Energy Capacity to do work What is energy



- Slides: 59

Energy

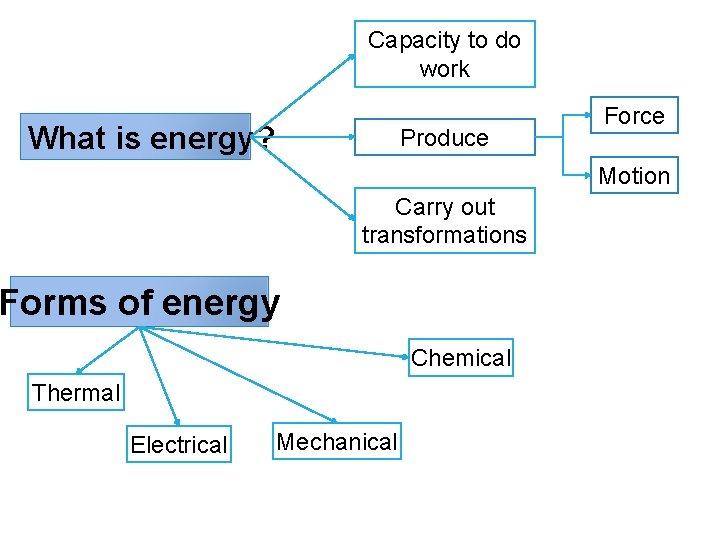
Capacity to do work What is energy? Produce Force Motion Carry out transformations Forms of energy Chemical Thermal Electrical Mechanical

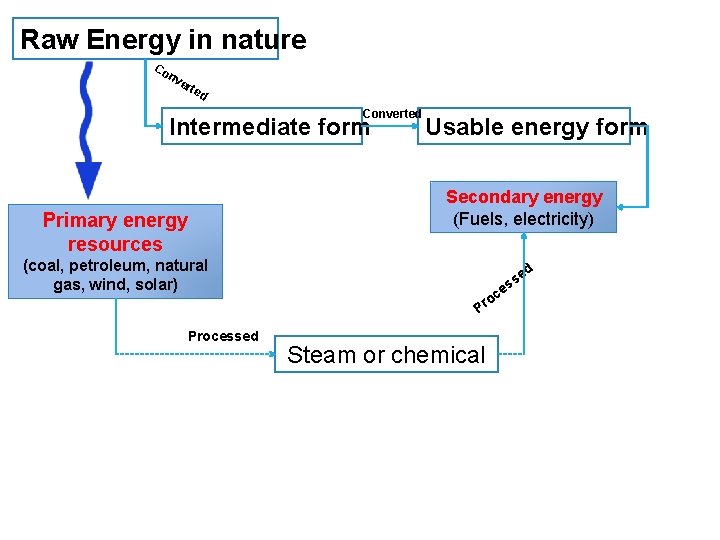
Raw Energy in nature Co nv ert ed Converted Intermediate form Primary energy resources Usable energy form Secondary energy (Fuels, electricity) (coal, petroleum, natural gas, wind, solar) d e c ro P Processed e ss Steam or chemical

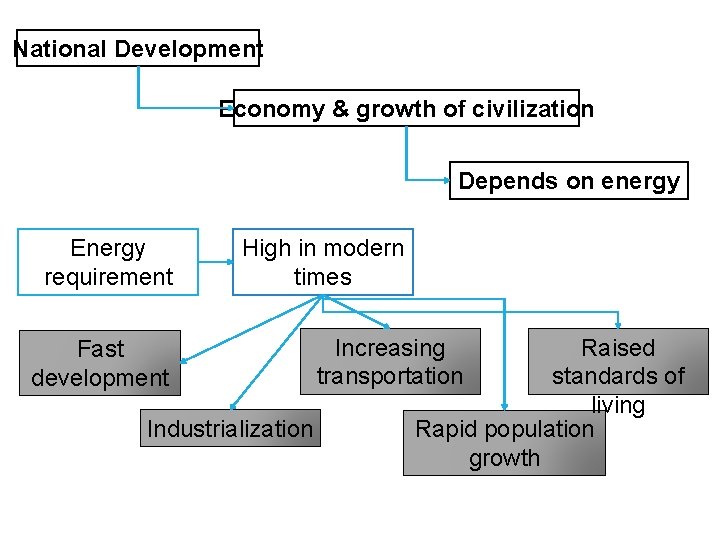
National Development Economy & growth of civilization Depends on energy Energy requirement High in modern times Fast development Industrialization Raised standards of living Rapid population growth Increasing transportation

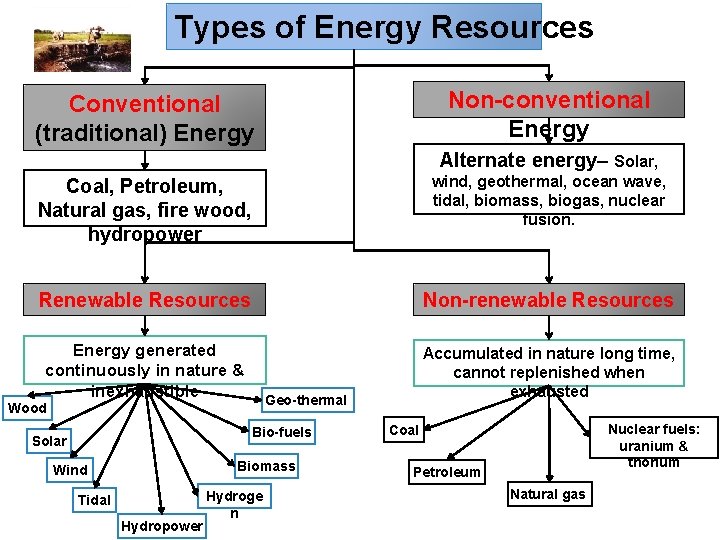
Types of Energy Resources Non-conventional Energy Conventional (traditional) Energy Alternate energy– Solar, Coal, Petroleum, Natural gas, fire wood, hydropower wind, geothermal, ocean wave, tidal, biomass, biogas, nuclear fusion. Renewable Resources Non-renewable Resources Energy generated continuously in nature & inexhaustible Accumulated in nature long time, cannot replenished when exhausted Geo-thermal Wood Bio-fuels Solar Biomass Wind Tidal Hydropower Hydroge n Nuclear fuels: uranium & thorium Coal Petroleum Natural gas

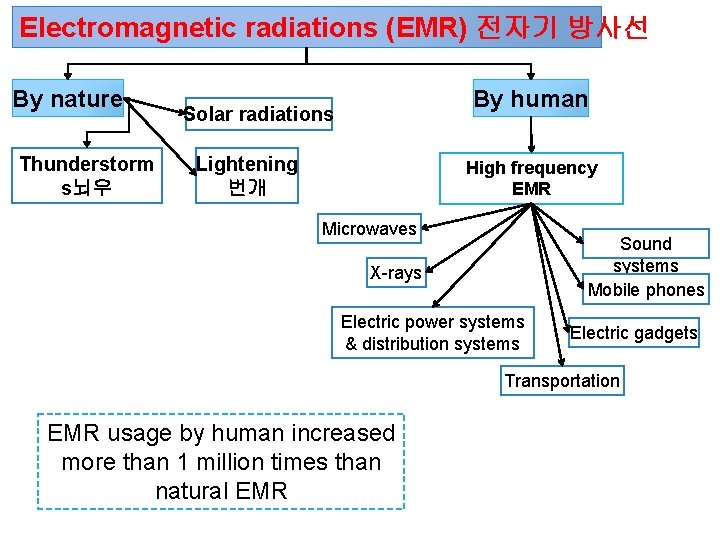
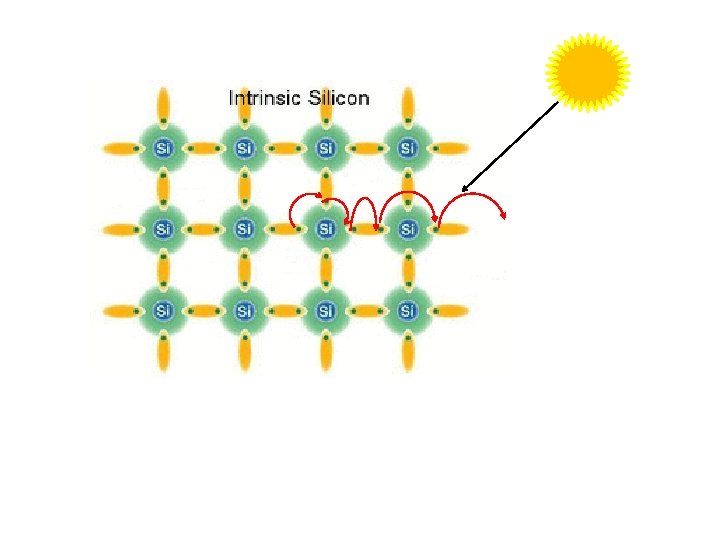
Electromagnetic radiations (EMR) 전자기 방사선 By nature Thunderstorm s뇌우 By human Solar radiations Lightening 번개 High frequency EMR Microwaves Sound systems Mobile phones X-rays Electric power systems & distribution systems Electric gadgets Transportation EMR usage by human increased more than 1 million times than natural EMR

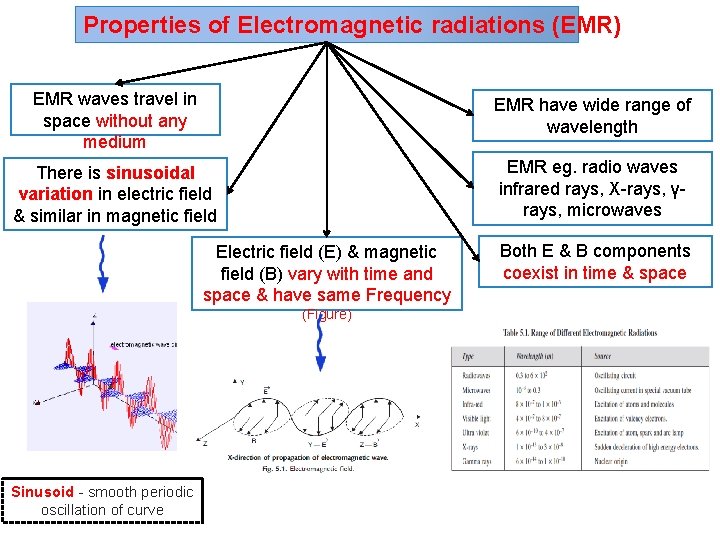
Properties of Electromagnetic radiations (EMR) EMR waves travel in space without any medium EMR have wide range of wavelength There is sinusoidal variation in electric field & similar in magnetic field EMR eg. radio waves infrared rays, X-rays, γrays, microwaves Electric field (E) & magnetic field (B) vary with time and space & have same Frequency (Figure) Sinusoid - smooth periodic oscillation of curve Both E & B components coexist in time & space

