ERASMUS PLUS 2015 2017 IIS LS PICCOLO CAPO



































- Slides: 54

ERASMUS PLUS 2015 -2017 IIS LS PICCOLO CAPO D’ ORLANDO A 4 INTERDISCIPLINARY CLASS CHEMISTRY BIOLOGY Realised by teacher Bontempo Anna Maria Translation of teacher Sottile Michele

LIPIDS l Lipids (from the Greek lipòs, fat) include numerous substances having different features and properties. They do not dissolve in water but do dissolve in apolar solvents such as carbon tetrachloride, chloroform, etc.

Classification (Nutritional) l. Depositing Lipids or triglycerides (98 %), which have an energetic function. l. Cellular Lipids (2 %) phospholipids, glycolipids and cholesterol, which instead have a structural function.

l. Complex or Saponifiable Lipids have one or more molecoles of fatty acids which, via alcaline hydrolysis, become detached and take the form of soap, that is, of alcaline salts. l. Simple. Lipids or non Saponifiable, which do not contain such molecoles and are not hydrolizable. (Chemical Classification)

Simple. Lipids l Terpenes l Steroides l Prostaglandines

Complex Lipids l l l Acylglycerols Phospholipids Glycolipids Wax Sterides

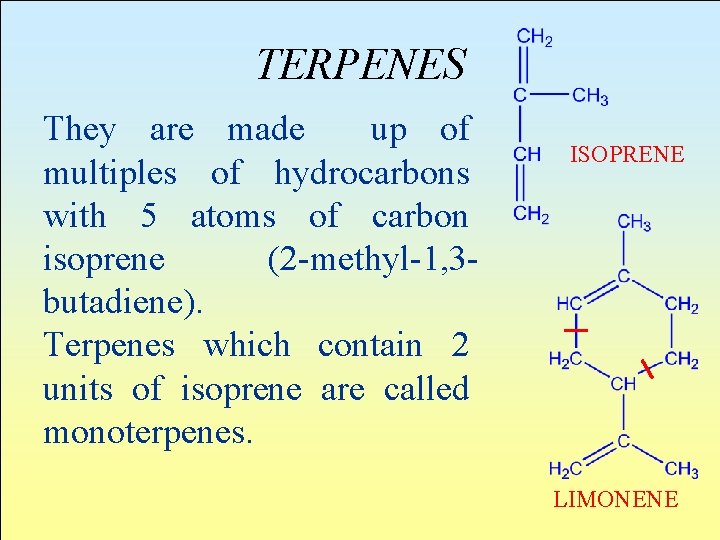
TERPENES They are made up of multiples of hydrocarbons with 5 atoms of carbon isoprene (2 -methyl-1, 3 butadiene). Terpenes which contain 2 units of isoprene are called monoterpenes. ISOPRENE LIMONENE

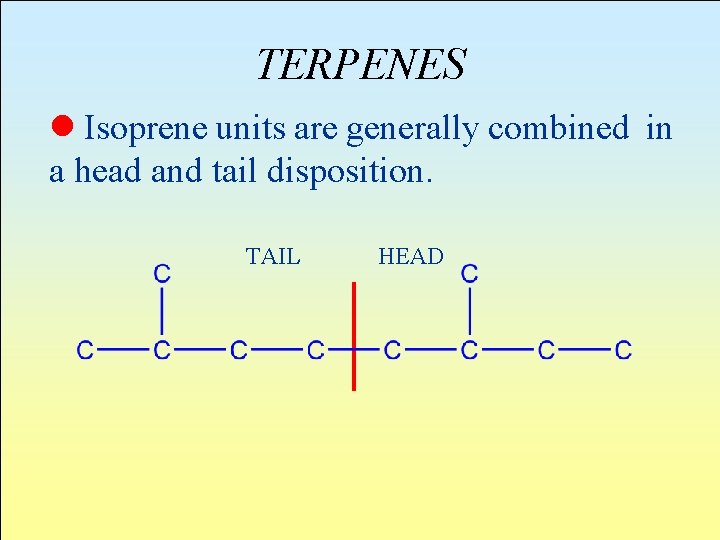
TERPENES l Isoprene units are generally combined in a head and tail disposition. TAIL HEAD

