Heat Engine A heat engine is a device














- Slides: 27

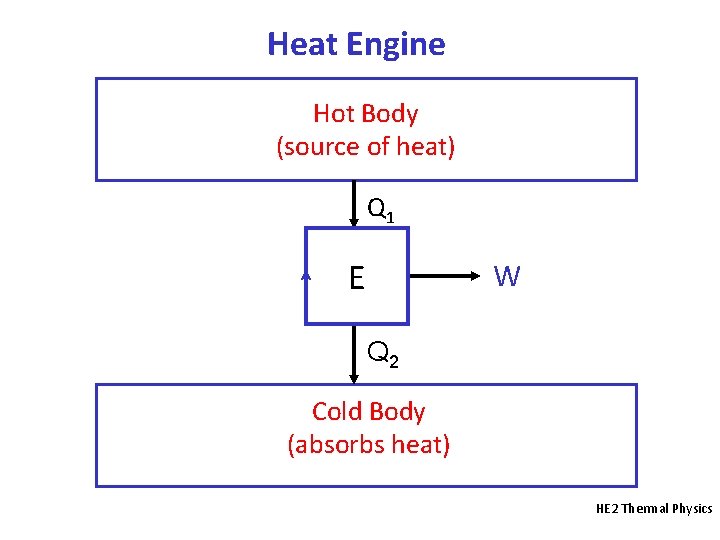
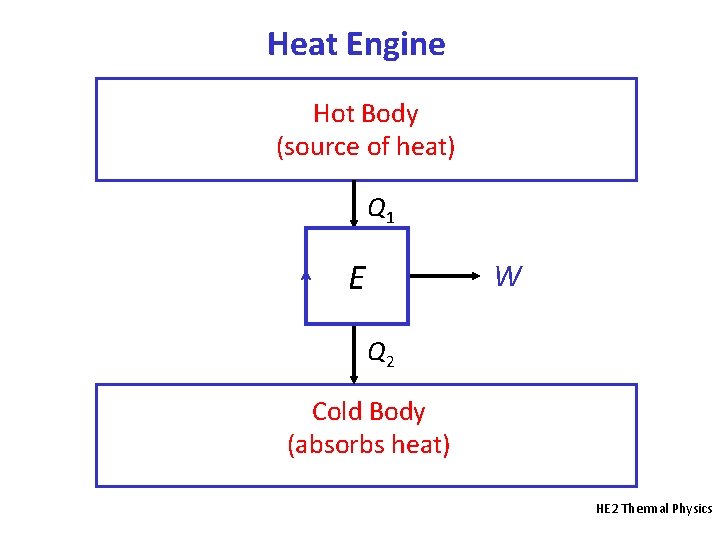
Heat Engine • A heat engine is a device that absorbs heat (Q) and uses it to do useful work (W) on the surroundings when operating in a cycle. • Sources of heat include the combustion of coal, petroleum or carbohydrates and nuclear reactions. • Working substance: the matter inside the heat engine that undergoes addition or rejection of heat and that does work on the surroundings. Examples include air and water vapour (steam). • In a cycle, the working substance is in the same thermodynamic state at the end as at the start. HE 2 Thermal Physics

Heat Engine Hot Body (source of heat) Q 1 E W Q 2 Cold Body (absorbs heat) HE 2 Thermal Physics

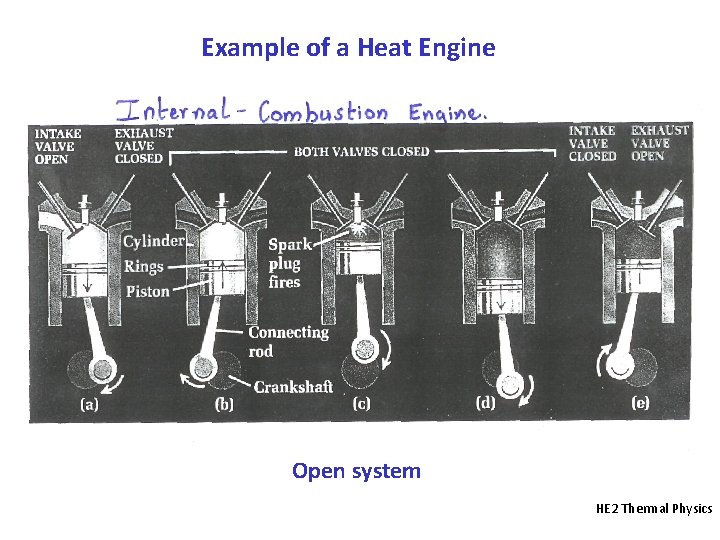
Example of a Heat Engine Open system HE 2 Thermal Physics