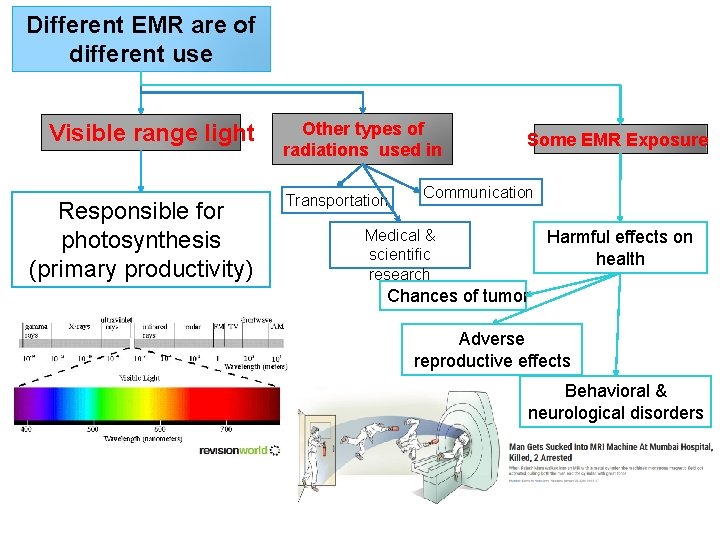
Different EMR are of different use Visible range light Responsible for photosynthesis (primary productivity) Other types of radiations used in Transportation Some EMR Exposure Communication Medical & scientific research Harmful effects on health Chances of tumor Adverse reproductive effects Behavioral & neurological disorders

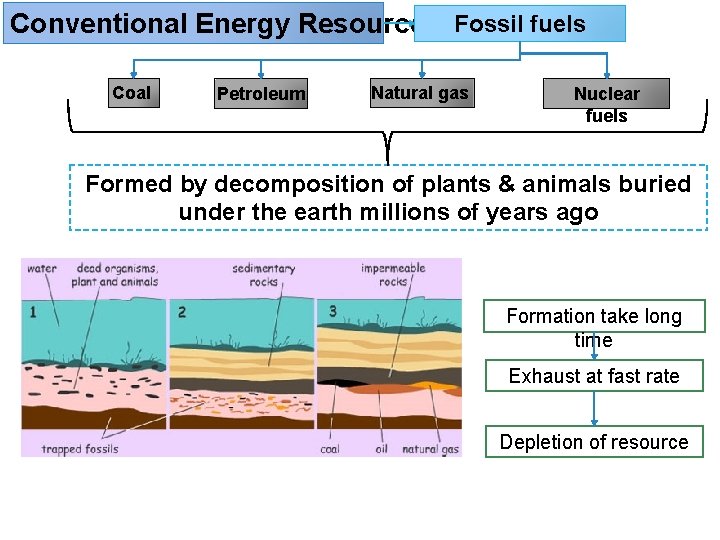
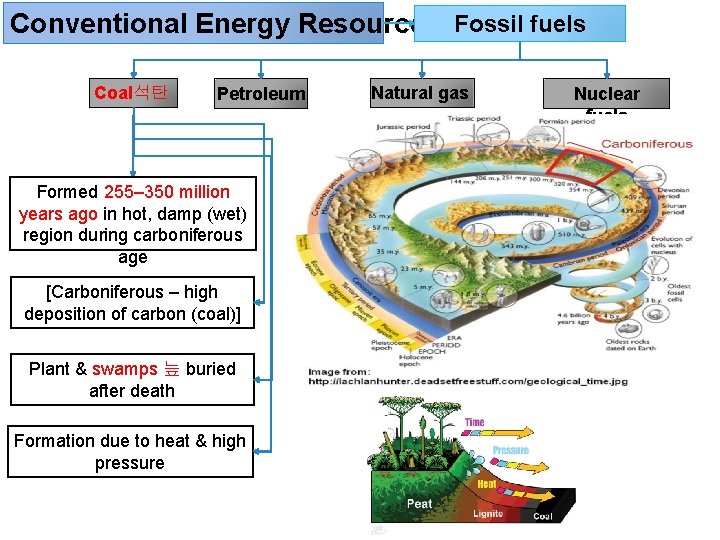
Conventional Energy Resources Fossil fuels Coal Petroleum Natural gas Nuclear fuels Formed by decomposition of plants & animals buried under the earth millions of years ago Formation take long time Exhaust at fast rate Depletion of resource

Conventional Energy Resources Fossil fuels Coal석탄 Petroleum Formed 255– 350 million years ago in hot, damp (wet) region during carboniferous age [Carboniferous – high deposition of carbon (coal)] Plant & swamps 늪 buried after death Formation due to heat & high pressure Natural gas Nuclear fuels

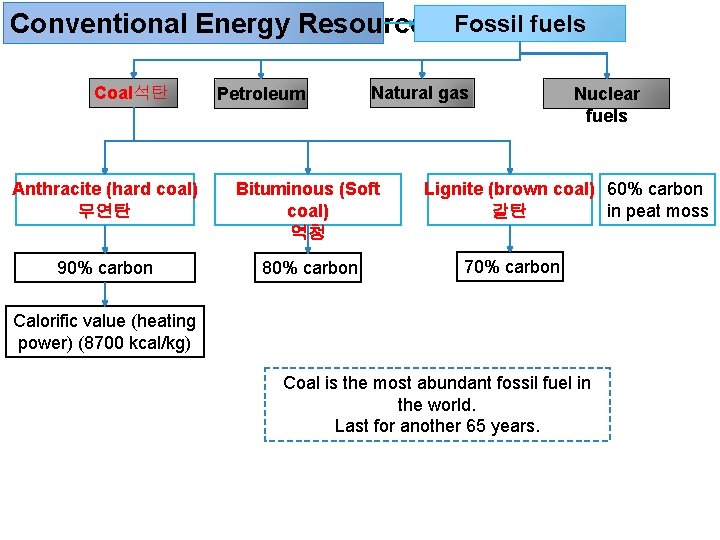
Conventional Energy Resources Fossil fuels Coal석탄 Petroleum Natural gas Anthracite (hard coal) 무연탄 Bituminous (Soft coal) 역청 90% carbon 80% carbon Nuclear fuels Lignite (brown coal) 60% carbon in peat moss 갈탄 70% carbon Calorific value (heating power) (8700 kcal/kg) Coal is the most abundant fossil fuel in the world. Last for another 65 years.

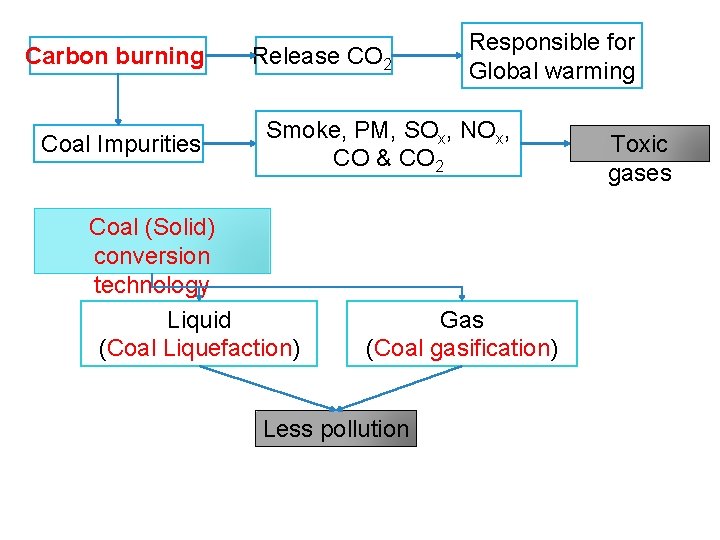
Carbon burning Coal Impurities Release CO 2 Responsible for Global warming Smoke, PM, SOx, NOx, CO & CO 2 Coal (Solid) conversion technology Liquid (Coal Liquefaction) Gas (Coal gasification) Less pollution Toxic gases

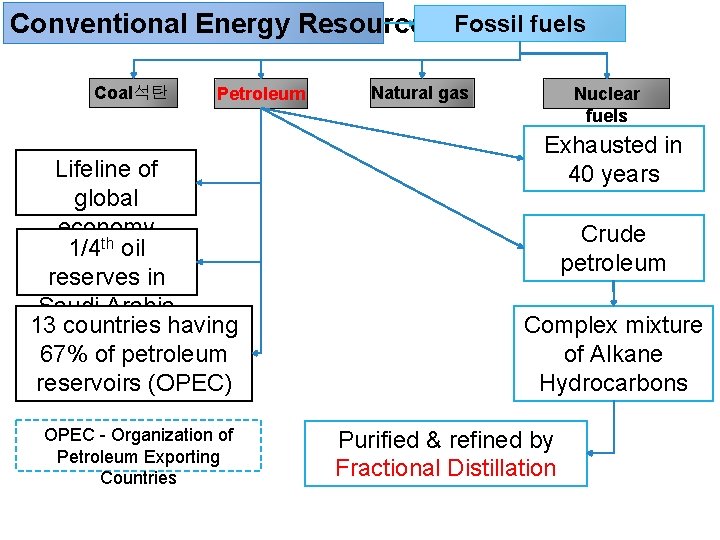
Conventional Energy Resources Fossil fuels Coal석탄 Petroleum Lifeline of global economy 1/4 th oil reserves in Saudi Arabia 13 countries having 67% of petroleum reservoirs (OPEC) OPEC - Organization of Petroleum Exporting Countries Natural gas Nuclear fuels Exhausted in 40 years Crude petroleum Complex mixture of Alkane Hydrocarbons Purified & refined by Fractional Distillation

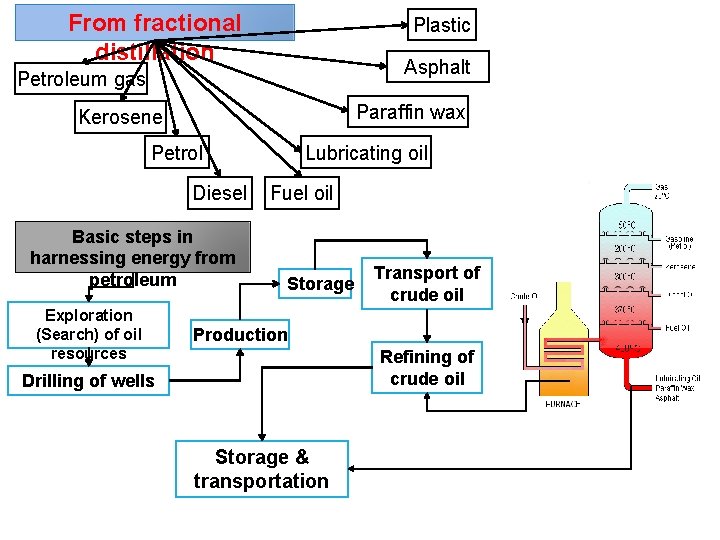
From fractional distillation Plastic Asphalt Petroleum gas Paraffin wax Kerosene Petrol Diesel Basic steps in harnessing energy from petroleum Exploration (Search) of oil resources Lubricating oil Fuel oil Storage Transport of crude oil Production Refining of crude oil Drilling of wells Storage & transportation