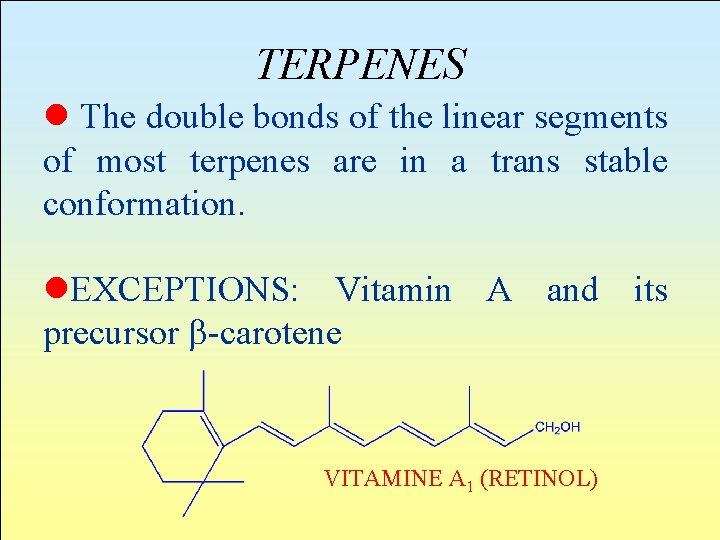
TERPENES l The double bonds of the linear segments of most terpenes are in a trans stable conformation. l. EXCEPTIONS: Vitamin A and its precursor b-carotene VITAMINE A 1 (RETINOL)

ISOPRENE RULE l This classification states that these substances are formed as if they derived from the head-tail polymerization of isoprene.

STEROIDS Steroids derive from the nucleus of a perhydrocyclopentanephenenthrenic nucleus, which contains 3 melted rings of cycloesane in a phenathrene disposition which represents the basic structure of all steroid compounds.

PROSTAGLANDINS l Prostaglandins (PG), derived from polyinsatured fatty acids, are made up of 20 carbon atoms, with a pentatomic ring and 2 lateral chains, one of which has a carboxylic terminal group. PGA 1

Fatty Acids Chemically, they are made up of a carboxylic function, COOH, linked, in its terminal position, to a carbon saturated chain, or else it can show one or more double bonds. They are the common and fundamental components of complex lipids. Inside these they are combined in an ester bond, or else in an amidic one to various types of alcohol (glycerol, sphyngosine, univalent superior alcohols, sterols).

Presence l Fatty acids are present in large quantities (mostly in the form of triglycerides) in food and in cosmetic-use preparations. They also constitute a fundamental component of the cellular membrane which has an important role in the maintaining of homeostasis.

General Features l Fatty acids are known for the length of their carbon chain and for the number and position of their double bonds. Insaturated fatty acids prevail over saturated ones in fats of vegetable origin and in animals which live at low temperatures, while saturated ones are mostly found in fats of animal origin.

Chemical and Physical Features l The nature and position of double bonds in carbon chains determine the chemical and physical features of the fat, such as fusion and solubility points. In natural lipids one finds solely unsaturated fatty acids in a cis form and in non-conjugated double bonds.

Nomenclature l The denomination of a fatty acid takes into account chain length, the number of unsaturations and their position. For example, it is possible to indicate a fatty acid as stearic acid 18: 0, where 18 indicates the number of C atoms and 0 indicates that there are no unsaturations.

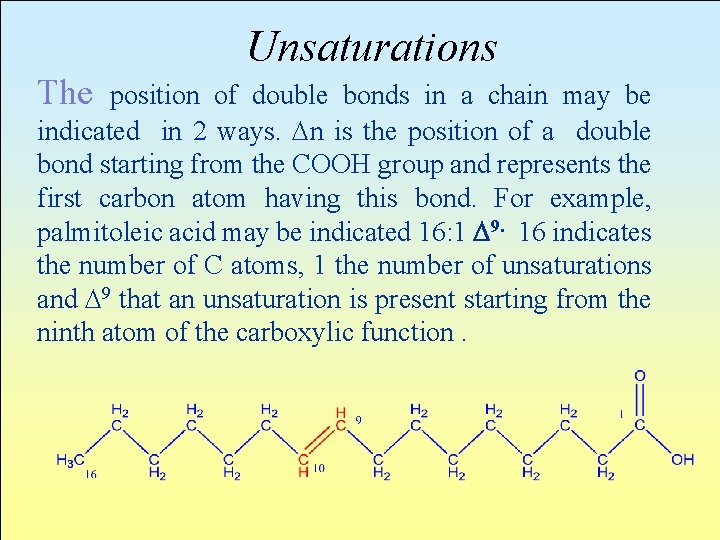
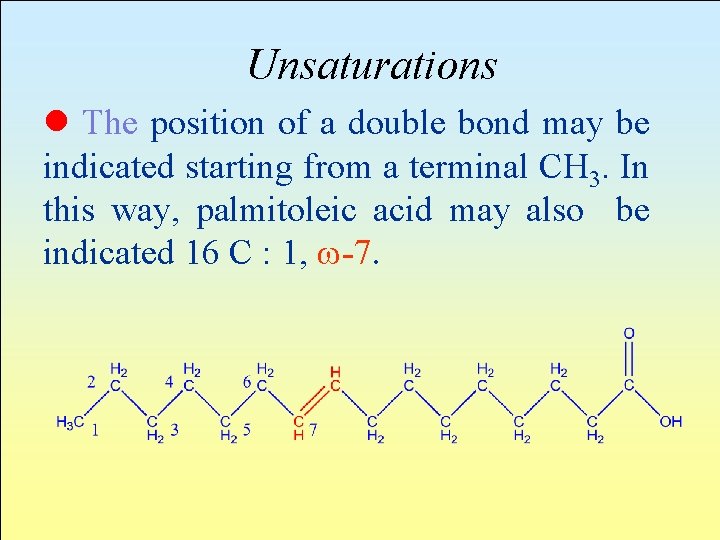
Unsaturations The position of double bonds in a chain may be indicated in 2 ways. Dn is the position of a double bond starting from the COOH group and represents the first carbon atom having this bond. For example, palmitoleic acid may be indicated 16: 1 D 9. 16 indicates the number of C atoms, 1 the number of unsaturations and D 9 that an unsaturation is present starting from the ninth atom of the carboxylic function.