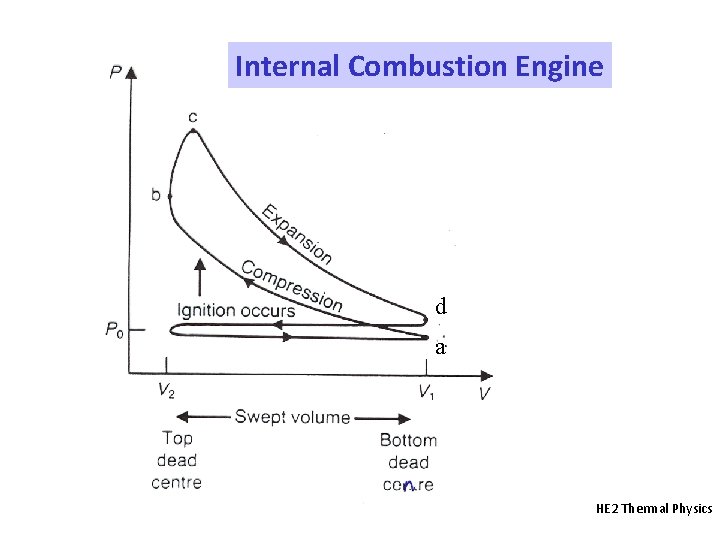
Internal Combustion Engine d a HE 2 Thermal Physics

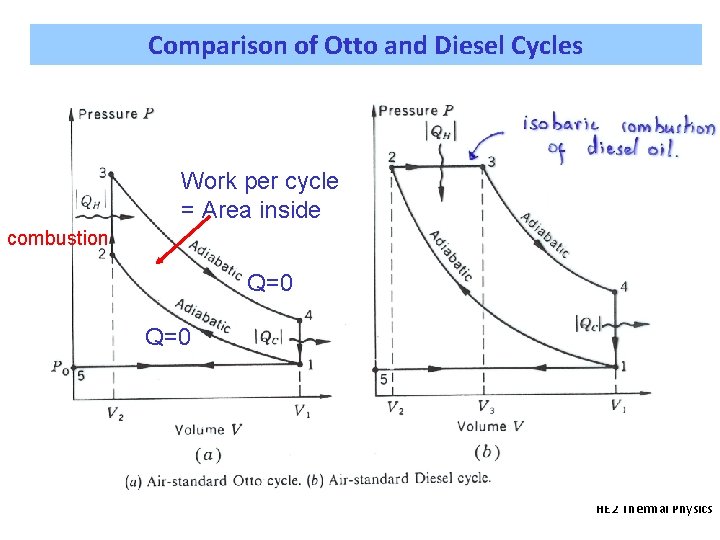
Comparison of Otto and Diesel Cycles Work per cycle = Area inside combustion Q=0 HE 2 Thermal Physics

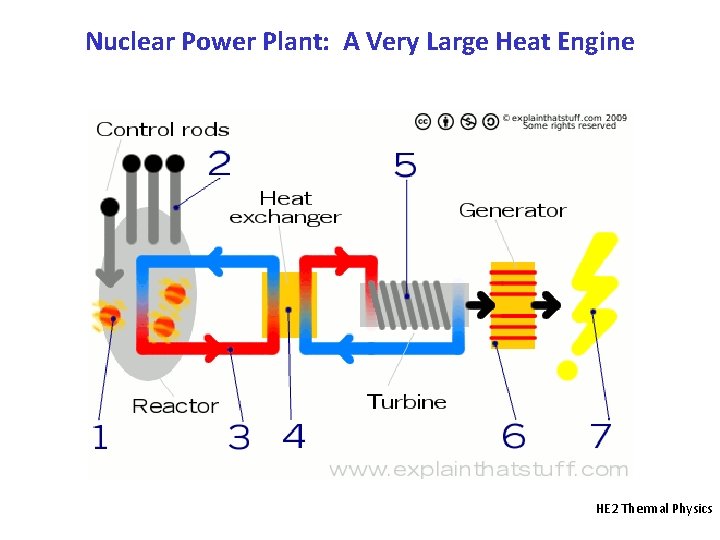
Nuclear Power Plant: A Very Large Heat Engine HE 2 Thermal Physics

Heat Engine Hot Body (source of heat) Q 1 E W Q 2 Cold Body (absorbs heat) HE 2 Thermal Physics

