Department of Chemical Engineering Professor De Chen Institutt


























- Slides: 41

Department of Chemical Engineering Professor De Chen Institutt for kjemisk prosessteknologi, NTNU Gruppe for katalyse og petrokjemi Kjemiblokk V, rom 407 chen@nt. ntnu. no 1 - 11/23/2020

Kjemisk reaksjonsteknikk Chemical Reaction Engineering H. Scott Fogler: Elements of Chemical Engineering www. engin. umich. edu/~cre University of Michigan, USA Department of Chemical Engineering Time plan: Week 34 -47, Tuesday: 08: 15 -10: 00 Thursday: 11: 15: 13: 00 Problem solving: Tuseday: 16: 15 -17: 00 2 - 11/23/2020

Department of Chemical Engineering 3 - 11/23/2020

Kjemisk reaksjonsteknikk Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place. Department of Chemical Engineering 4 - 11/23/2020

Lecture notes will be published on It’s learning after the lecture (Pensumliste ligger på It’s learning Deles ut på de første forelesningene) Department of Chemical Engineering Øvingsopplegget ligger på It’s learning Deles ut på de første forelesningene 5 - 11/23/2020

Felleslaboratorium Faglærer: Professor Heinz Preisig For information: It’s learning Introduction lecture: Department of Chemical Engineering Place : in PFI-50001, the lecture room on the top of the building Date: Tuesday 21 of August Time: 12: 15 - 14: 00 6 - 11/23/2020

TKP 4110 Chemical Reaction Engineering Øvingene starter onsdag 26 august kl 1615 i K 5. Lillebø, Andreas Helland: andreas. lillebo@chemeng. ntnu. no Stud. ass. : Kristian Selvåg : krisse@stud. ntnu. no Department of Chemical Engineering Øyvind Juvkam Eraker: oyvindju@stud. ntnu. no Emily Ann Melsæther: melsathe@stud. ntnu. no 7 - 11/23/2020

Lecture 1 Kjemisk reaksjonsteknikk Chemical Reaction Engineering Department of Chemical Engineering 1. Industrial reactors 2. Reaction engineering 3. Mass balance 4. Ideal reactors 8 - 11/23/2020

Steam Cracking (Rafnes) Department of Chemical Engineering 9 - 11/23/2020

Batch reactor Department of Chemical Engineering 10 - 11/23/2020

Fixed bed reactor Department of Chemical Engineering 11 - 11/23/2020

CSTR bioreactor Department of Chemical Engineering 12 - 11/23/2020

Artificial leaf, photochemical reactor Department of Chemical Engineering 13 - 11/23/2020

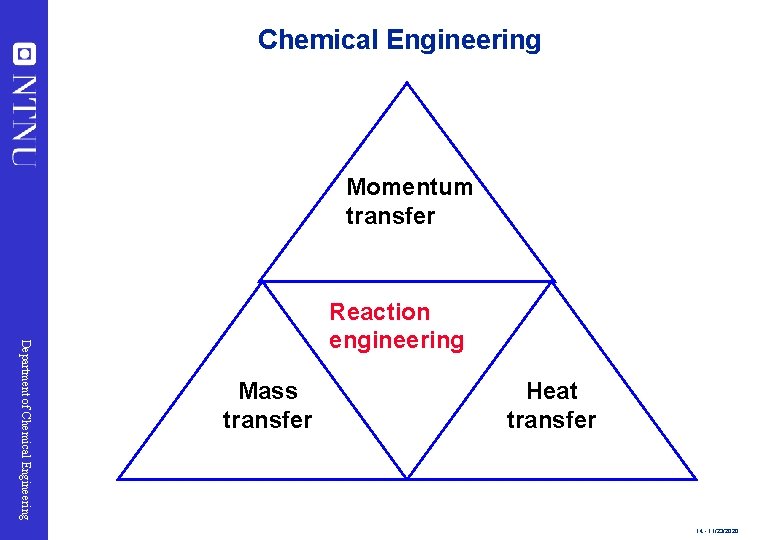
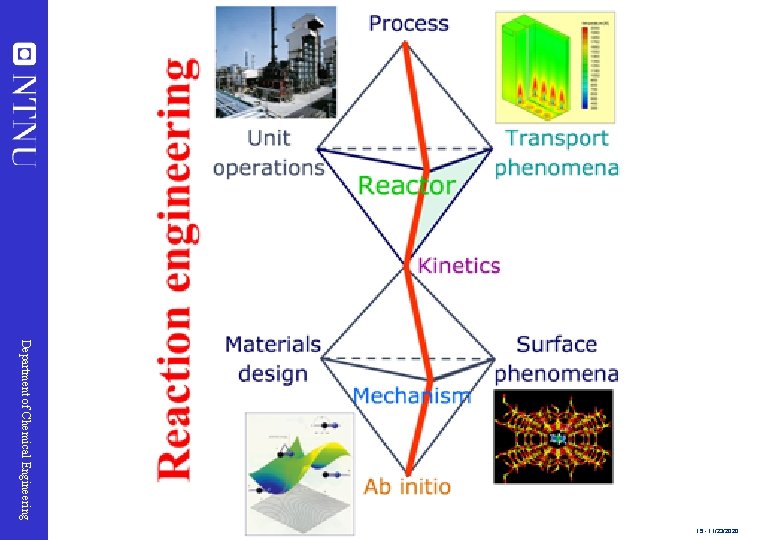
Chemical Engineering Momentum transfer Department of Chemical Engineering Reaction engineering Mass transfer Heat transfer 14 - 11/23/2020