Unsaturations l The position of a double bond may be indicated starting from a terminal CH 3. In this way, palmitoleic acid may also be indicated 16 C : 1, -7.

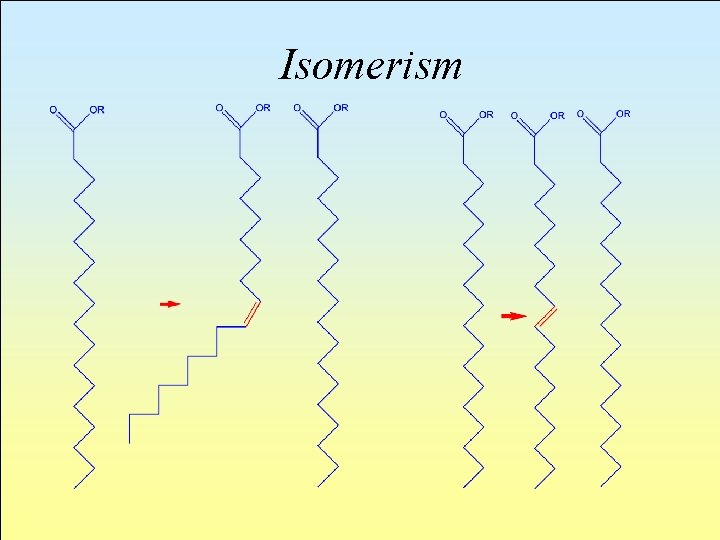
Isomerism l Geometrical isomerisms (cis- e trans-) have fundamental importance from a biological point of view. In the solid phase, the molecules of a fat come together as close as possible. The closer they are, the more effective the adhesive forces (Van der Waals Forces) acting among themselves will be and therefore the fusion point will also be higher.

l The chains of trans-unsaturated acids may spread rather homogeneously and, by tightly coming side by side, establish elevated intermolecular interactions and therefore elevated fusion points as well. A cisunsaturated chain forms folds which oppose the moving closer together of structures, with a consequent decrease in intermolecular interaction and a lowering of the fusion point.

Isomerism

Essential Fatty Acids l These are fatty acids which the organism synthesizes only in a in minimum part, and therefore are not sufficient to satisfy daily necessities. So they must be introduced through the daily diet. Among these we should mention linoleic acid, linolenic acid, arachidonic acid and palmitoleic acid.

The low availability of these acids in some people’s diet has led the food industry to introduce more familiar products into the market (such as milk), with added -3 fatty acids that are naturally present in fish.

Triglycerides l Triglycerides (or triacilglyceroles) are the result of the esterification of glycerole with three fatty acids having a long chain. If only one glycerole hydroxyl is esterificated, the result is a monoglyceride but if the esterification is of two hydroxyls, then we obtain a diglyceride.

Triglicerides l These last two categories of compounds are of technological interest because they are used to favour manufacturing processes and to make for a softer product. Glycerole: Triglyceride:

Possible Lipid Modifications l Hydrolysis or Souring l Ketone Rancidation l Oxydative Rancidation

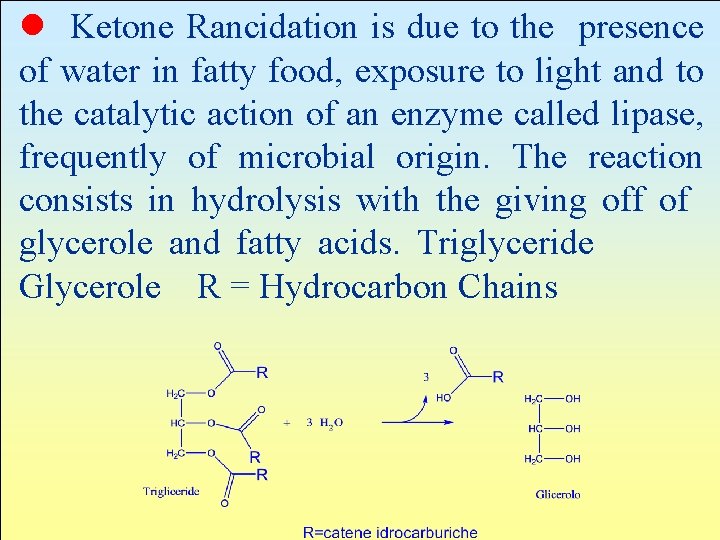
l Ketone Rancidation is due to the presence of water in fatty food, exposure to light and to the catalytic action of an enzyme called lipase, frequently of microbial origin. The reaction consists in hydrolysis with the giving off of glycerole and fatty acids. Triglyceride Glycerole R = Hydrocarbon Chains