Efficiency of a Heat Engine Efficiency, h = Work out/Heat in: Apply First Law to the working substance: DU = Q 1 – Q 2 – W But in a cycle, DU = 0 Thus, W = Q 1 – Q 2. Substituting: Lesson: h is maximum when Q 2 is minimum. HE 2 Thermal Physics

The Stirling Engine • Closed system See: http: //www. animatedengines. com/ltdstirling. shtml • Operates between two bodies with (small) different temperatures. • Can use “stray” heat HE 2 Thermal Physics

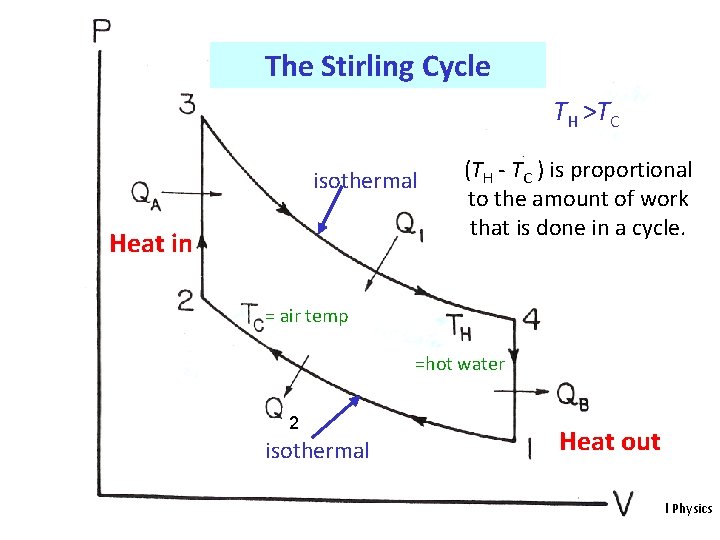
The Stirling Cycle TH >TC isothermal Heat in (TH - TC ) is proportional to the amount of work that is done in a cycle. = air temp =hot water 2 isothermal Heat out HE 2 Thermal Physics

HE 2 Thermal Physics

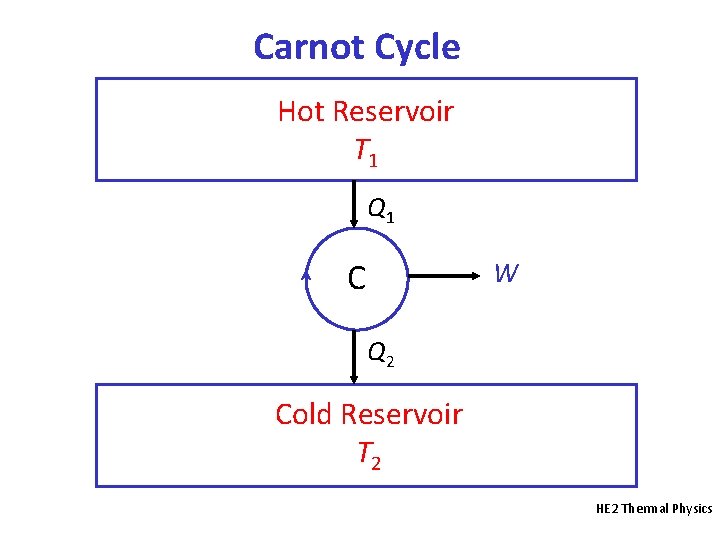
Carnot Cycle Hot Reservoir T 1 Q 1 C W Q 2 Cold Reservoir T 2 HE 2 Thermal Physics

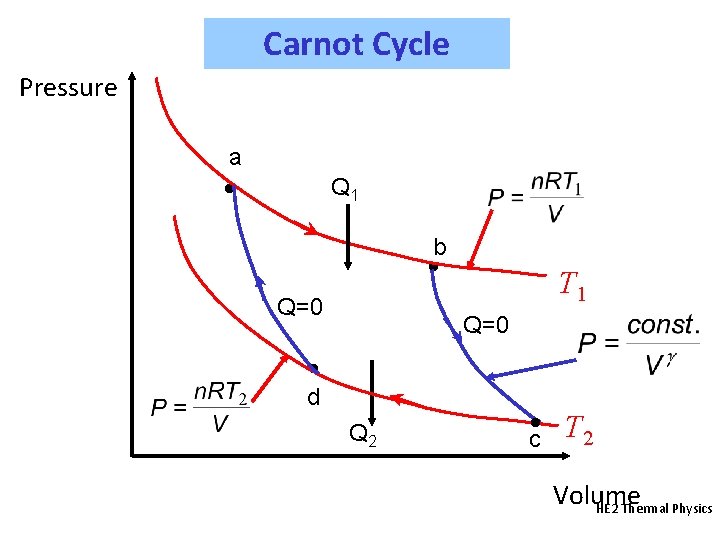
Carnot Cycle Pressure a • Q 1 b • Q=0 T 1 Q=0 • d Q 2 • c T 2 Volume HE 2 Thermal Physics

