Dissolution of calcite in sediments metabolic dissolution Ca

Dissolution of calcite in sediments -metabolic dissolution

Ca. CO 3 accumulation in marine sediments • Ca. CO 3 is the predominant biogenic sediment constituent over much of the ocean • Changes in the marine alkalinity budget • can drive changes in atmospheric PCO 2 • involve changes in the accumulation rate of Ca. CO 3 in marine sediment ==> The study of climate change is linked to the study of Ca. CO 3 in marine sediments

Ca. CO 3 dissolution ~ 50% of calcite dissolution in the ocean occurs at the sea floor Up to 50% of seafloor calcite dissolution may occur in sediments that lie above the calcite saturation horizon … what drives this dissolution?

“Metabolic” calcite dissolution Oxic respiration results in the release of acids to solution : Acids are neutralized by (and similar reactions for neutralizing H+)

Sediments vs water column In the water column, A very large reservoir of dissolved CO 32 -, B(OH)4… result of respiration: lower p. H and CO 32 - in deep water over its ~ 1000 yr trip from NAtl to NEPac How are sediments different? spatial scale… 8 cm mixed layer, w~1 cm/ky… res time ~ 8000 y supply of dissolved CO 32 -, B(OH)4 - limited by diffusion from BW Abundant Ca. CO 3 (s) in sediments ==> Dissolution is favored in sediments if pore waters are driven to undersaturation

Theoretical consideration of metabolic dissolution
![Predicted [CO 32 -] vs. depth General… A specific case, supersaturated bottom water… Predicted [CO 32 -] vs. depth General… A specific case, supersaturated bottom water…](http://slidetodoc.com/presentation_image/a574ed1d6cd540a29dd66390b587c281/image-7.jpg)
Predicted [CO 32 -] vs. depth General… A specific case, supersaturated bottom water…
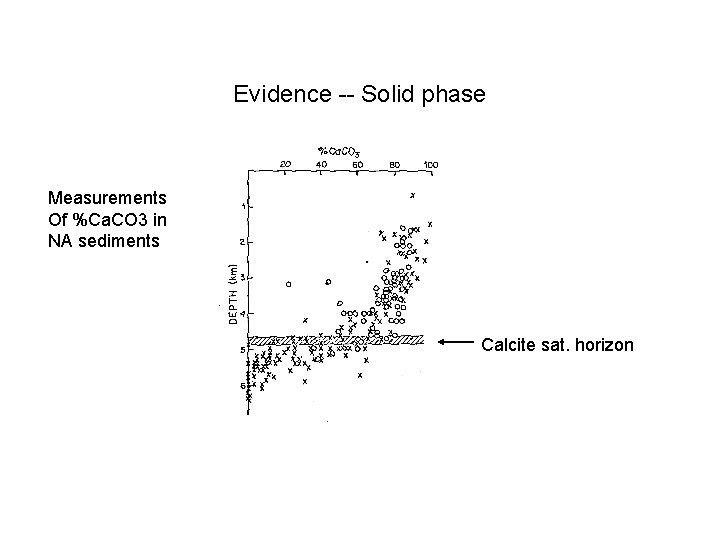
Evidence -- Solid phase Measurements Of %Ca. CO 3 in NA sediments Calcite sat. horizon