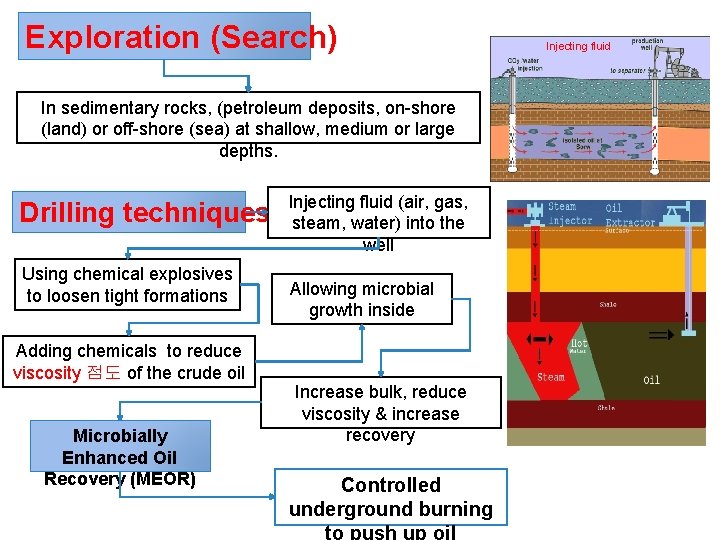
Exploration (Search) In sedimentary rocks, (petroleum deposits, on-shore (land) or off-shore (sea) at shallow, medium or large depths. Drilling techniques Using chemical explosives to loosen tight formations Adding chemicals to reduce viscosity 점도 of the crude oil Microbially Enhanced Oil Recovery (MEOR) Injecting fluid (air, gas, steam, water) into the well Allowing microbial growth inside Increase bulk, reduce viscosity & increase recovery Controlled underground burning to push up oil Injecting fluid

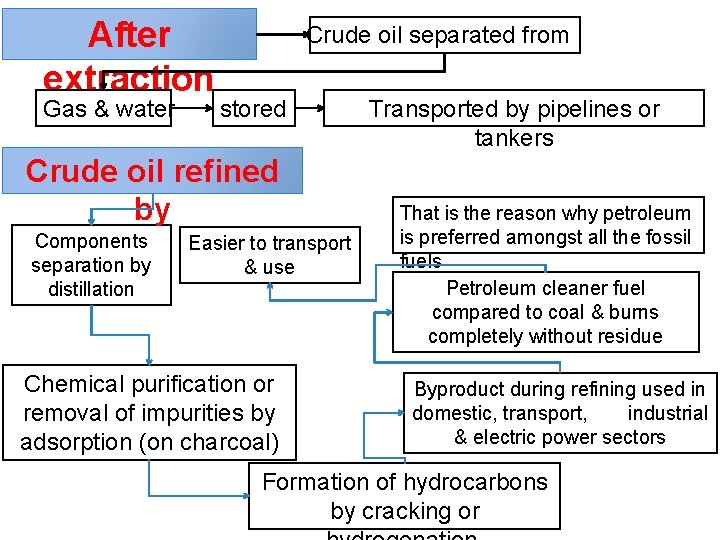
After extraction Gas & water Crude oil separated from stored Crude oil refined by Components separation by distillation Easier to transport & use Chemical purification or removal of impurities by adsorption (on charcoal) Transported by pipelines or tankers That is the reason why petroleum is preferred amongst all the fossil fuels Petroleum cleaner fuel compared to coal & burns completely without residue Byproduct during refining used in domestic, transport, industrial & electric power sectors Formation of hydrocarbons by cracking or

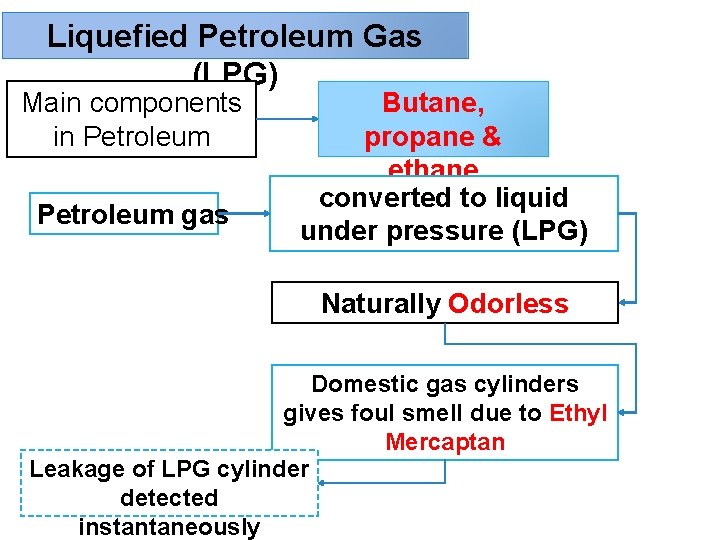
Liquefied Petroleum Gas (LPG) Main components in Petroleum gas Butane, propane & ethane converted to liquid under pressure (LPG) Naturally Odorless Domestic gas cylinders gives foul smell due to Ethyl Mercaptan Leakage of LPG cylinder detected instantaneously

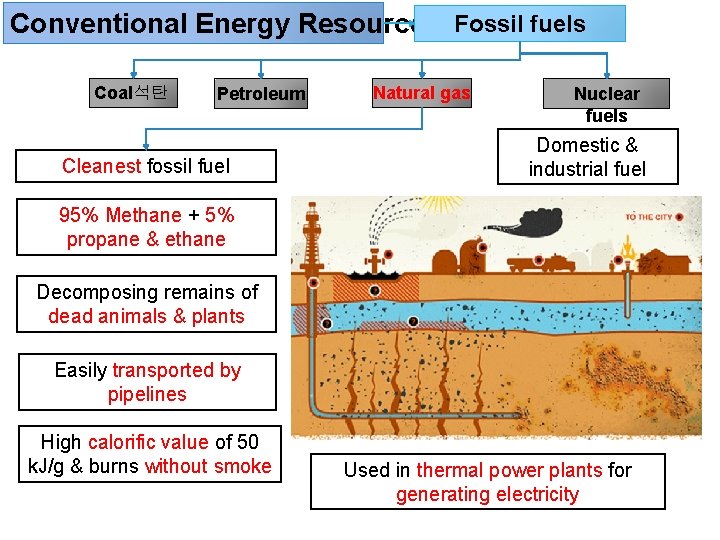
Conventional Energy Resources Fossil fuels Coal석탄 Petroleum Cleanest fossil fuel Natural gas Nuclear fuels Domestic & industrial fuel 95% Methane + 5% propane & ethane Decomposing remains of dead animals & plants Easily transported by pipelines High calorific value of 50 k. J/g & burns without smoke Used in thermal power plants for generating electricity

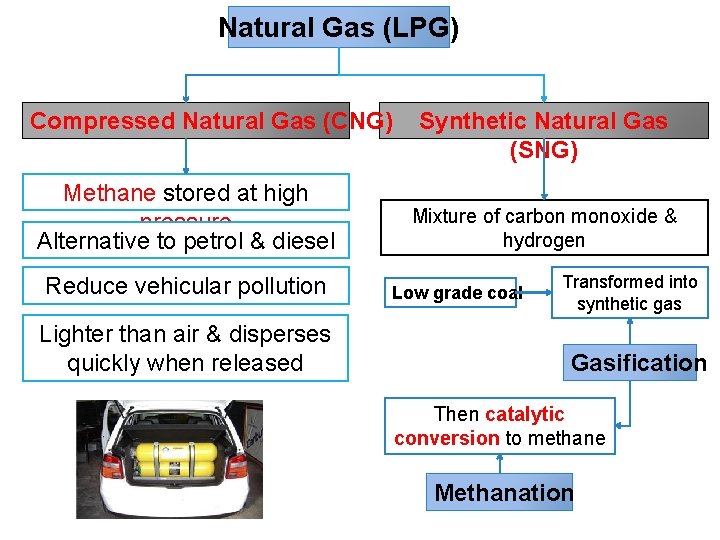
Natural Gas (LPG) Compressed Natural Gas (CNG) Methane stored at high pressure Alternative to petrol & diesel Reduce vehicular pollution Lighter than air & disperses quickly when released Synthetic Natural Gas (SNG) Mixture of carbon monoxide & hydrogen Low grade coal Transformed into synthetic gas Gasification Then catalytic conversion to methane Methanation

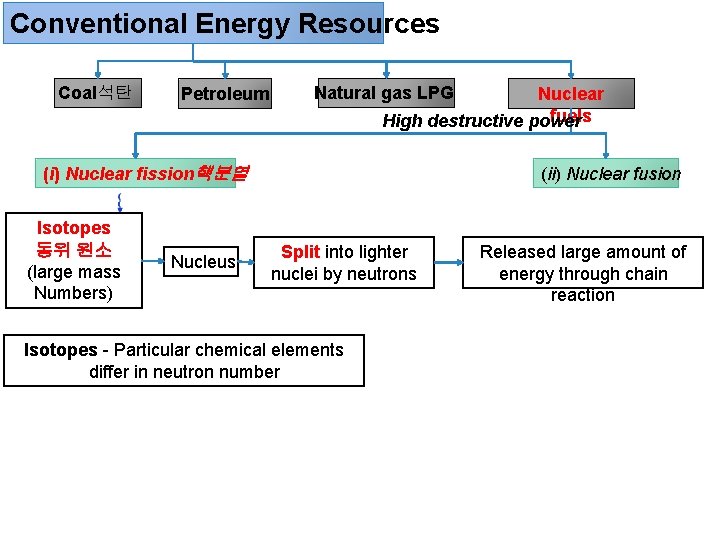
Conventional Energy Resources Coal석탄 Petroleum Natural gas LPG Nuclear fuels High destructive power (i) Nuclear fission핵분열 Isotopes 동위 원소 (large mass Numbers) Nucleus (ii) Nuclear fusion Split into lighter nuclei by neutrons Isotopes - Particular chemical elements differ in neutron number Released large amount of energy through chain reaction

