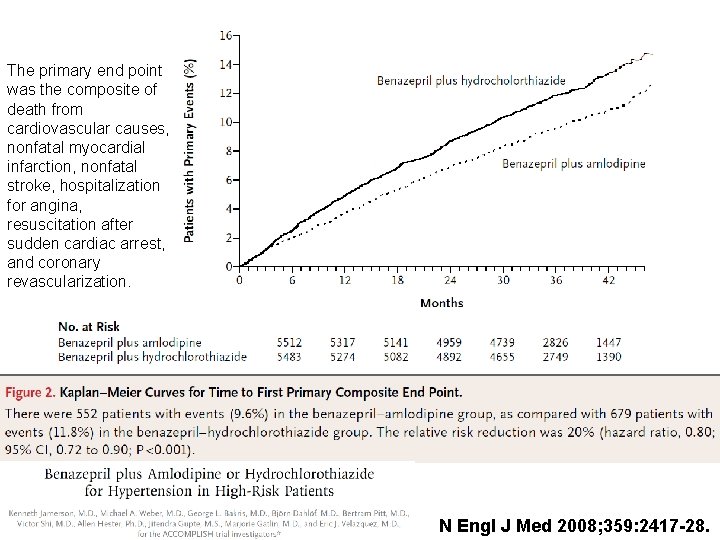
The primary end point was the composite of death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, hospitalization for angina, resuscitation after sudden cardiac arrest, and coronary revascularization. N Engl J Med 2008; 359: 2417 -28.

Background Clinical practice guidelines recommend blockers of the renin-angiotensin system alone or in combination with other agents to reduce blood pressure and albuminuria in patients with type 2 diabetes. Dihydropyridine calcium channel blockers, however, may lower blood pressure but not albuminuria in these patients. Here we tested the hypothesis that combining an ACE inhibitor with either a thiazide diuretic or a calcium channel blocker will cause similar reductions in blood pressure and albuminuria in hypertensive type 2 diabetics.
Methods We conducted a double blind randomized controlled trial on 332 hypertensive, albuminuric type 2 diabetic patients treated with benazepril with either amlodipine or hydrochlorothiazide for 1 year. The trial employed a non-inferiority design.
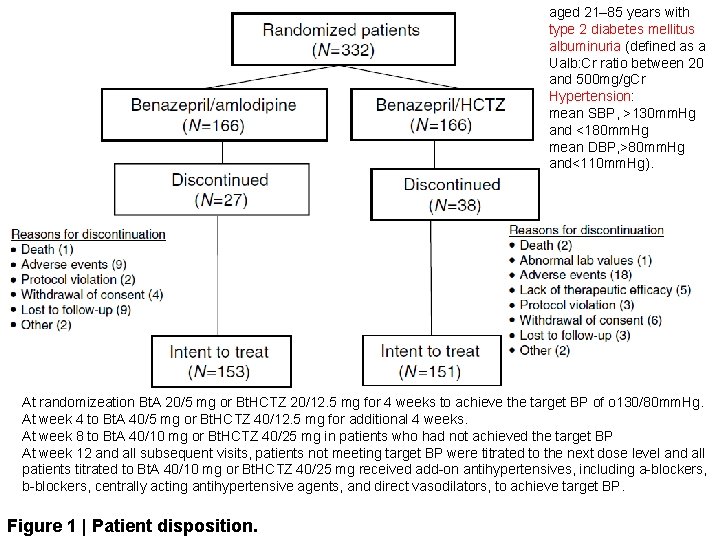
aged 21– 85 years with type 2 diabetes mellitus albuminuria (defined as a Ualb: Cr ratio between 20 and 500 mg/g. Cr Hypertension: mean SBP, >130 mm. Hg and <180 mm. Hg mean DBP, >80 mm. Hg and<110 mm. Hg). At randomizeation Bt. A 20/5 mg or Bt. HCTZ 20/12. 5 mg for 4 weeks to achieve the target BP of o 130/80 mm. Hg. At week 4 to Bt. A 40/5 mg or Bt. HCTZ 40/12. 5 mg for additional 4 weeks. At week 8 to Bt. A 40/10 mg or Bt. HCTZ 40/25 mg in patients who had not achieved the target BP At week 12 and all subsequent visits, patients not meeting target BP were titrated to the next dose level and all patients titrated to Bt. A 40/10 mg or Bt. HCTZ 40/25 mg received add-on antihypertensives, including a-blockers, b-blockers, centrally acting antihypertensive agents, and direct vasodilators, to achieve target BP. Figure 1 | Patient disposition.








Results Both combinations significantly reduced the urinary albumin to creatinine ratio and sitting blood pressure of the entire cohort. The percentage of patients progressing to overt proteinuria was similar for both groups. When we examined patients who had only microalbuminuria and hypertension we found that a larger percentage of the diuretic and ACE inhibitor normalized their albuminuria.

Conclusion We conclude that initial treatment using benzaepril with a diuretic resulted in a greater reduction in albuminuria compared to the group of ACE inhibitor and calcium channel blocker. In contrast, blood pressure reduction, particularly the diastolic component, favored the combination with amilodipine. The dissociation between reductions in blood pressure and albuminuria may be related to factors other than blood pressure.

- Slides: 63