Evidence -- Pore water Organic matter oxidation by O 2: With no dissolution: With dissolution, add to this reaction: Now:
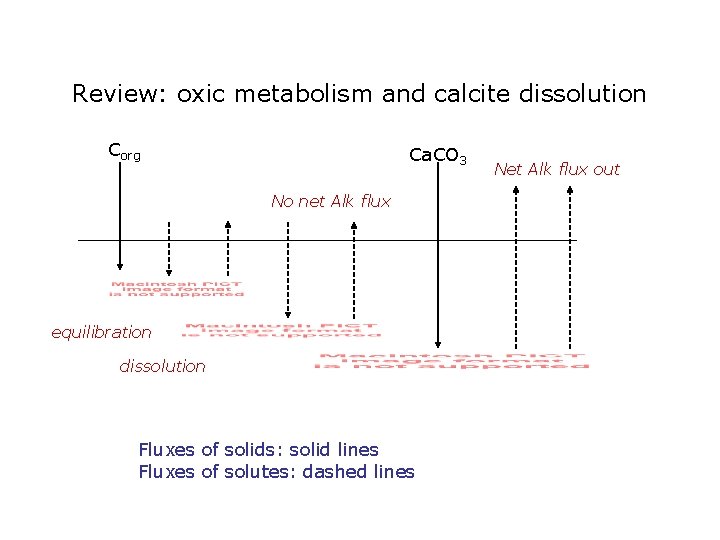
Review: oxic metabolism and calcite dissolution Corg Ca. CO 3 No net Alk flux equilibration dissolution Fluxes of solids: solid lines Fluxes of solutes: dashed lines Net Alk flux out
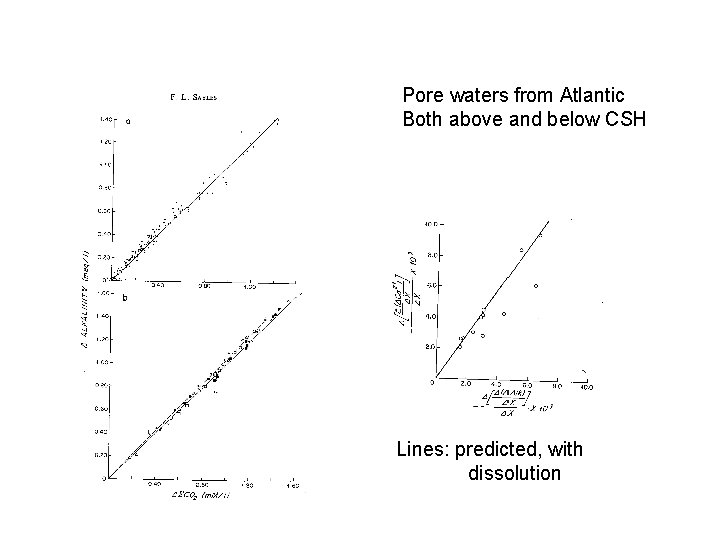
Pore waters from Atlantic Both above and below CSH Lines: predicted, with dissolution

Evidence - Pore water In situ microelectrode profiling ** measure pore water p. H and O 2 at high resolution ** interpret profiles using a model of coupled oxic metabolism / calcite dissolution ** the combination allows (1) qualitative demonstration of metabolic dissolution (2) quantification of dissolution rate

Metabolic dissolution : models For each solute: Assume: acid-base equilibrium in solution ! For Ca. CO 3: For mass: Where: Assume: no calcite precipitation !