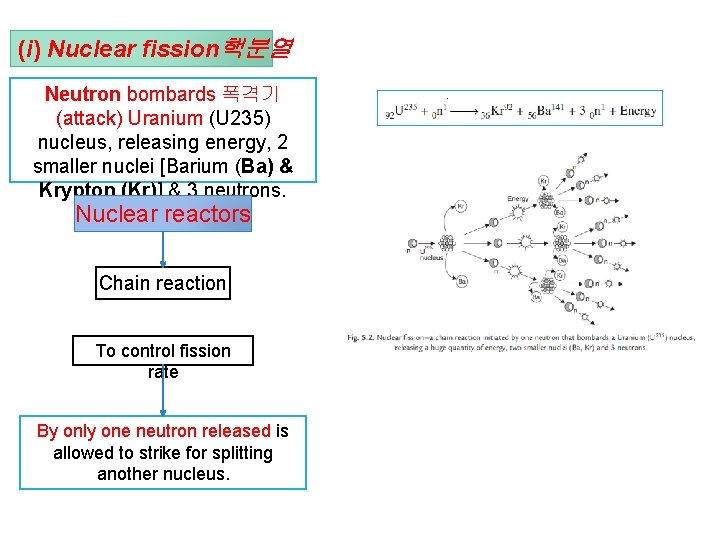
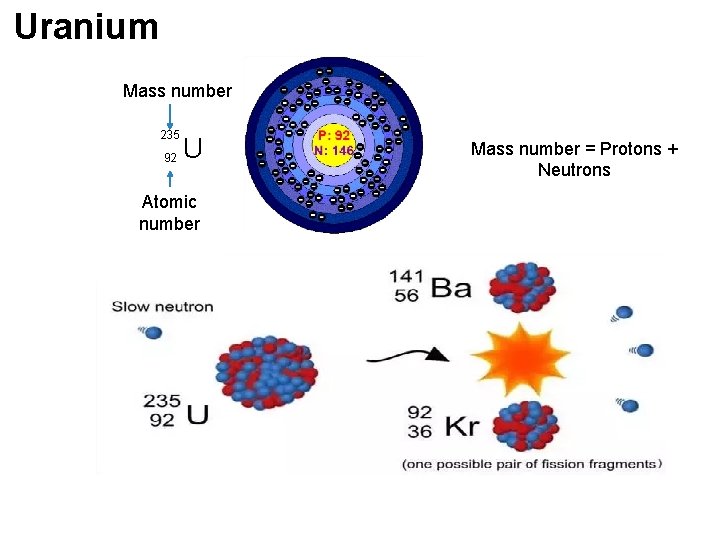
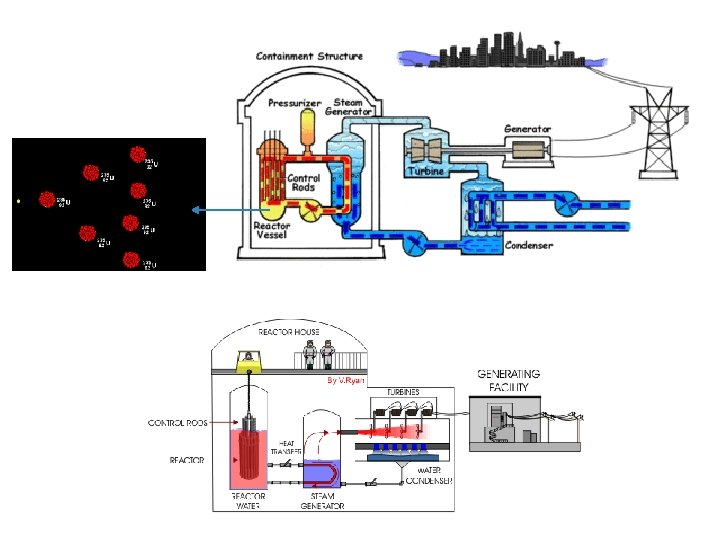
(i) Nuclear fission핵분열 Neutron bombards 폭격기 (attack) Uranium (U 235) nucleus, releasing energy, 2 smaller nuclei [Barium (Ba) & Krypton (Kr)] & 3 neutrons. Nuclear reactors Chain reaction To control fission rate By only one neutron released is allowed to strike for splitting another nucleus.

Uranium Mass number 235 92 U Atomic number Mass number = Protons + Neutrons


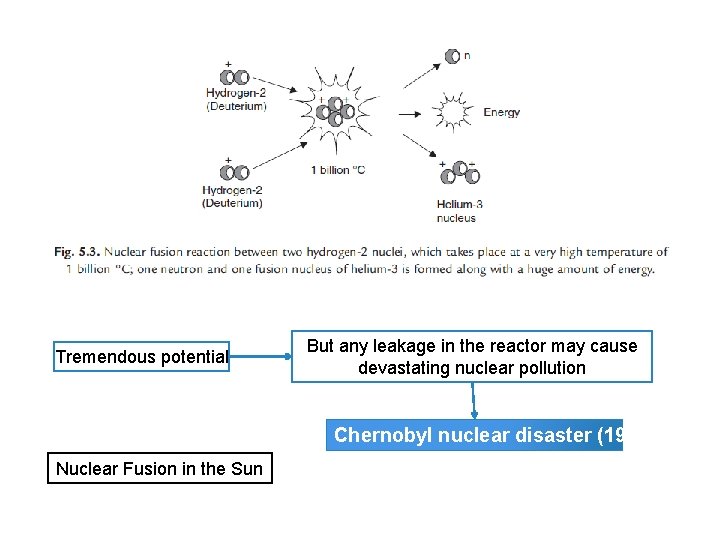
Conventional Energy Resources Coal석탄 Petroleum Natural gas LPG Nuclear fuels (ii) Nuclear fusion 핵융합 (i) Nuclear fission핵분열 Two light element isotopes (Hydrogen-2 (Deuterium) atoms Forced together at high temperatures (1 billion o. C) Releasing enormous energy Until they fuse to form heavier nucleus (Helium) Difficult to initiate the process but it releases more energy than nuclear fission Also nuclear fusion reaction between one H 2 (Deuterium) & One H 3 (Tritium) at 100 million o. C form heaver nucleus of Helium-4 + one neutron + Energy

Tremendous potential But any leakage in the reactor may cause devastating nuclear pollution Chernobyl nuclear disaster (1986) Nuclear Fusion in the Sun

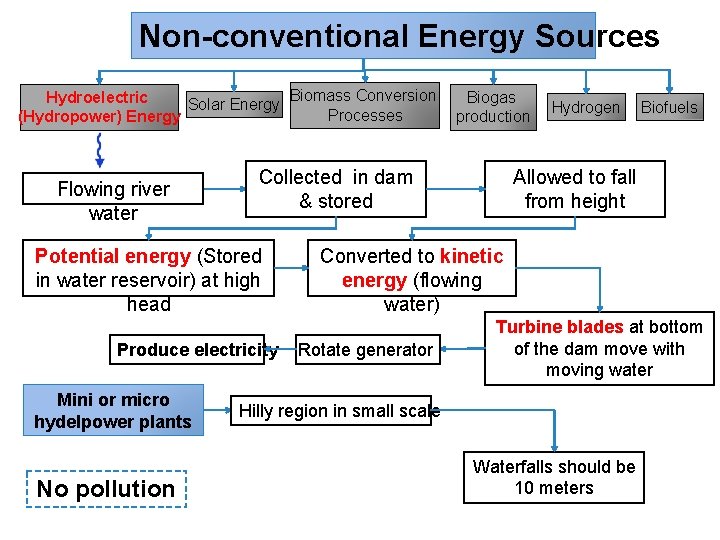
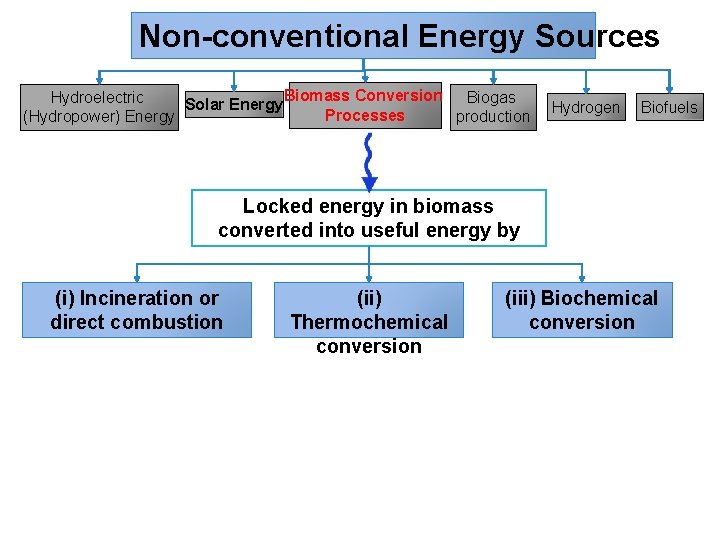
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Flowing river water Produce electricity Mini or micro hydelpower plants No pollution Biofuels Allowed to fall from height Collected in dam & stored Potential energy (Stored in water reservoir) at high head Hydrogen Converted to kinetic energy (flowing water) Rotate generator Turbine blades at bottom of the dam move with moving water Hilly region in small scale Waterfalls should be 10 meters

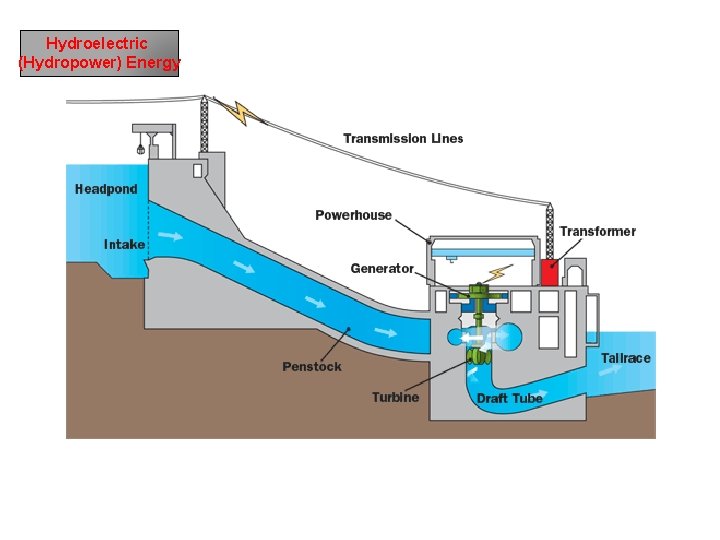
Hydroelectric (Hydropower) Energy

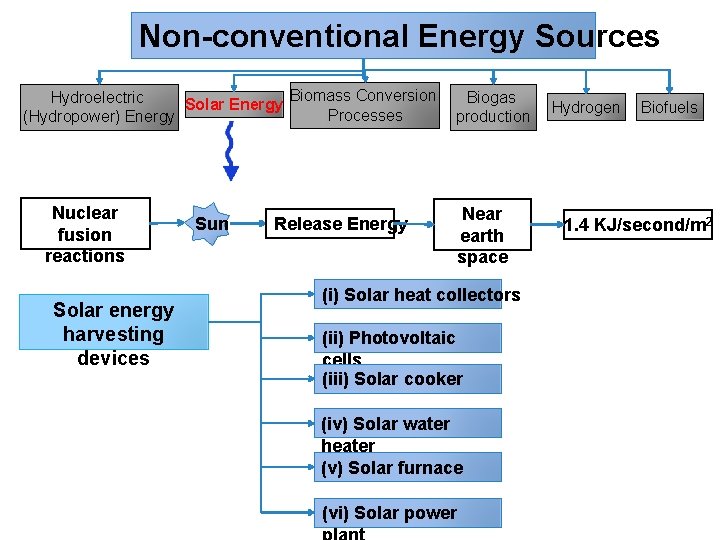
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Nuclear fusion reactions Solar energy harvesting devices Sun Release Energy Near earth space (i) Solar heat collectors (ii) Photovoltaic cells (iii) Solar cooker (iv) Solar water heater (v) Solar furnace (vi) Solar power Hydrogen Biofuels 1. 4 KJ/second/m 2