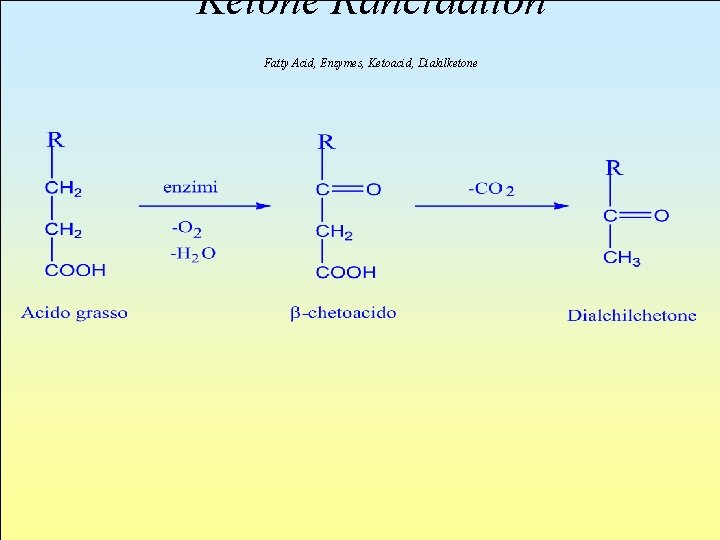
l This alteration strikes fatty acids having low molecular weight previously hydrolized, and is mainly due to the action of enzymes of microbial origin. Reactions consist in oxydation and subsequent decarboxylation, causing the formation of a dialkilketone with one less C atom than the original one.

Ketone Rancidation Fatty Acid, Enzymes, Ketoacid, Diakilketone

Oxydative Rancidation It is the most important alteration concerning fatty acids and is due to absorption of oxygen by unsaturated, free or esterified fatty acids. The reaction is brought about by light, heat, traces of peroxide metals and sometimes by the presence of an enzyme called la lipoxydasis.

The fats which are more easily prone to this alteration are those that, following rectification, have lost natural antioxydants such as Vitamin E. Oxydative rancidation occurs according to the typical process of radical type reactions, in three phases, induction, propagation and termination.

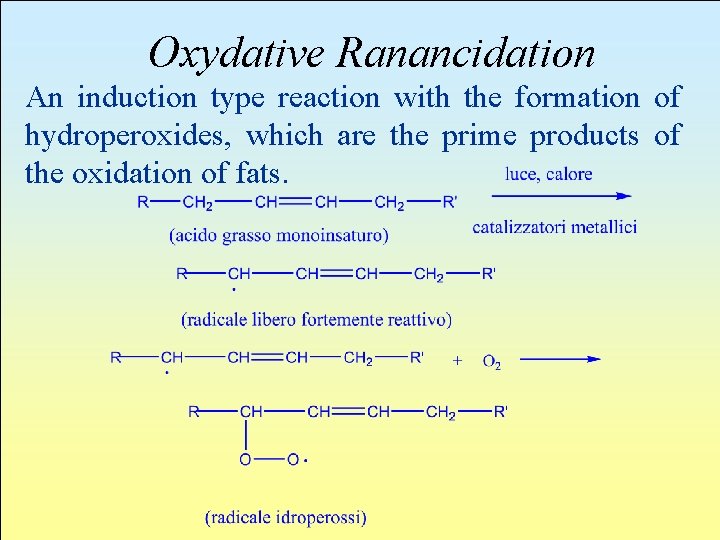
Oxydative Ranancidation An induction type reaction with the formation of hydroperoxides, which are the prime products of the oxidation of fats.