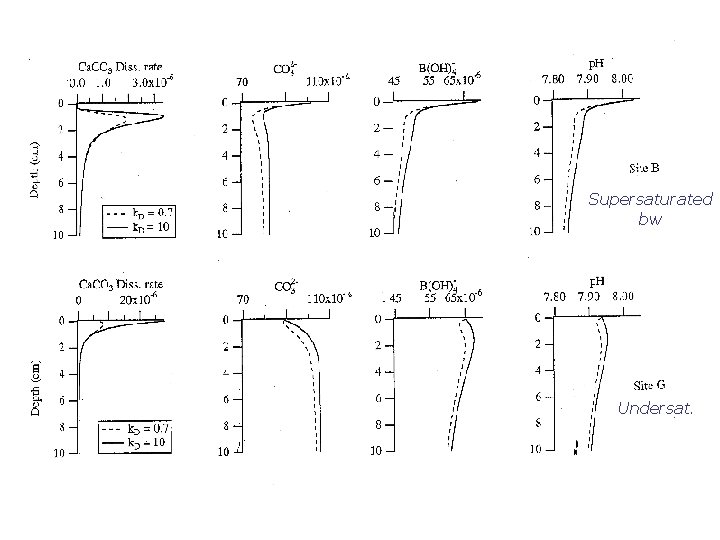
Supersaturated bw Undersat.
In situ MEP Archer et al. (1989) GCA 53, 2831 -2845 Data from: Station 12 -- above CSH Station 13 -- below CSH
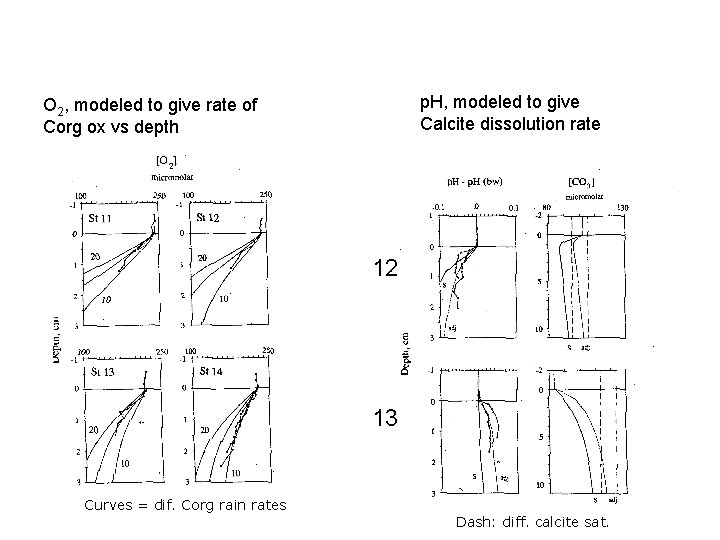
p. H, modeled to give Calcite dissolution rate O 2, modeled to give rate of Corg ox vs depth 12 13 Curves = dif. Corg rain rates Dash: diff. calcite sat.
In situ MEP North Atlantic Hales et al. , 1994, DSR 41, 695 -719 From 2 sites: 5 -- 2159 m 7 -- 4236 m
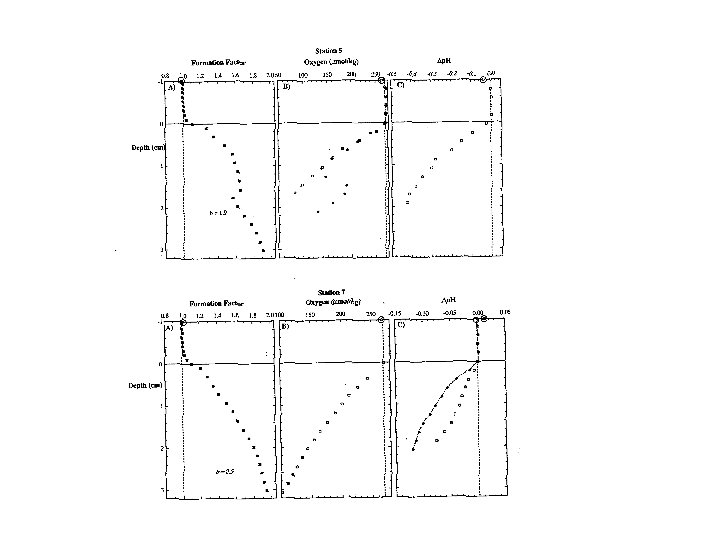
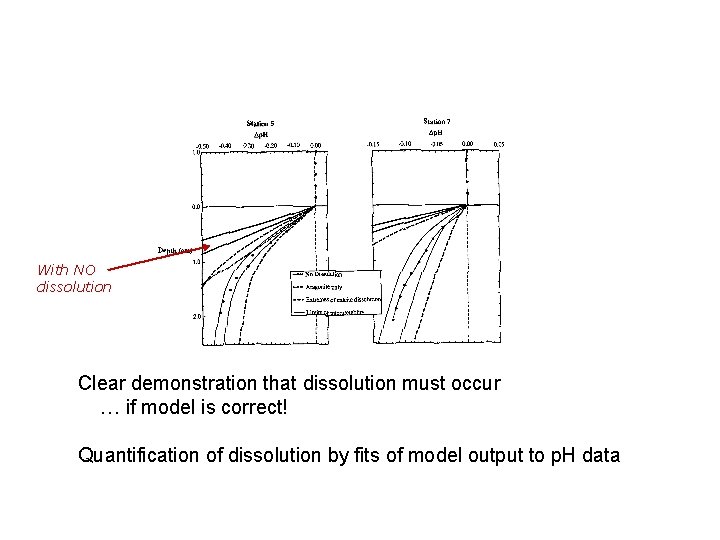
With NO dissolution Clear demonstration that dissolution must occur … if model is correct! Quantification of dissolution by fits of model output to p. H data