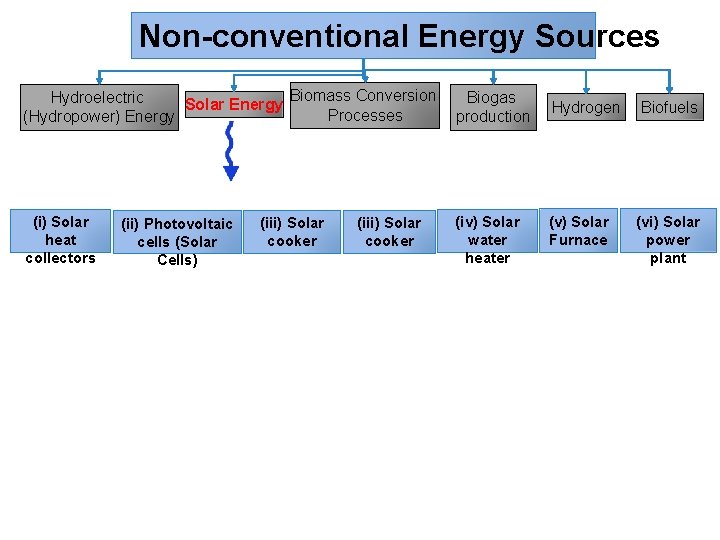
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production (i) Solar heat collectors (ii) Photovoltaic cells (Solar Cells) (iii) Solar cooker (iv) Solar water heater Hydrogen (v) Solar Furnace Biofuels (vi) Solar power plant

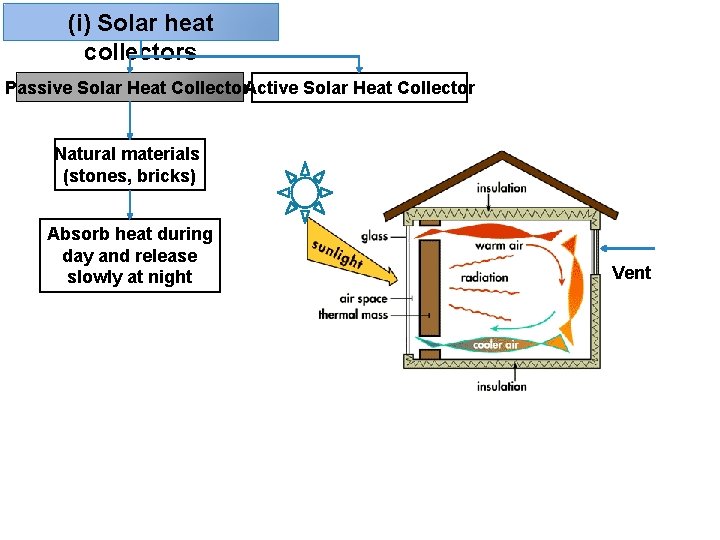
(i) Solar heat collectors Passive Solar Heat Collector. Active Solar Heat Collector Natural materials (stones, bricks) Absorb heat during day and release slowly at night Vent

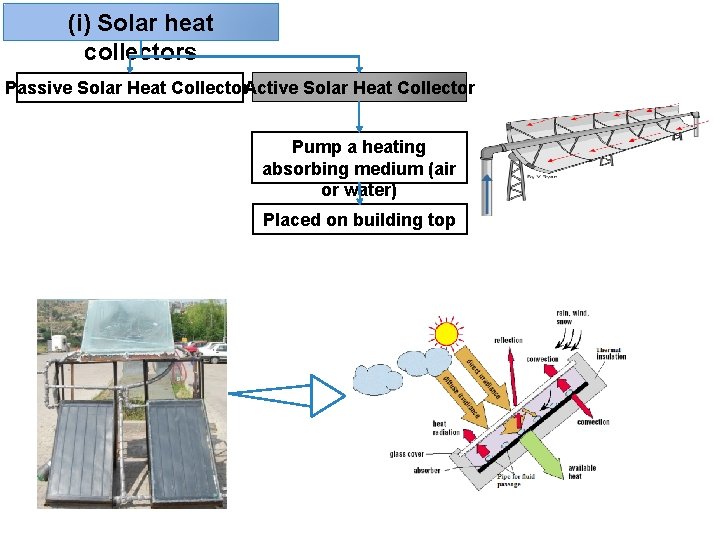
(i) Solar heat collectors Passive Solar Heat Collector. Active Solar Heat Collector Pump a heating absorbing medium (air or water) Placed on building top

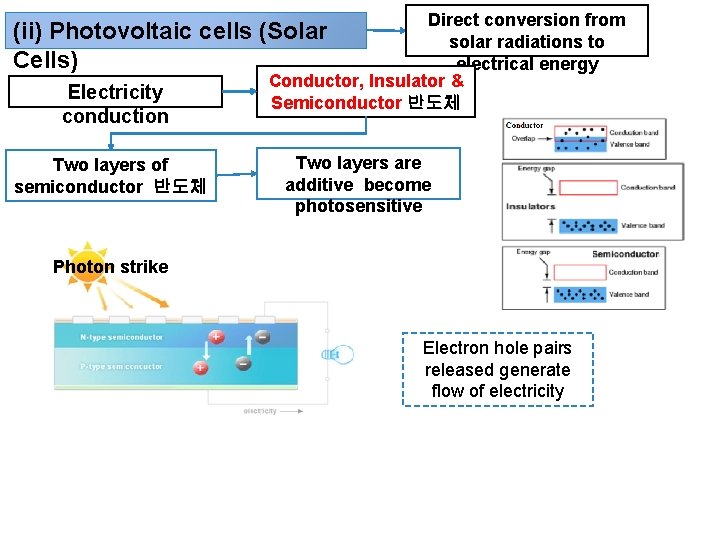
Direct conversion from solar radiations to electrical energy Conductor, Insulator & Semiconductor 반도체 (ii) Photovoltaic cells (Solar Cells) Electricity conduction Two layers of semiconductor 반도체 Two layers are additive become photosensitive Photon strike Electron hole pairs released generate flow of electricity

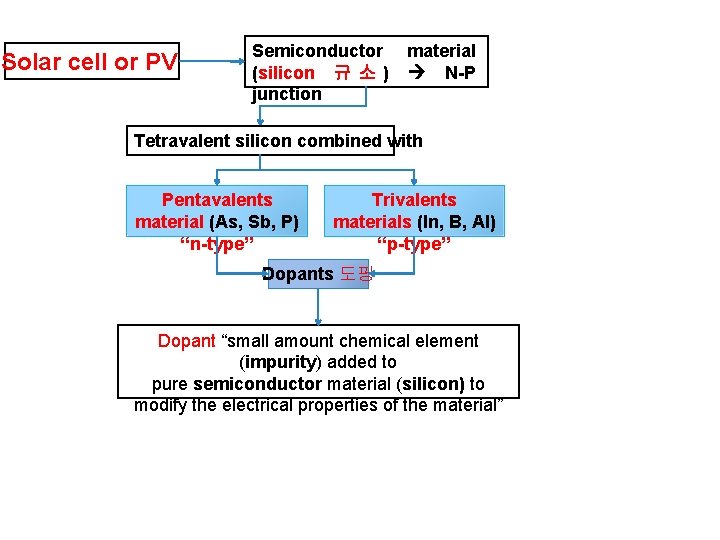
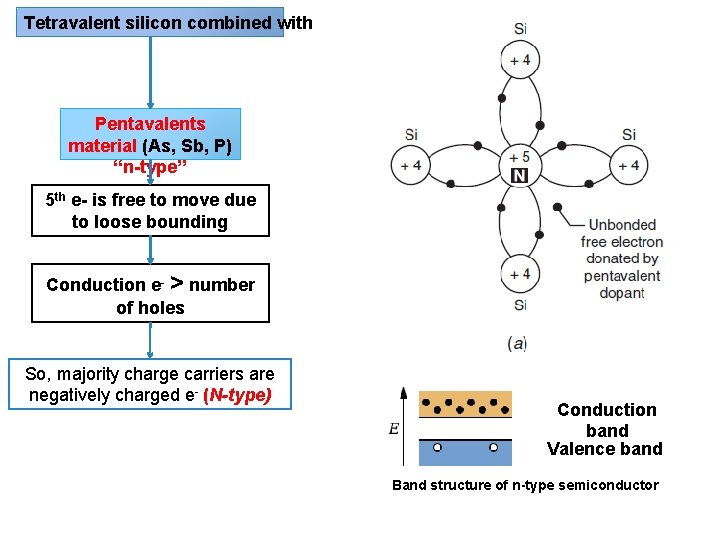
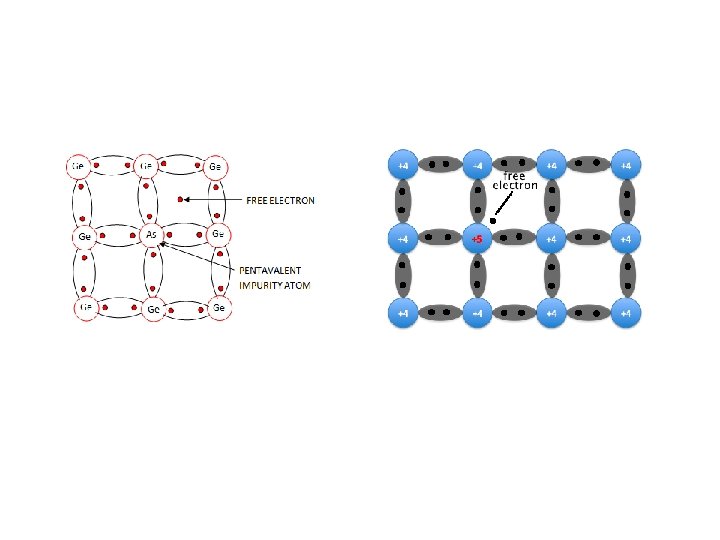
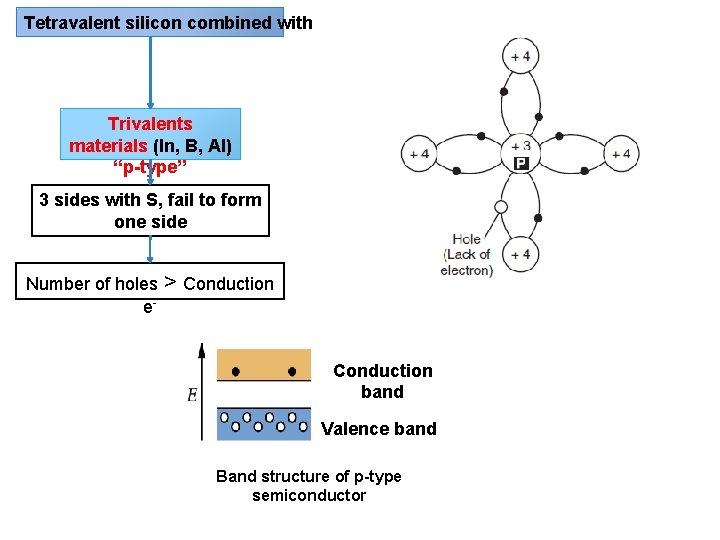
Solar cell or PV Semiconductor material (silicon 규 소 ) N-P junction Tetravalent silicon combined with Pentavalents material (As, Sb, P) “n-type” Trivalents materials (In, B, Al) “p-type” Dopants 도핑 Dopant “small amount chemical element (impurity) added to pure semiconductor material (silicon) to modify the electrical properties of the material”