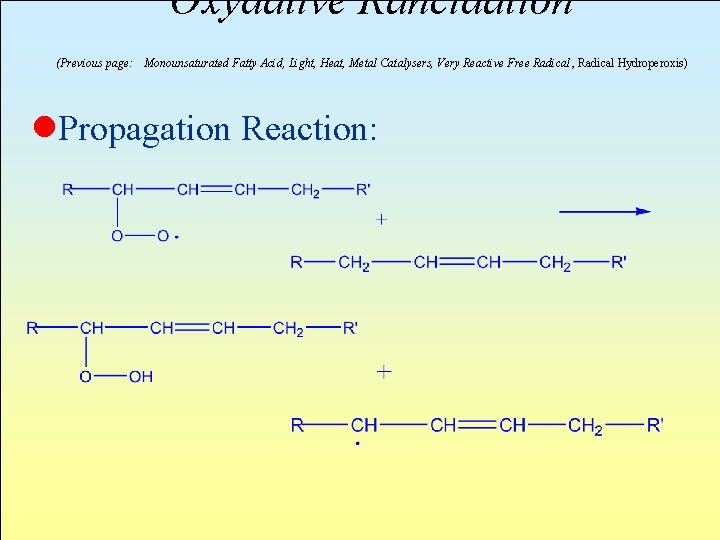
Oxydative Rancidation (Previous page: Monounsaturated Fatty Acid, Light, Heat, Metal Catalysers, Very Reactive Free Radical , Radical Hydroperoxis) l. Propagation Reaction:

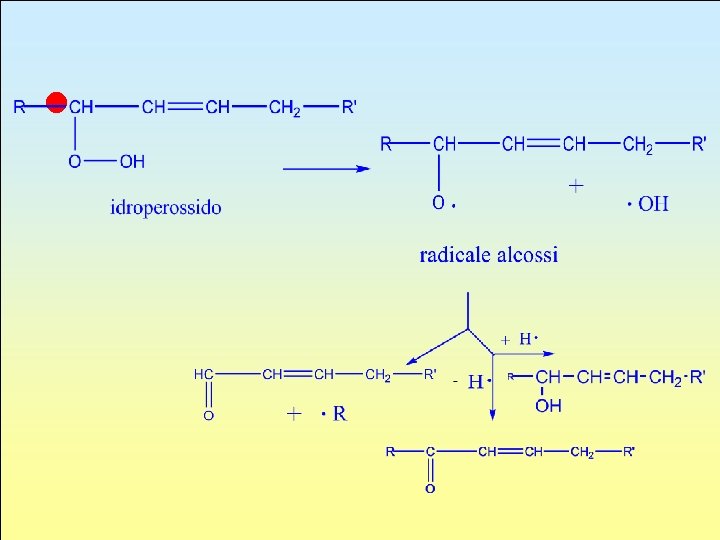
l

Phospholipids (Previous page: Hydroperoxide, Radical Alcolssis) l The phosphoglyceride from which all the others derive is phosphatic acid (PA). It has a structure similar to a triglyceride except for the fact that in position 3 it is esterified with a phosphoric group.

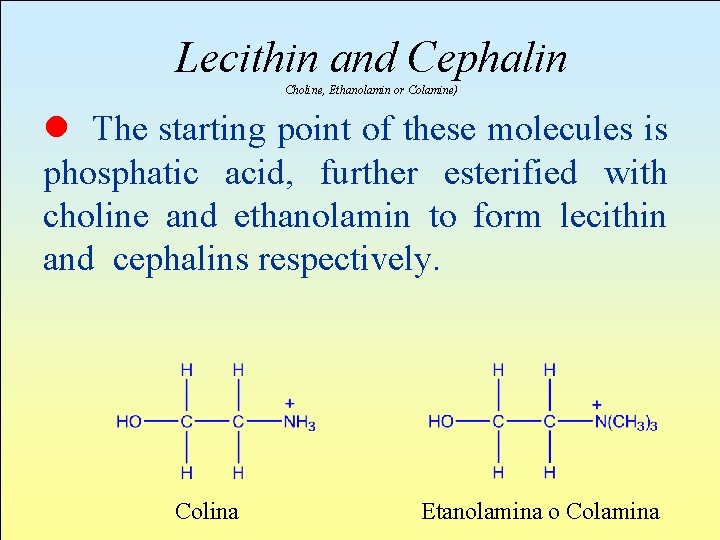
Lecithin and Cephalin Choline, Ethanolamin or Colamine) l The starting point of these molecules is phosphatic acid, further esterified with choline and ethanolamin to form lecithin and cephalins respectively. Colina Etanolamina o Colamina

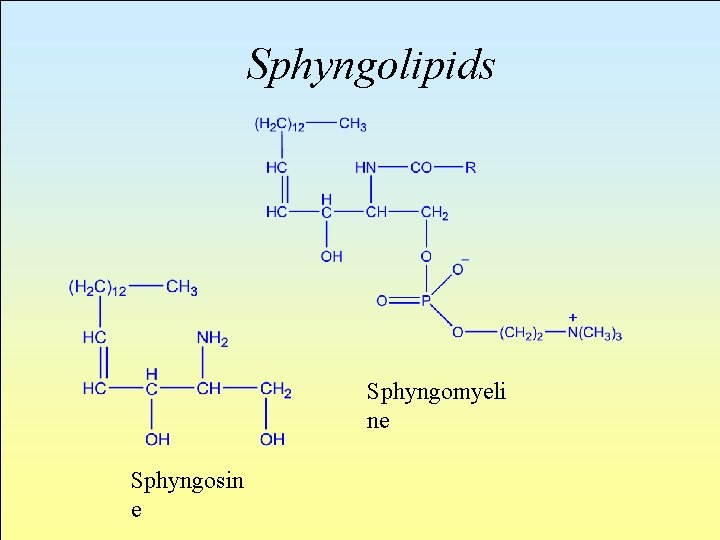
Sphyngolipids In their molecule they have an unsaturated aminoalcohol and sphyingosin or its derivatives instead of glycerole. In the simplest case (sphyngomyelin), the sphyngoine NH 2 is bonded to a fatty acid and the primary OH is esterified with phosphoric acid bonded to choline.