Evidence -- Pore water in situ whole-core squeezing Martin and Sayles, 1996, GCA 60, 243 -263 In situ measurement of pore water TCO 2, Alk, Ca 2+ … demonstration of dissolution without model Either shipboard or in situ measurement of NO 3 -, shipboard O 2 … quantification of ox. vs depth Model of coupled Corg ox. and calcite dissolution to quantify dissolution rate
Ceara Rise, w. tropical Atlantic Site A: just above CSH
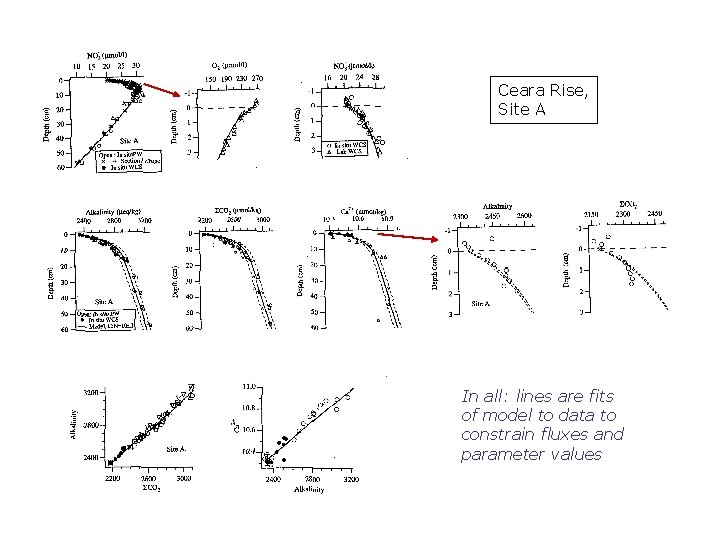
Ceara Rise, Site A In all: lines are fits of model to data to constrain fluxes and parameter values
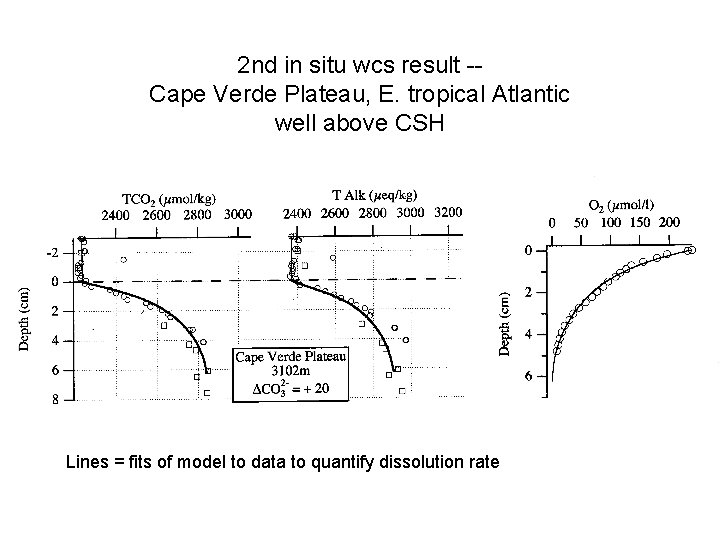
