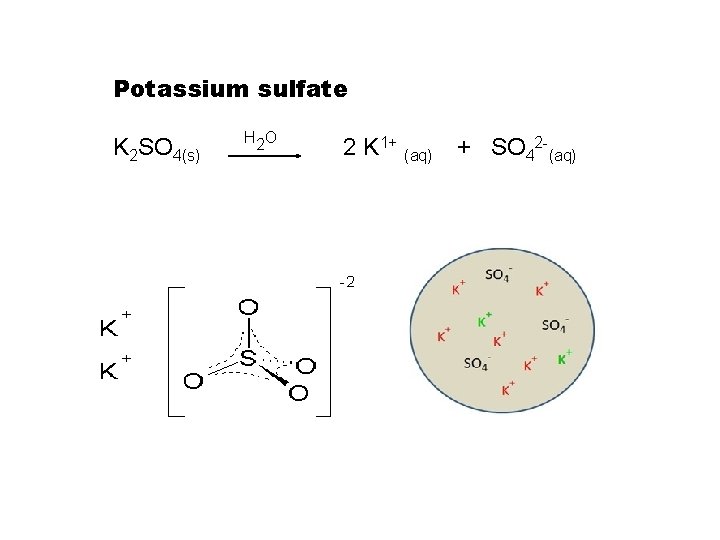
Dissolution of SALTS Potassium sulfate K 2 SO













- Slides: 17

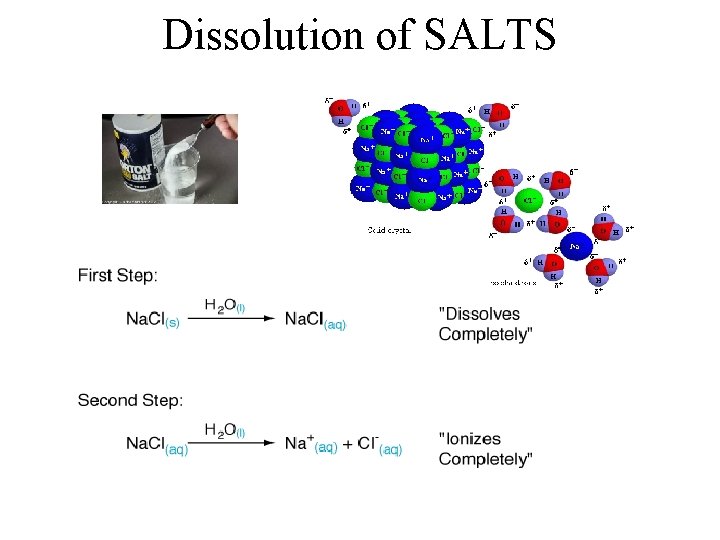
Dissolution of SALTS

Potassium sulfate K 2 SO 4(s) H 2 O 2 K 1+ (aq) + SO 42 -(aq)


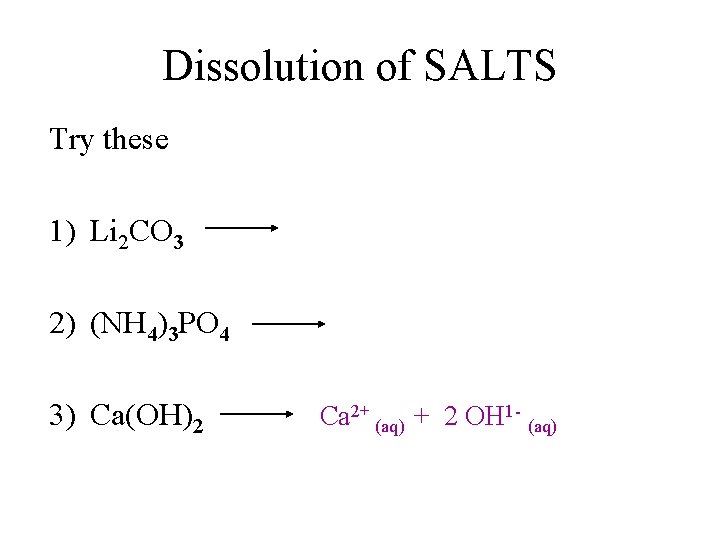
Dissolution of SALTS Try these 1) Li 2 CO 3 2) (NH 4)3 PO 4 3) Ca(OH)2 Ca 2+ (aq) + 2 OH 1 - (aq)

Molarity Calculations 60. 0 grams of Na. OH is dissolved in enough distilled water to make 2. 00 L of solution. What is the molarity of the solution? Molar mass of Na. OH = Moles of Na. OH present = Molarity = moles of solute liters of solution M= M= moles of Na. OH L of H 20

138 grams of copper(II) sulfate in 750 m. L of solution has what concentration? 138 g Cu. SO 4 1 mole of Cu. SO 4 = 0. 8625 moles Cu. SO 4 160 g Molarity = ? moles ? liters 750 m. L 1 L = 0. 750 L 1000 m. L Molarity = 0. 8625 moles = 1. 15 M Cu. SO 4 0. 750 L

You want to make 1. 50 L of 4. 00 M silver nitrate solution. How many grams of silver nitrate are needed? 2

If you want 250 m. L of a 0. 08 M concentrate solution of barium hydroxide, how many grams of barium hydroxide is needed?

DILUTION You need to make 1. 00 L of a 2. 00 M solution of HCl. You only have a 10. 0 M solution of HCl available. How much 10. 0 M HCl do you need to make 1. 00 L of a 2. 00 M solution? various concentrations of hydrochloric acid

How much 18 M sulfuric acid is needed to make 1250 m. L of a 4. 5 molarity solution? For DILUTION (concentration 1)(volume 1) = (concentration 2)(volume 2) (18 M) (V) = (4. 5 M) (1250 m. L) divide by 18 M V = 312. 5 m. L of 18 M sulfuric acid

If 61 m. L of 8 M hydrobromic acid is mixed with 689 m. L of water, what is the concentration of the resulting solution?

Dissolution Concentration Al 2(SO 4)3 H 2 O 2 Al 3+ + 3 SO 4 2 - What is the sulfate ion concentration in this 0. 15 M aluminum sulfate solution?

Dissolution Concentration What are the concentrations of the two ions that are formed from a 2. 31 M calcium acetate solution?

Concentration of a Gas If the maximum concentration of krypton gas in water is 0. 00089 M at 404 K and 1250 mm. Hg, what volume of gas can potentially be removed from 20 liters of solution?

If the maximum concentration of carbon dioxide gas in water is 0. 0284 M at 0 o. C and 760 mm Hg, what volume of this gas could possibly be removed from 2. 00 liters of this solution? 0. 0284 moles CO 2 = x 1 L solution 2 L solution PV =n. RT (760 mm. Hg) V = 0. 0568 moles(62. 4)(273 K) V = 1. 27 L of CO 2 x = 0. 0568 moles CO 2

Molarity with a Chemical Reaction How many milliliters of 18. 0 M H 2 SO 4 are required to react with 250 m. L of 2. 5 M Al(OH)3 to create aluminum sulfate and water? What do we need to solve this problem?

Molarity with a Chemical Reaction What volume of a 8. 00 M HBr solution is needed to completely react 600 m. L of a 2. 00 M Ca(OH)2 solution? Ca(OH)2 + 2 HBr 2 H 2 O + Ca. Br 2 2. 00 moles Ca(OH)2 = x moles Ca(OH)2 x = 1. 20 moles Ca(OH)2 1 L 0. 600 L 1. 20 moles Ca(OH)2 2 moles HBr 1 L solution = 0. 30 L of 8 M HBr 1 mole Ca(OH)2 8. 00 moles HBr