Sphyngolipids Sphyngomyeli ne Sphyngosin e

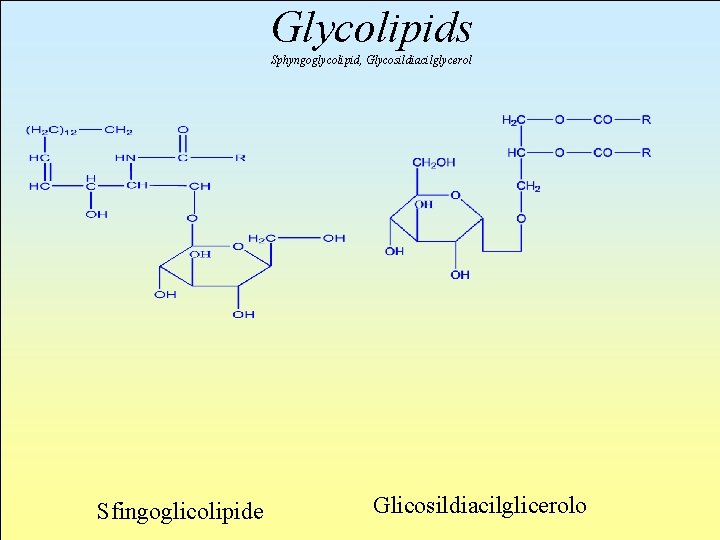
Glycolipids The glycolipid class features a residual glycosidic bonded to a lipid structure. The most important examples of this class are glycosildiacilglyceroles and sphyngoglycolipids.

Glycolipids Sphyngoglycolipid, Glycosildiacilglycerol Sfingoglicolipide Glicosildiacilglicerolo

Wax is an ester of fatty acids having an elevated number of carbon atoms with monohydroxylic aliphatic alcohol. It may be saturated and unsaturated, with a linear chain and a branchy chain.

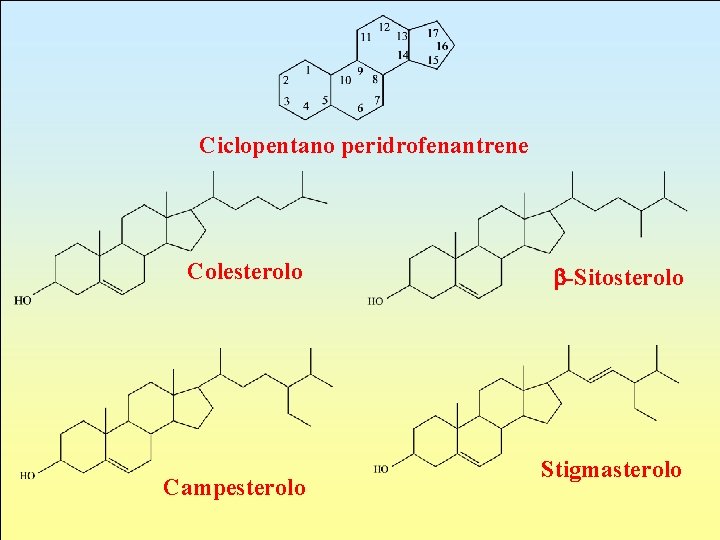
Sterids These are sterol esters with fatty acids. Sterol, which is solid alcohol and can be found in a free or esterified state, comes from peridrophenanthrene cyclopentane, which is a polycyclical hydrocarbon.