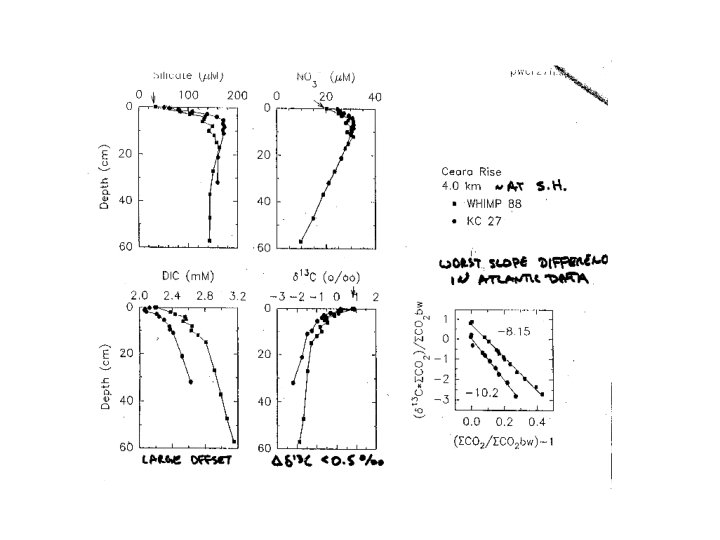
2 nd in situ wcs result -Cape Verde Plateau, E. tropical Atlantic well above CSH Lines = fits of model to data to quantify dissolution rate
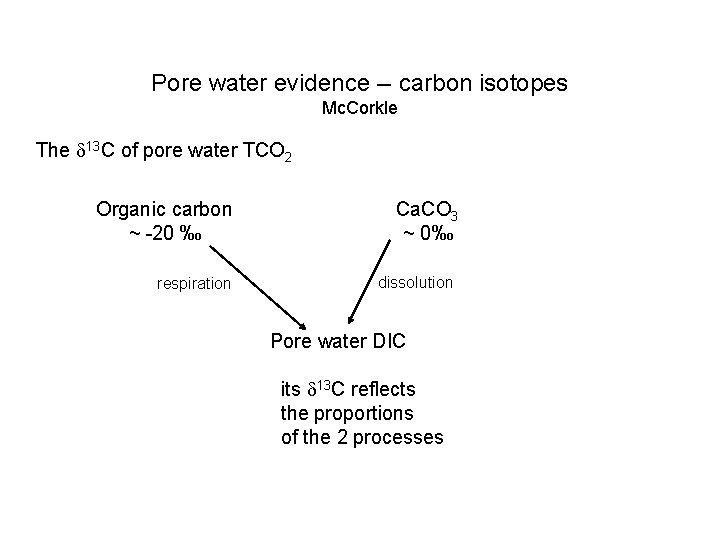
Pore water evidence -- carbon isotopes Mc. Corkle The 13 C of pore water TCO 2 Organic carbon ~ -20 ‰ respiration Ca. CO 3 ~ 0‰ dissolution Pore water DIC its 13 C reflects the proportions of the 2 processes
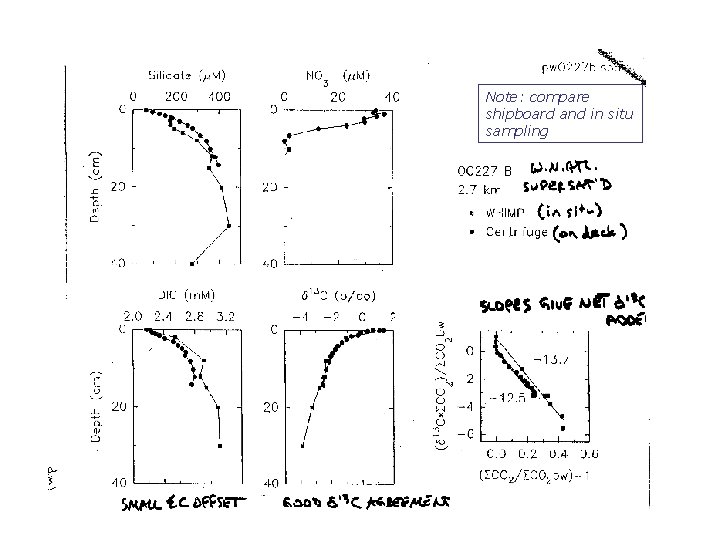
Note: compare shipboard and in situ sampling
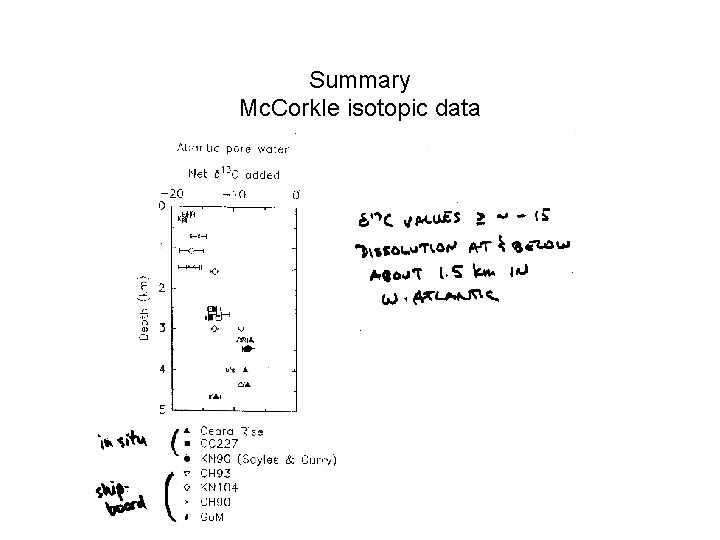
Summary Mc. Corkle isotopic data