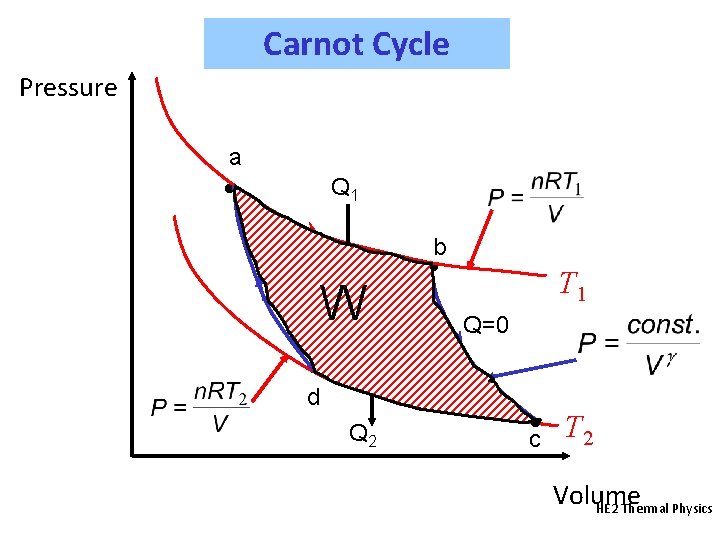
Carnot Cycle Pressure a • Q 1 b W Q=0 • T 1 Q=0 • d Q 2 • c T 2 Volume HE 2 Thermal Physics

Carnot Cycle From a to b: isothermal, so that DU = 0 and Q = - W Thus, Q 1 = +n. RT 1 ln(Vb/Va) (+ve quantity) From b to c: adiabatic, Q = 0, so that TVg-1 is constant. Thus, T 1 Vbg-1 = T 2 Vcg-1 or Similarly, from c to d: isothermal, so that DU = 0 and Q = - W Thus, Q 2 = +n. RT 2 ln(Vd/Vc) = -n. RT 2 ln(Vc/Vd) (-ve) Similarly, d to a: adiabatic, Q = 0, so that TVg-1 is constant. Thus, T 2 Vdg-1 = T 1 Vag-1 or HE 2 Thermal Physics

Carnot Cycle We see that: Which means that Now also: But as the volume ratios are equal: This is an important result. Temperature can be defined (on the absolute (Kelvin) scale) in terms of the heat flows in a Carnot Cycle. HE 2 Thermal Physics

What’s Special about a Carnot Cycle? (1) Heat is transferred to/from only two reservoirs at fixed temperatures, T 1 and T 2 - not at a variety of temperatures. (2) Heat transfer is the most efficient possible because the temperature of the working substance equals the temperature of the reservoirs. No heat is wasted in flowing from hot to cold. (3) The cycle uses an adiabatic process to raise and lower the temperature of the working substance. No heat is wasted in heating up the working substance. (4) Carnot cycles are reversible. Not all cycles are! HE 2 Thermal Physics

What’s Special about a Carnot Cycle? (5) The Carnot theorem states that the Carnot cycle (or any reversible cycle) is the most efficient cycle possible. The Carnot cycle defines an upper limit to the efficiency of a cycle. • Recall that for any cycle, the efficiency of a heat engine is given as: • For an engine using a Carnot cycle, the efficiency is also equal to: • Where T 1 and T 2 are the temperatures of the hot and cold reservoirs, respectively, in degrees Kelvin. As T 2 > 0, hc is always <1. HE 2 Thermal Physics

Efficiency of a Stirling Engine Question: What is the maximum possible efficiency of a Stirling engine operating between room temperature (25 °C) and boiling water (100 °C)? Maximum efficiency would be achieved by a Carnot cycle operating between reservoirs at T 1 = 373 K and T 2 = 298 K. Question: What is the maximum possible efficiency of a Stirling engine operating between room temperature (25 °C) and ice (0 °C)? HE 2 Thermal Physics

