Heat Kinetic Energy at the Nanoscale Heat vs
















- Slides: 19

Heat Kinetic Energy at the Nanoscale

Heat vs. Temperature n n Heat and temperature are related, but they are not the same Temperature is a measure of the average kinetic energy of particles (recall KE = ½mv 2); heat is energy that can be transferred

Heat n n Heat is the amount of energy that can be transferred from one object to another due to a temperature difference. Heat flows from warmer objects to cooler objects.

Heat vs. Temperature n n n Comparing a beaker of boiling water with one drop of boiling water, which is at a higher temperature? which contains more heat?

Temperature Units n Temperature is usually measured in o. F, or o. C n o. C = 5/9(o. F -32) n What is 75 o. F on the Celsius scale?

Temperature Units n The Kelvin scale is based on absolute zero When particles have lost all the kinetic energy they can lose, the object is at absolute zero, 0 K n K + 273 = o. C n

Heat Units n n Since heat is a form of energy, it is often reported in Joules, J Another common heat unit is the calorie 1 calorie = 1 cal = 4. 184 J

The calorie n n n One calorie is the amount of heat needed to raise the temperature of one gram of water by one degree Celsius How much heat to raise temperature of 2 g water by 1 o. C? How much heat to raise temperature of 1 g water by 2 o. C?

Specific Heat n n The amount of heat needed to raise the temperature of one gram of a substance by one degree Celsius is the specific heat, c. The specific heat of water is 1 cal/(g o. C)

Heat n n n q = mcΔT q is the amount of heat that flows m is the mass of the substance c is the specific heat of the substance ΔT is the temperature change

example n n If 100 calories flows into 20 g of water (c = 1 cal/(g o. C)) initially at 20 o. C, what will its temperature become? Why does hot pizza burn the roof of your mouth but not your tongue?

Heat & Phase Changes n Ice bath activity n n Did the temperature of the ice bath change? Did the temperature of the surroundings change? Did heat flow? Was energy conserved?

Heat & Phase Changes n A flow of heat can cause a change in the. . . n n kinetic energy of particles (q = mcΔT) potential energy of the particles (q = m. L) where L is the latent heat

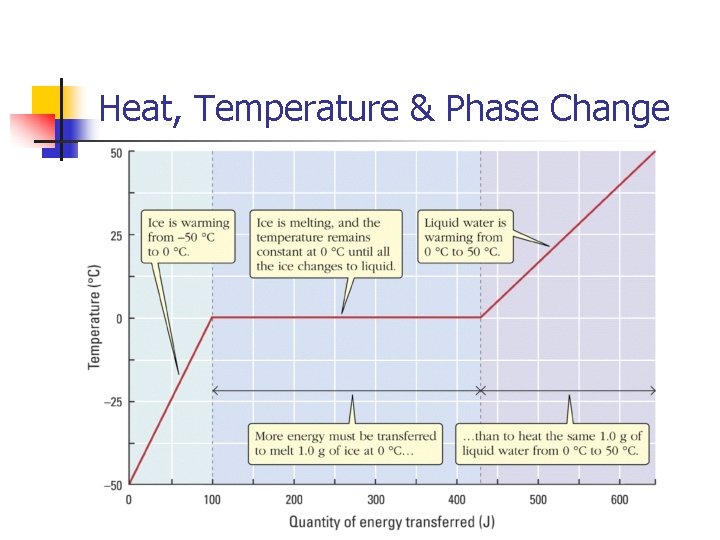
Heat & Phase Changes n Adding heat causes a temperature change UNTIL a phase change begins, then the heat causes the phase change. Ice -10°C heat Temperature change Ice 0°C heat Phase change Water 0°C

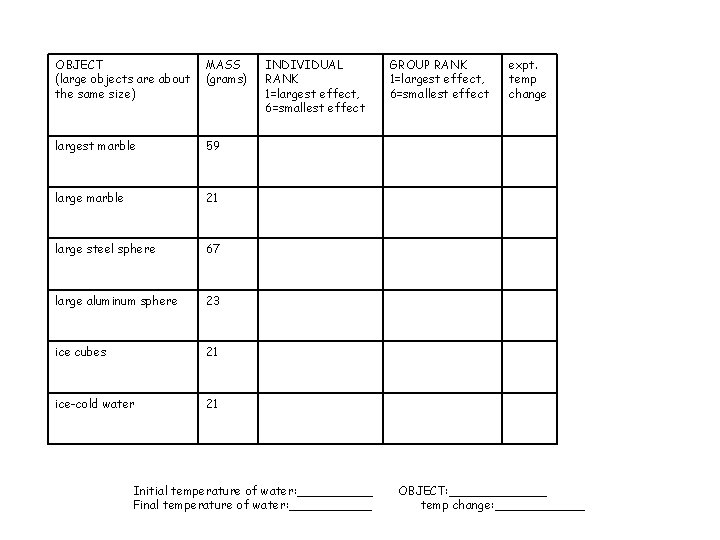
OBJECT (large objects are about the same size) MASS (grams) largest marble 59 large marble 21 large steel sphere 67 large aluminum sphere 23 ice cubes 21 ice-cold water 21 INDIVIDUAL RANK 1=largest effect, 6=smallest effect Initial temperature of water: _____ Final temperature of water: ______ GROUP RANK 1=largest effect, 6=smallest effect expt. temp change OBJECT: _______ temp change: ______

Heat, Temperature & Phase Change

Heat & Phase Changes n How much heat is needed to melt 250 g of ice at 0 o. C into water at 0 o. C? The latent heat of fusion for ice is 333 J/g. q = m. L q = (250 g)(333 J/g) = 83, 250 J

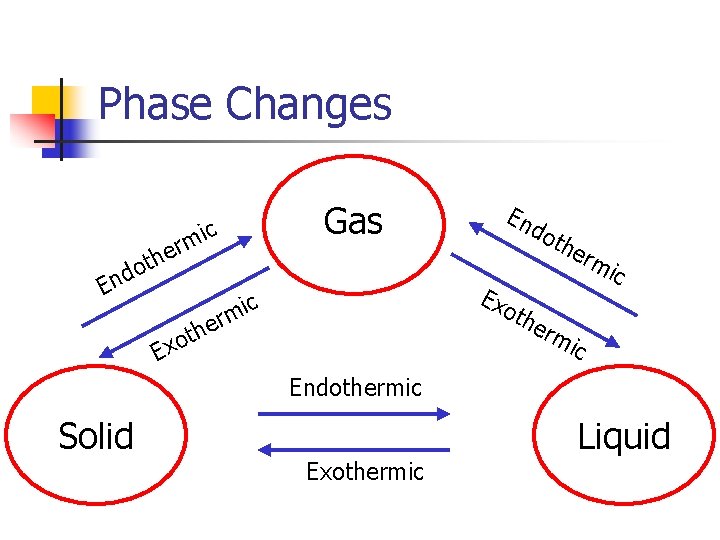
Heat Flow n n When heat is absorbed by a system, the process is called endothermic When heat is released from a system, the process is called exothermic

Phase Changes c mi er th o nd E Gas do Ex ic m er the oth h xot E En rm ic erm ic Endothermic Solid Exothermic Liquid