Tetravalent silicon combined with Pentavalents material (As, Sb, P) “n-type” 5 th e- is free to move due to loose bounding Conduction e- > number of holes So, majority charge carriers are negatively charged e- (N-type) Conduction band Valence band Band structure of n-type semiconductor


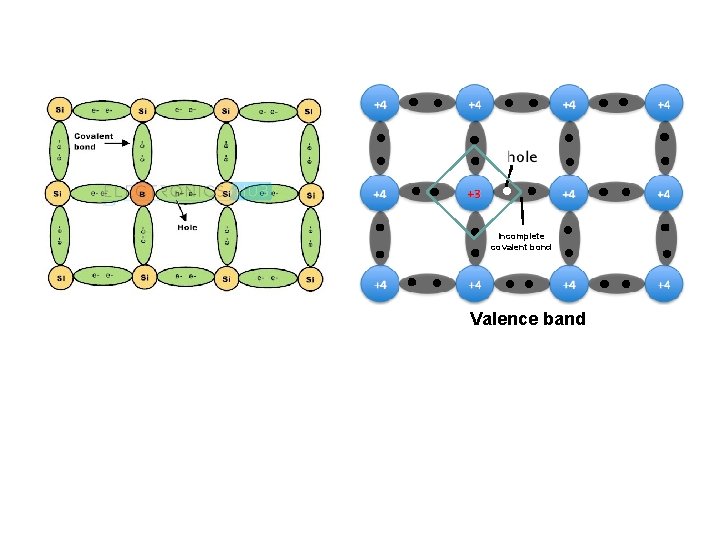
Tetravalent silicon combined with Trivalents materials (In, B, Al) “p-type” 3 sides with S, fail to form one side Number of holes > Conduction e. Conduction band Valence band Band structure of p-type semiconductor

Incomplete covalent bond Valence band

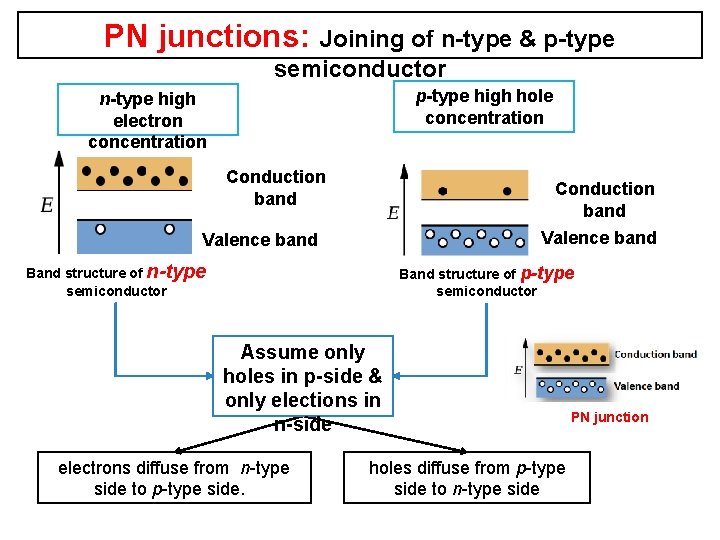
PN junctions: Joining of n-type & p-type semiconductor p-type high hole concentration n-type high electron concentration Conduction band Valence band Band structure of n-type semiconductor Band structure of p-type semiconductor Assume only holes in p-side & only elections in n-side electrons diffuse from n-type side to p-type side. PN junction holes diffuse from p-type side to n-type side

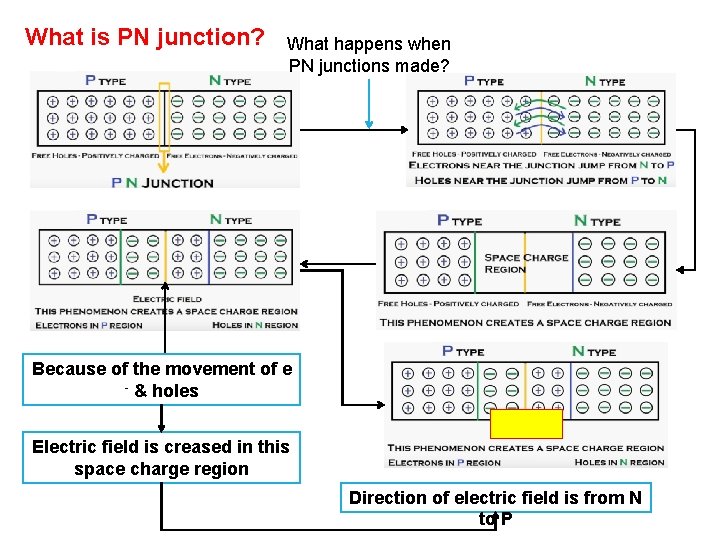
What is PN junction? What happens when PN junctions made? Because of the movement of e - & holes Electric field is creased in this space charge region Direction of electric field is from N to P

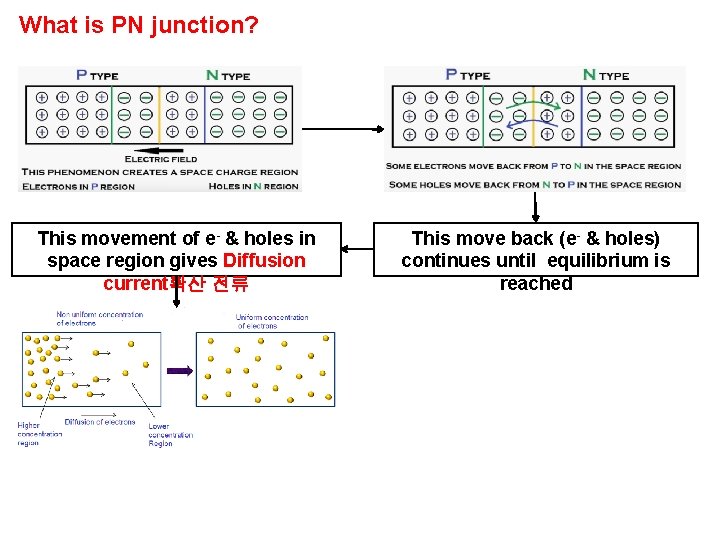
What is PN junction? This movement of e- & holes in space region gives Diffusion current확산 전류 This move back (e- & holes) continues until equilibrium is reached

http: //blog. naver. com/Post View. nhn? blog. Id=chylook& log. No=70097594996

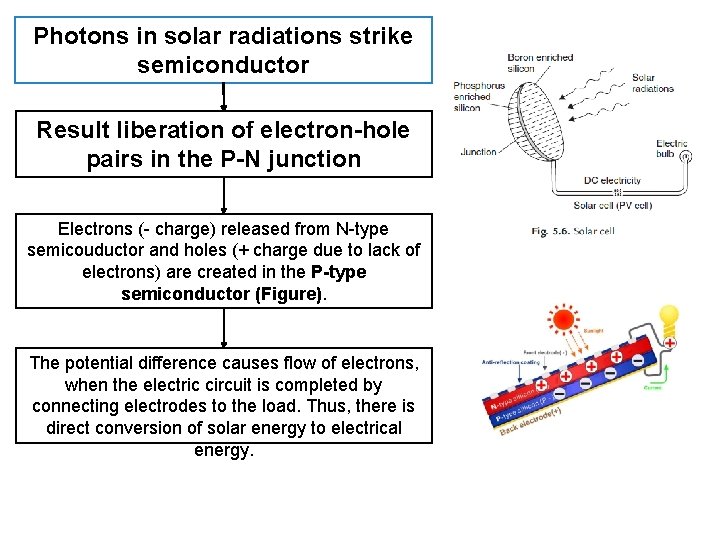
Photons in solar radiations strike semiconductor Result liberation of electron-hole pairs in the P-N junction Electrons (- charge) released from N-type semicouductor and holes (+ charge due to lack of electrons) are created in the P-type semiconductor (Figure). The potential difference causes flow of electrons, when the electric circuit is completed by connecting electrodes to the load. Thus, there is direct conversion of solar energy to electrical energy.