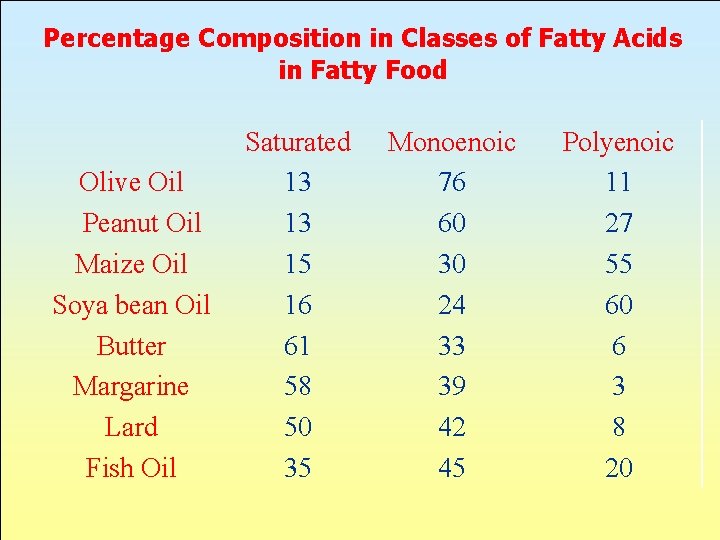
Percentage Composition in Classes of Fatty Acids in Fatty Food Olive Oil Peanut Oil Maize Oil Soya bean Oil Butter Margarine Lard Fish Oil Saturated 13 13 15 16 61 58 50 35 Monoenoic 76 60 30 24 33 39 42 45 Polyenoic 11 27 55 60 6 3 8 20

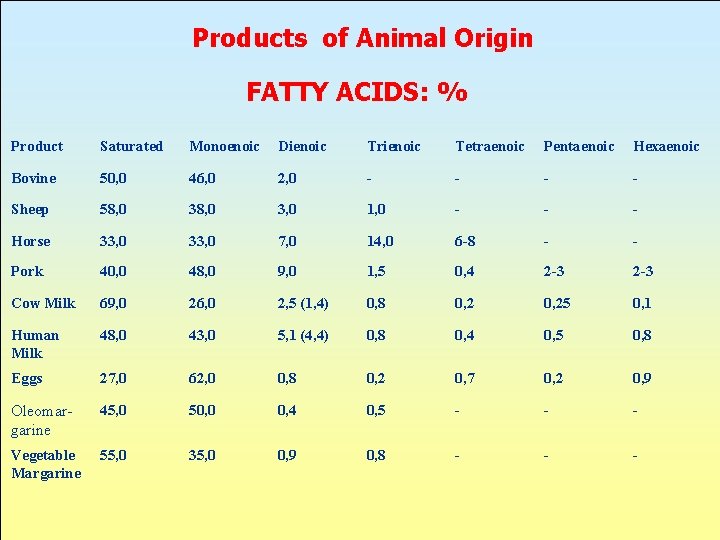
Products of Animal Origin FATTY ACIDS: % Product Saturated Monoenoic Dienoic Trienoic Tetraenoic Pentaenoic Hexaenoic Bovine 50, 0 46, 0 2, 0 - - Sheep 58, 0 3, 0 1, 0 - - - Horse 33, 0 7, 0 14, 0 6 -8 - - Pork 40, 0 48, 0 9, 0 1, 5 0, 4 2 -3 Cow Milk 69, 0 26, 0 2, 5 (1, 4) 0, 8 0, 25 0, 1 Human Milk 48, 0 43, 0 5, 1 (4, 4) 0, 8 0, 4 0, 5 0, 8 Eggs 27, 0 62, 0 0, 8 0, 2 0, 7 0, 2 0, 9 Oleomargarine 45, 0 50, 0 0, 4 0, 5 - - - Vegetable Margarine 55, 0 35, 0 0, 9 0, 8 - - -

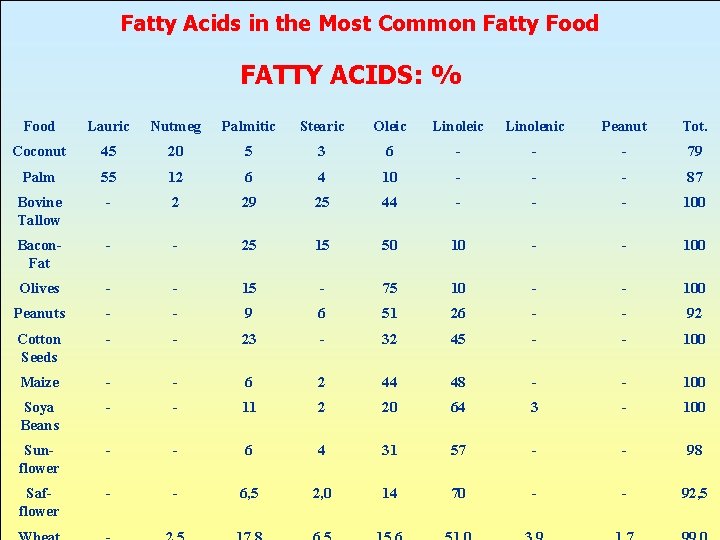
Fatty Acids in the Most Common Fatty Food FATTY ACIDS: % Food Lauric Nutmeg Palmitic Stearic Oleic Linolenic Peanut Tot. Coconut 45 20 5 3 6 - - - 79 Palm 55 12 6 4 10 - - - 87 Bovine Tallow - 2 29 25 44 - - - 100 Bacon. Fat - - 25 15 50 10 - - 100 Olives - - 15 - 75 10 - - 100 Peanuts - - 9 6 51 26 - - 92 Cotton Seeds - - 23 - 32 45 - - 100 Maize - - 6 2 44 48 - - 100 Soya Beans - - 11 2 20 64 3 - 100 Sunflower - - 6 4 31 57 - - 98 Safflower - - 6, 5 2, 0 14 70 - - 92, 5