Department of Chemical Engineering 15 - 11/23/2020

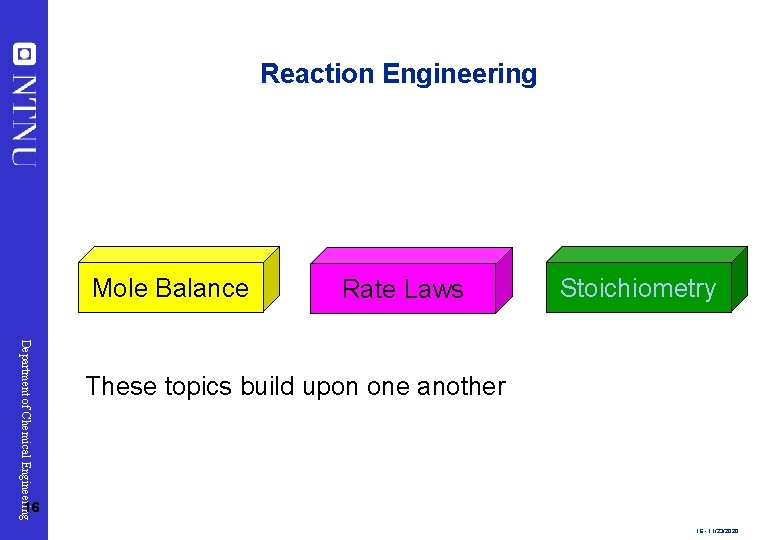
Reaction Engineering Mole Balance Rate Laws Stoichiometry Department of Chemical Engineering These topics build upon one another 16 16 - 11/23/2020

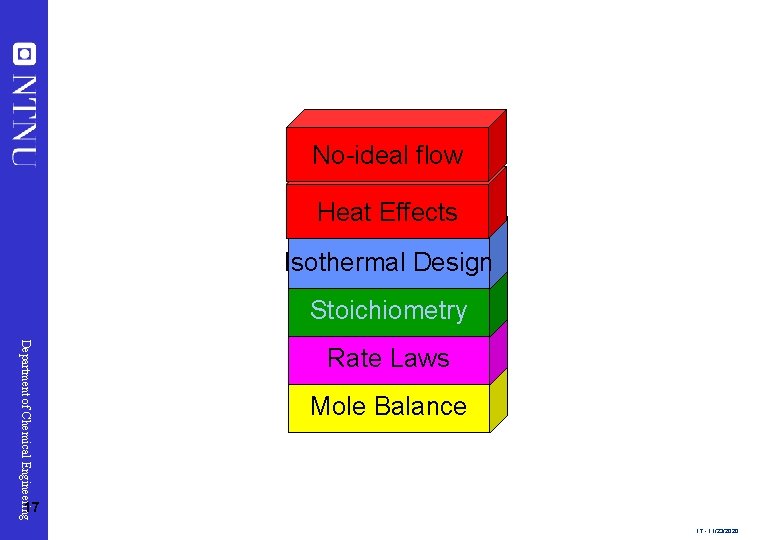
No-ideal flow Heat Effects Isothermal Design Stoichiometry Department of Chemical Engineering Rate Laws Mole Balance 17 17 - 11/23/2020