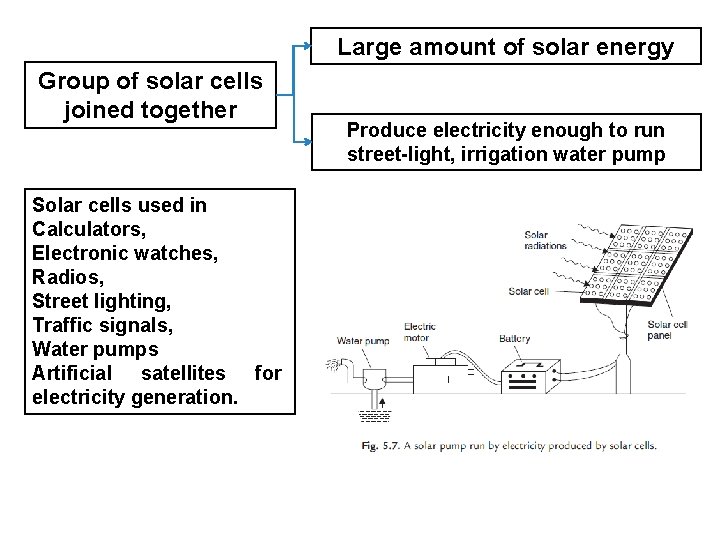
Large amount of solar energy Group of solar cells joined together Solar cells used in Calculators, Electronic watches, Radios, Street lighting, Traffic signals, Water pumps Artificial satellites for electricity generation. Produce electricity enough to run street-light, irrigation water pump

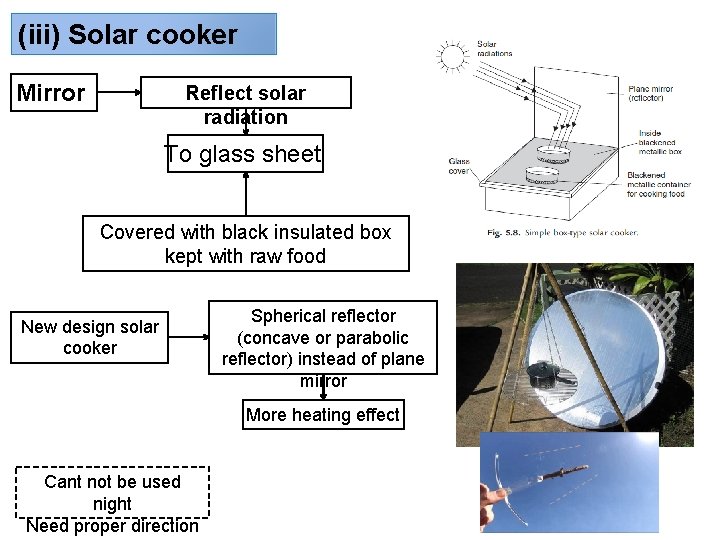
(iii) Solar cooker Mirror Reflect solar radiation To glass sheet Covered with black insulated box kept with raw food New design solar cooker Spherical reflector (concave or parabolic reflector) instead of plane mirror More heating effect Cant not be used night Need proper direction

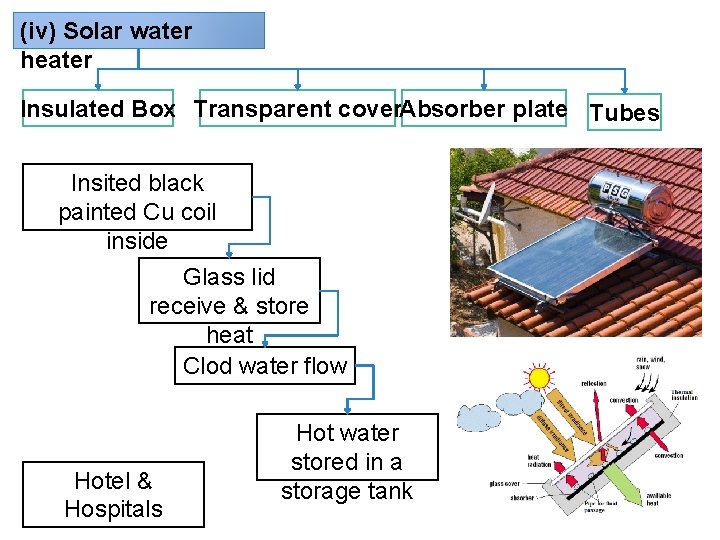
(iv) Solar water heater Insulated Box Transparent cover. Absorber plate Tubes Insited black painted Cu coil inside Glass lid receive & store heat Clod water flow Hotel & Hospitals Hot water stored in a storage tank

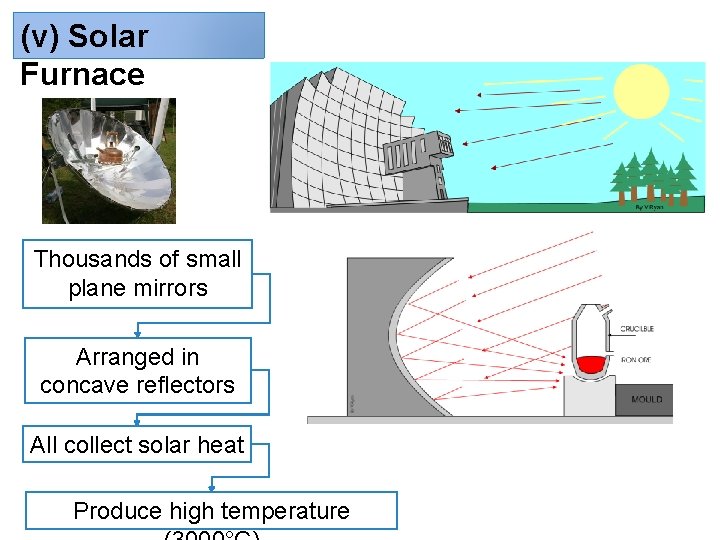
(v) Solar Furnace Thousands of small plane mirrors Arranged in concave reflectors All collect solar heat Produce high temperature

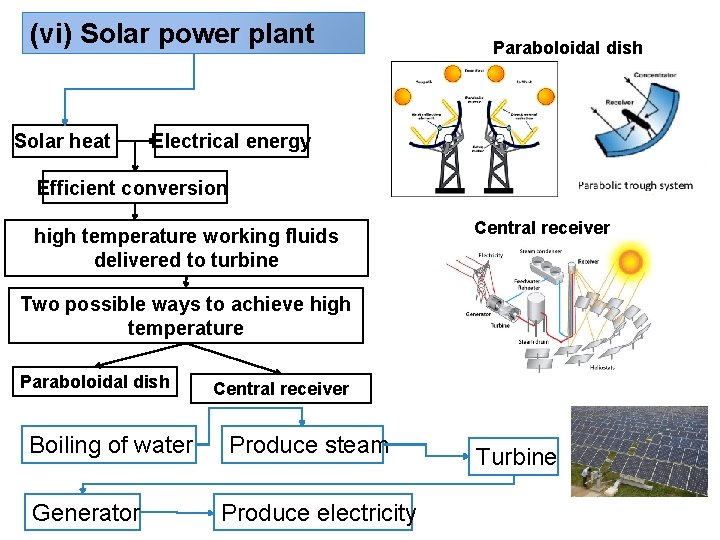
(vi) Solar power plant Solar heat Paraboloidal dish Electrical energy Efficient conversion high temperature working fluids delivered to turbine Central receiver Two possible ways to achieve high temperature Paraboloidal dish Boiling of water Generator Central receiver Produce steam Produce electricity Turbine

Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Hydrogen Biofuels Locked energy in biomass converted into useful energy by (i) Incineration or direct combustion (ii) Thermochemical conversion (iii) Biochemical conversion

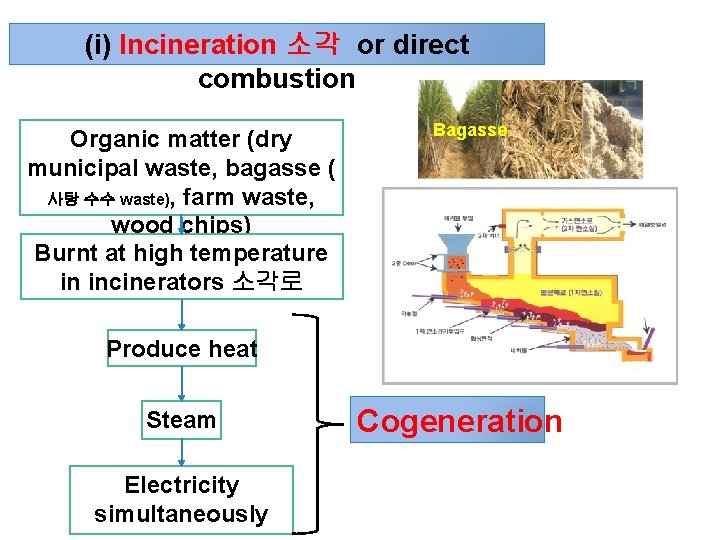
(i) Incineration 소각 or direct combustion Organic matter (dry municipal waste, bagasse ( 사탕 수수 waste), farm waste, wood chips) Burnt at high temperature in incinerators 소각로 Bagasse Produce heat Steam Electricity simultaneously Cogeneration

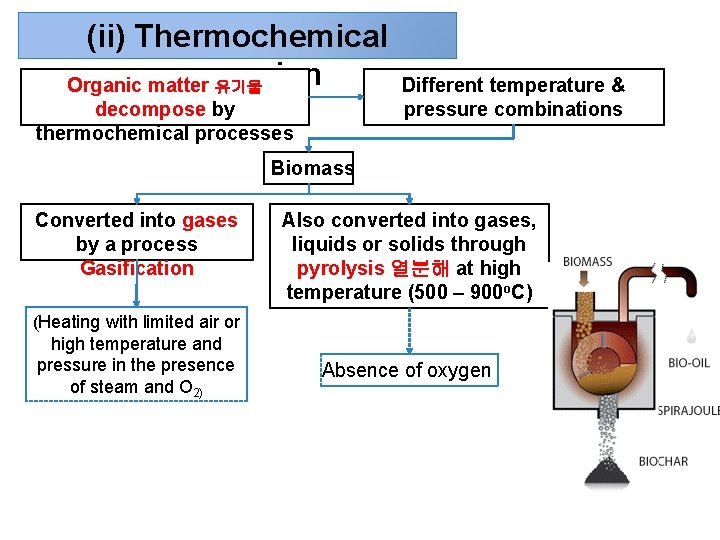
(ii) Thermochemical conversion Organic matter 유기물 decompose by thermochemical processes Different temperature & pressure combinations Biomass Converted into gases by a process Gasification (Heating with limited air or high temperature and pressure in the presence of steam and O 2) Also converted into gases, liquids or solids through pyrolysis 열분해 at high temperature (500 – 900 o. C) Absence of oxygen