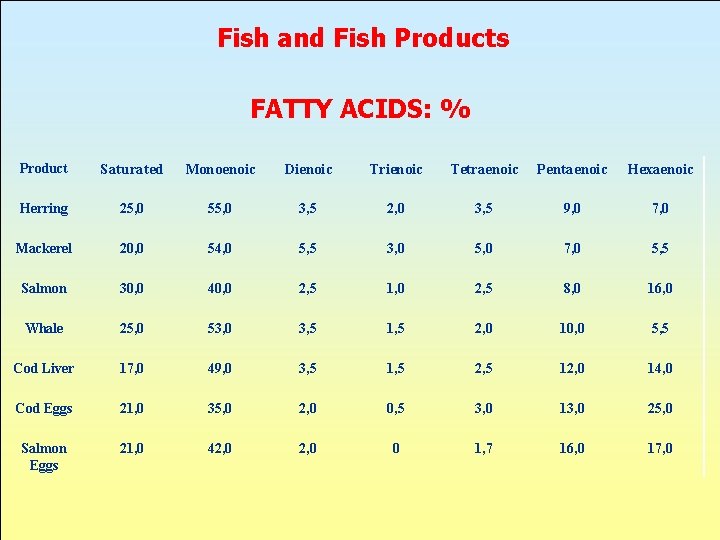
Fish and Fish Products FATTY ACIDS: % Product Saturated Monoenoic Dienoic Trienoic Tetraenoic Pentaenoic Hexaenoic Herring 25, 0 55, 0 3, 5 2, 0 3, 5 9, 0 7, 0 Mackerel 20, 0 54, 0 5, 5 3, 0 5, 0 7, 0 5, 5 Salmon 30, 0 40, 0 2, 5 1, 0 2, 5 8, 0 16, 0 Whale 25, 0 53, 0 3, 5 1, 5 2, 0 10, 0 5, 5 Cod Liver 17, 0 49, 0 3, 5 1, 5 2, 5 12, 0 14, 0 Cod Eggs 21, 0 35, 0 2, 0 0, 5 3, 0 13, 0 25, 0 Salmon Eggs 21, 0 42, 0 0 1, 7 16, 0 17, 0

Funzioni Chimico-biologiche dei grassi Nella dieta • sono una fonte “concentrata” insostituibile di energia: 9 cal/gr • sono fonte di acidi grassi essenziali • veicolano le vitamine liposolubili • rendono i cibi più appetibili e conferiscono senso di sazietà

Funzioni Chimico-biologiche dei grassi Nell’organismo • sono l’unica fonte di “energia di riserva” disponibile ed utilizzabile a medio-lungo termine senza che l’organismo risenta di alcun danno • sono costituenti della struttura cellulare e partecipano al funzionamento delle membrane • sono precursori di sostanze indispensabili per il buon funzionamento delle attività cellulari (ormoni, vitamine, prostaglandine) • agiscono sul livello dei lipidi ematici e contribuiscono a regolare le concentrazioni di colesterolo • agiscono come isolanti termici, proteggono e sostengono gli organi • modellano il corpo disponendosi in modo diverso nell’uomo e nella donna

Importanza Metabolica dei Fosfolipidi

Limiti di Variabilità del Contenuto di Colesterolo in alcuni Alimenti ALIMENTO Colesterolo mg/100 g di parte edibile Uovo intero 450 -480 Tuorlo d’uovo 1700 -1900 Albume d’uovo 0 Cervello 1500 -2500 Cuore 120 -180 Fegato 270 -350 Muscolo di manzo 90 -120 Muscolo di vitello 70 -90 Muscolo di maiale 50 -70 Muscolo di pollo 40 -80 Muscolo di agnello 60 -80 Pesci vari 30 -100 Burro 210 -270 Camembert 120 -150 Emmental 130 -155

Contenuto di Colesterolo in alcuni Alimenti (mg x 100 g) Burro 250 Lardo Margarina Strutto Oli di oliva Olio olio di pomodoro Olio di palma Altri oli vegetali 95 Tracce-105 95 0 -tracce 12 3, 8 0 -tracce

Ciclopentano peridrofenantrene Colesterolo Campesterolo b-Sitosterolo Stigmasterolo

Zoosteroli di origine animale (Colesterolo) Fitosteroli di origine vegetale (Sitosterolo) Micosteroli nei lieviti e nei funghi (Ergosterolo) nelle spugne (Spongosteroli)