Summary Pore water p. H, Alk, TCO 2 data
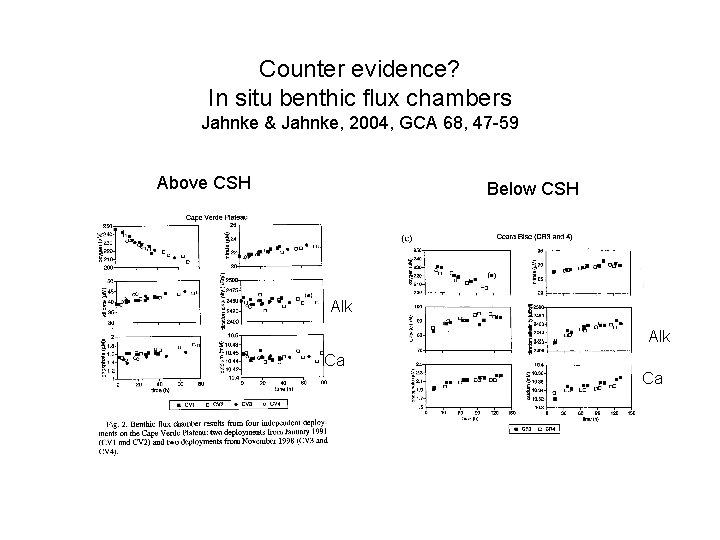
Counter evidence? In situ benthic flux chambers Jahnke & Jahnke, 2004, GCA 68, 47 -59 Above CSH Below CSH Alk Ca Ca
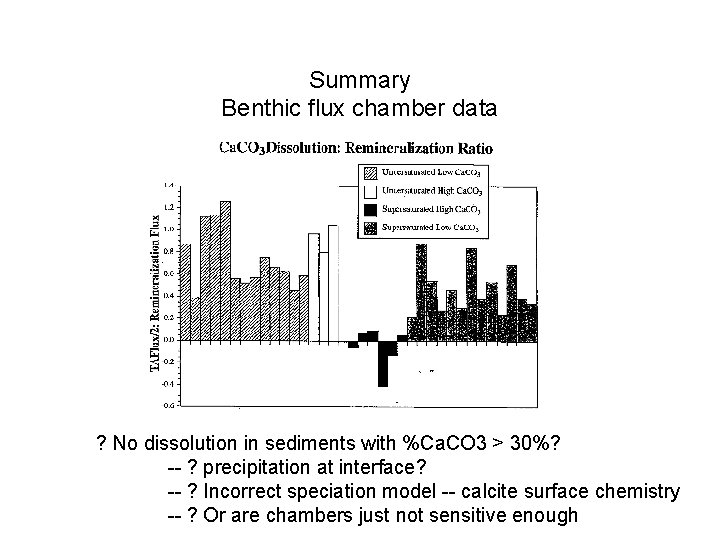
Summary Benthic flux chamber data ? No dissolution in sediments with %Ca. CO 3 > 30%? -- ? precipitation at interface? -- ? Incorrect speciation model -- calcite surface chemistry -- ? Or are chambers just not sensitive enough

One more approach -230 Th activity changes near swi Martin, 2004
Best place to observe this effect: sites with very high %Ca. CO 3 Ontong-Java Plateau

Bottom water saturation state %Ca. CO 3



If metabolic dissolution occurs, how important is it? 2 competing processes: diffusion across sediment-water interface Ca. CO 3 dissolution Balance between them depends on: Ratio, Corg rain : Ca. CO 3 rain -- today, constant at ~ 0. 8 depth distribution of Corg oxidation in sediments saturation state of bottom water

Depth distribution of Corg oxidation in sediments in the deep sea Martin and Sayles (2006) DSR II, 53, 771 -792 If Then “e-folding depth is 1/p 1

How does metabolic dissolution vary with depth distribution of oxidation and saturation state? Model test: Keep Corg and Ca. CO 3 rain rates constant, vary sat. state of bw and efold depth of oxidation

What is a “best guess” of metabolic dissolution -- rain rate ratio constant at ~ 0. 8 -- observed Corg ox. Distribution in sediments Dissolution rate increases with Increasing rain at given saturation Ca. CO 3 burial efficiency is ~ constant At given sat. , but decreases with Decreasing bw. saturation “mde” ~ constant at given sat. , But increases as sat. decreases
- Slides: 40