Kelvin-Planck Statement of the Second Law of Thermodynamics “It is impossible to construct a device that - operating in a cycle - will produce no other effect than the extraction of heat from a single body and the performance of an equivalent amount of work” Or…A cyclical engine cannot convert heat from a single body completely into work. Some heat must be rejected at a lower temperature. Thus, efficiency, h < 1! HE 2 Thermal Physics

Heat Engine Hot Body (source of heat) Q 1 E W= -Q 1 Q 2 = 0 Cold Body (absorbs heat) HE 2 Thermal Physics

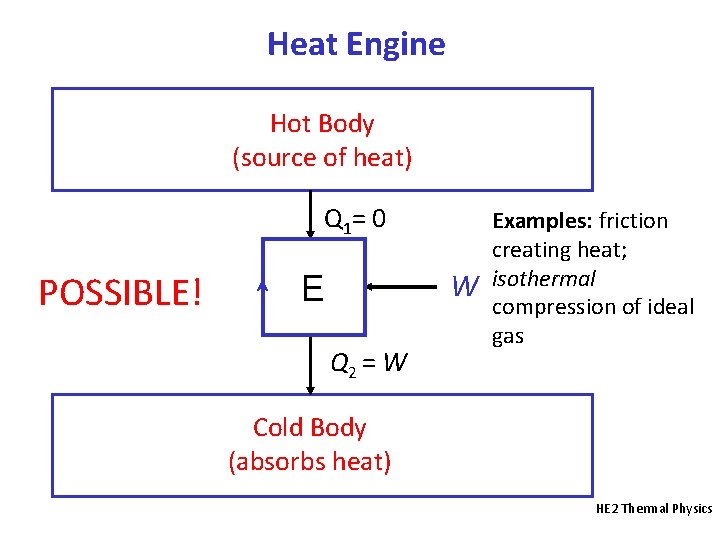
Heat Engine Hot Body (source of heat) Q 1= 0 POSSIBLE! W E Q 2 = W Examples: friction creating heat; isothermal compression of ideal gas Cold Body (absorbs heat) HE 2 Thermal Physics

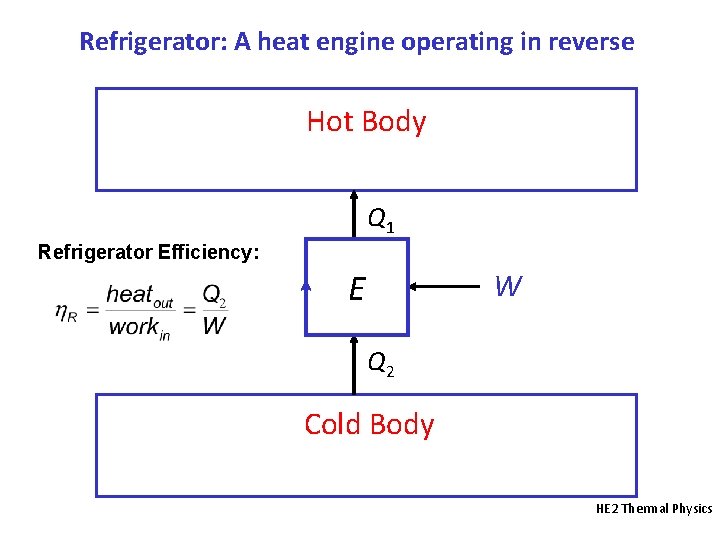
Refrigerator: A heat engine operating in reverse Hot Body Q 1 Refrigerator Efficiency: E W Q 2 Cold Body HE 2 Thermal Physics

Refrigerator Efficiency First Law tells us that Q 2 + W -Q 1 = 0. Thus, W = Q 1 – Q 2 For a Carnot refrigerator, the efficiency is: Efficiency is usually >1! The smaller the T difference, the more efficient is the refrigerator. HE 2 Thermal Physics

Clausius Statement of the Second Law of Thermodynamics (applies to refrigerators) “It is impossible to construct a device that - operating in a cycle - will produce no other effect than heat transfer from a colder body to hotter body. ” “Or…Heat cannot flow from a cold body to a hotter body by itself. Work has to be done in the process. ” HE 2 Thermal Physics

HE 2 Thermal Physics

Efficiency of a Heat Pump The purpose of a heat pump is to extract heat from a cold body (such as the River Thames) and “pump” it to a hot body (such as an office building). The efficiency is defined as the amount of heat pumped in to the hot body per the amount of work done: The First Law tells us that W = Q 1 -Q 2 So, substituting, we find: hhp is always > 1! For maximum h, T 2 should be T 1 (just slightly less). HE 2 Thermal Physics