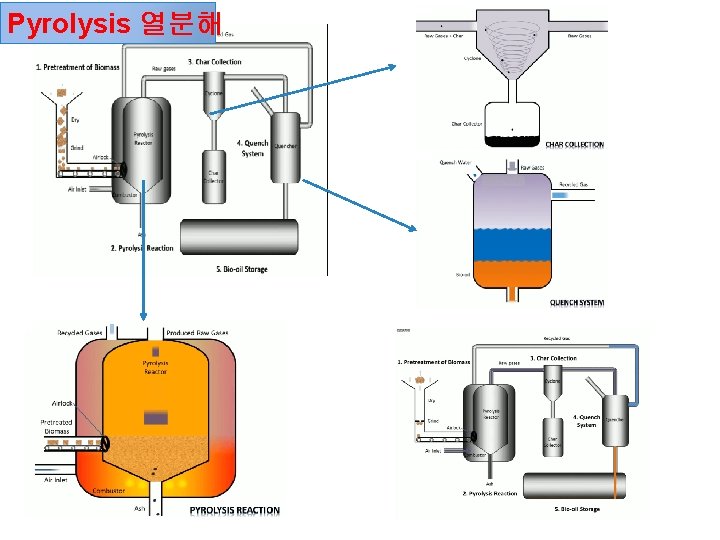
Pyrolysis 열분해

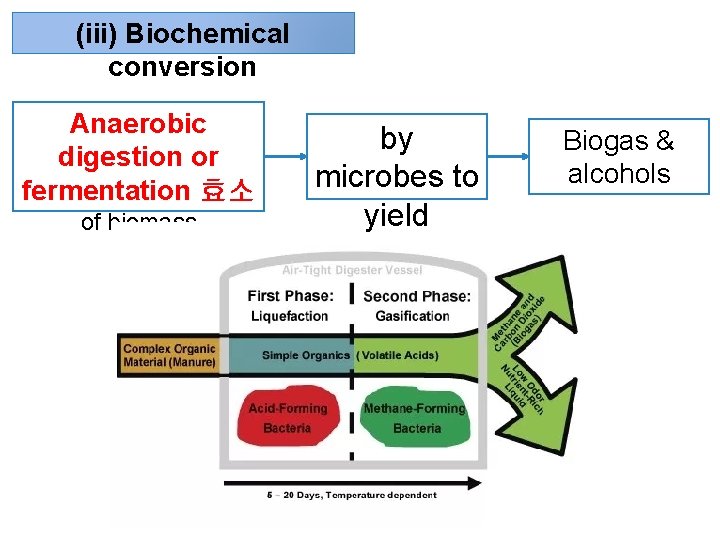
(iii) Biochemical conversion Anaerobic digestion or fermentation 효소 of biomass by microbes to yield Biogas & alcohols

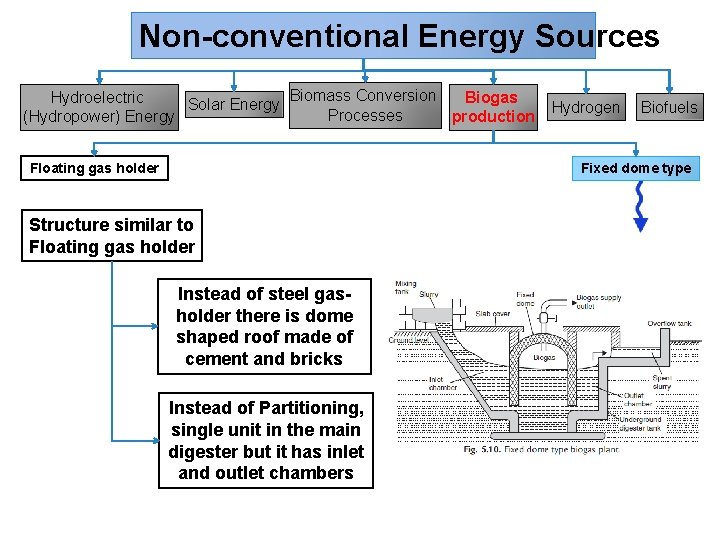
Non-conventional Energy Sources Biomass Conversion Biogas Hydroelectric Solar Energy Processes (Hydropower) Energy production Floating gas holder Well-shaped digester tank Partitioned tank wall Biofuels Fixed dome type Placed underground & made up of bricks Gas holder controlled by a pipe Hydrogen Steel drum floats to hold the bio-gas produced Gas outlet regulated by valve One side receives dung-water mixture by pipe other side discharge slurry through outlet pipe

Non-conventional Energy Sources Biomass Conversion Biogas Hydroelectric Solar Energy Processes (Hydropower) Energy production Floating gas holder Hydrogen Biofuels Fixed dome type Structure similar to Floating gas holder Instead of steel gasholder there is dome shaped roof made of cement and bricks Instead of Partitioning, single unit in the main digester but it has inlet and outlet chambers

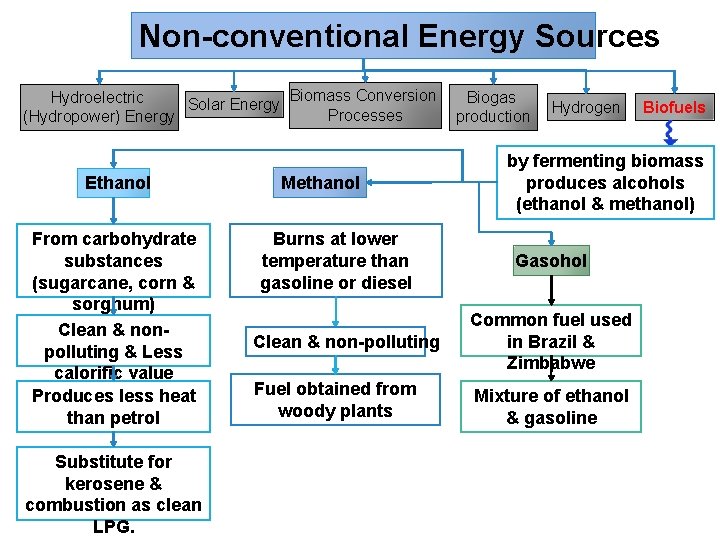
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Ethanol From carbohydrate substances (sugarcane, corn & sorghum) Clean & nonpolluting & Less calorific value Produces less heat than petrol Substitute for kerosene & combustion as clean LPG. Methanol Burns at lower temperature than gasoline or diesel Hydrogen Biofuels by fermenting biomass produces alcohols (ethanol & methanol) Gasohol Clean & non-polluting Common fuel used in Brazil & Zimbabwe Fuel obtained from woody plants Mixture of ethanol & gasoline

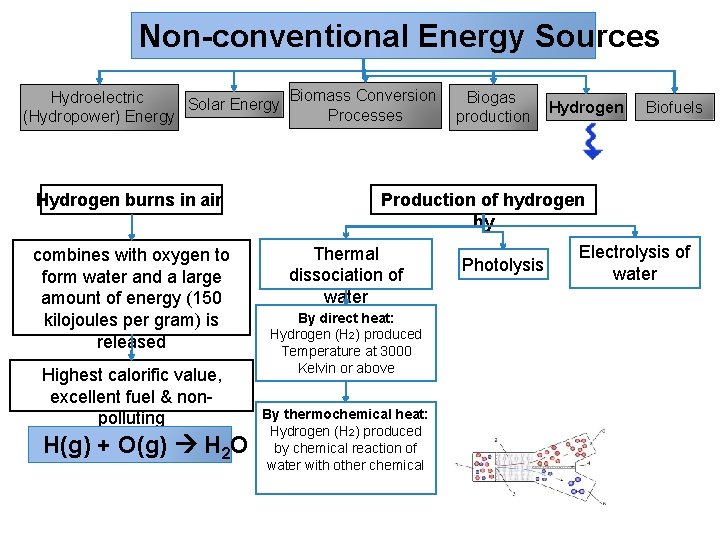
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Hydrogen burns in air combines with oxygen to form water and a large amount of energy (150 kilojoules per gram) is released Highest calorific value, excellent fuel & nonpolluting H(g) + O(g) H 2 O Hydrogen Biofuels Production of hydrogen by Thermal dissociation of water By direct heat: Hydrogen (H 2) produced Temperature at 3000 Kelvin or above By thermochemical heat: Hydrogen (H 2) produced by chemical reaction of water with other chemical Photolysis Electrolysis of water

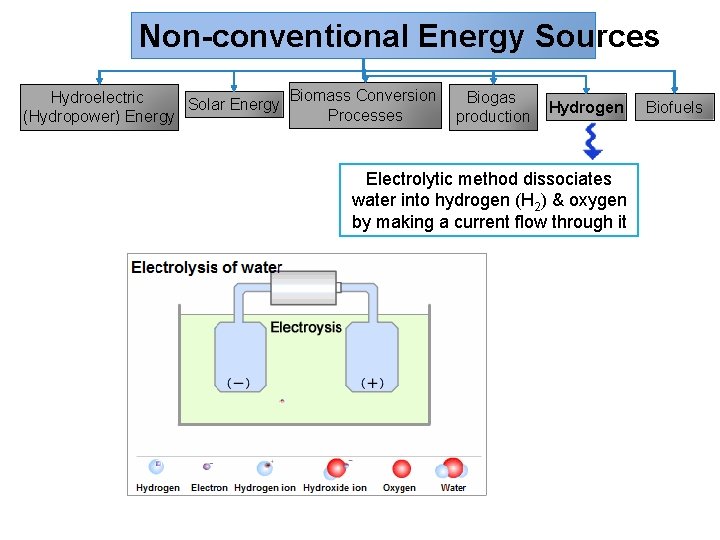
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Hydrogen Electrolytic method dissociates water into hydrogen (H 2) & oxygen by making a current flow through it Biofuels

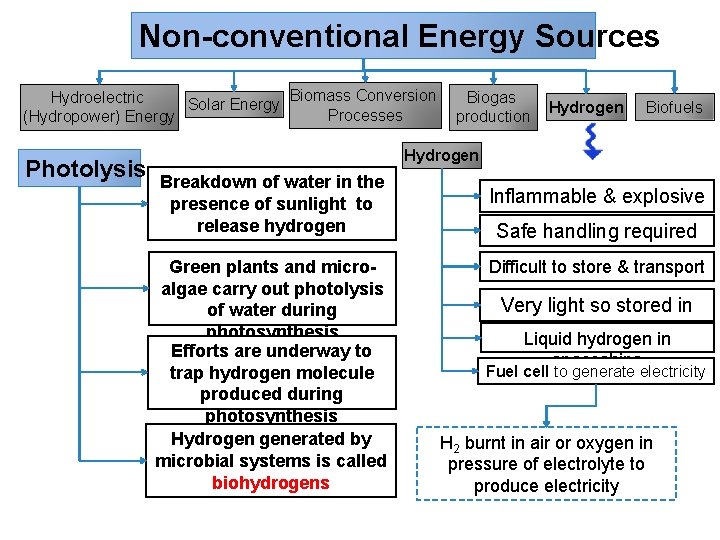
Non-conventional Energy Sources Biomass Conversion Hydroelectric Biogas Solar Energy Processes (Hydropower) Energy production Photolysis Hydrogen Biofuels Hydrogen Breakdown of water in the presence of sunlight to release hydrogen Green plants and microalgae carry out photolysis of water during photosynthesis Efforts are underway to trap hydrogen molecule produced during photosynthesis Hydrogen generated by microbial systems is called biohydrogens Inflammable & explosive Safe handling required Difficult to store & transport Very light so stored in bulk Liquid hydrogen in spaceships Fuel cell to generate electricity H 2 burnt in air or oxygen in pressure of electrolyte to produce electricity