Chemical kinetics and reactor design are at the heart of producing almost all industrial chemicals Department of Chemical Engineering It is primary a knowledge of chemical kinetics and reactor design that distinguishes the chemical engineer from other engineers 18 - 11/23/2020

Reaction Engineering 1. Week 34, Aug. 21, chapter 1, Introduction, mole balance, and ideal Department of Chemical Engineering reactors, 2. Week 34, Aug. 23, chapter 2, Conversion and reactor size 3. Week 35, Aug. 28, chapter 3, Reaction rates 4. Week 35, Aug. 30, chapter 3, Stoichometric numbers 5. Week 36, Sept. 4, chapter 4, isothermal reactor design (1) 6. Week 36, Sept. 6, chapter 4, isothermal reactor design (2) 7. Week 37, Sept. 11, chapter 10, catalysis and kinetics (1) 8. Week 37, Sept. 13, chapter 10, catalysis and kinetics (2) 9. Week 38, Sept. 18, chapter 10, catalysis and kinetics (2) 10. Week 38, Sept. 20, chapter 5, 7, kinetic modeling (1) 11. Week 39, Sept. 25, chapter 5, 7, kinetic modeling (2) 12. Week 39, Sept. 28 chapter 6, multiple reactions (1) 13. Week 40, Oct. 2, chapter 6 multiple reactions (2) 14. Week 40, Oct. 4, summary of chapter 1 -7, and 10 19 - 11/23/2020

Reaction Engineering 41 (9/10, 11/10) 8. 1 - 8. 2 (JPA) 42 (16/10, 18/10) 8. 3 – 8. 5 (JPA) 43 (23/10, 25/10) 8. 6 - 8. 7 (JPA) 44 (30/10, 1/11) 11 (JPA) 45 (6/11, 8/11) 11 (JPA) Department of Chemical Engineering 46 (13/11, 15/11) 12. 1 -12. 4 (JPA) 47 (20/11, 22/11) 12. 5 -12. 8 (JPA) 50 (Mandag 13/12) Reaktorberegninger for ikke-isoterme systemer. Energibalanser, stasjonær drift. Omsetning ved likevekt. Optimal fødetemperatur. CSTR med varmeeffekter og flere løsninger ved stasjonær drift, ustabilitet. Masseoverføring, ytre diffusjonseffekter i heterogene systemer. Fylte reaktorer (packed beds). Kjernemodellen (shrinking core). Oppløsning av partikler og regenerering av katalysator. Diffusjon og reaksjon i katalysatorpartikler, Thieles modul, effektivitetsfaktor. Masseoverføring og reaksjon i flerfasereaktorer. Oppsummering. Eksamen, kl 0900 -1300. 20 - 11/23/2020

Chemical Identity and reaction A chemical species is said to have reacted when it has lost its chemical identity. There are three ways for a species to loose its identity: Department of Chemical Engineering 1. Decomposition 2. Combination 3. Isomerization CH 3 H 2 + H 2 C=CH 2 N 2 + O 2 2 NO C 2 H 5 CH=CH 2 CH 2=C(CH 3)2 21 21 - 11/23/2020

Reaction Rate The reaction rate is the rate at which a species looses its chemical identity per unit volume. The rate of a reaction (mol/dm 3/s) can be expressed as either: Department of Chemical Engineering The rate of Disappearance of reactant: -r. A or as The rate of Formation (Generation) of product: r. P 22 22 - 11/23/2020

Reaction Rate Department of Chemical Engineering Consider the isomerization A B r. A = the rate of formation of species A per unit volume -r. A = the rate of a disappearance of species A per unit volume r. B = the rate of formation of species B per unit volume 23 23 - 11/23/2020

Reaction Rate For a catalytic reaction, we refer to -r. A', which is the rate of disappearance of species A on a per mass of catalyst basis. (mol/gcat/s) NOTE: d. CA/dt is not the rate of reaction Department of Chemical Engineering 24 24 - 11/23/2020

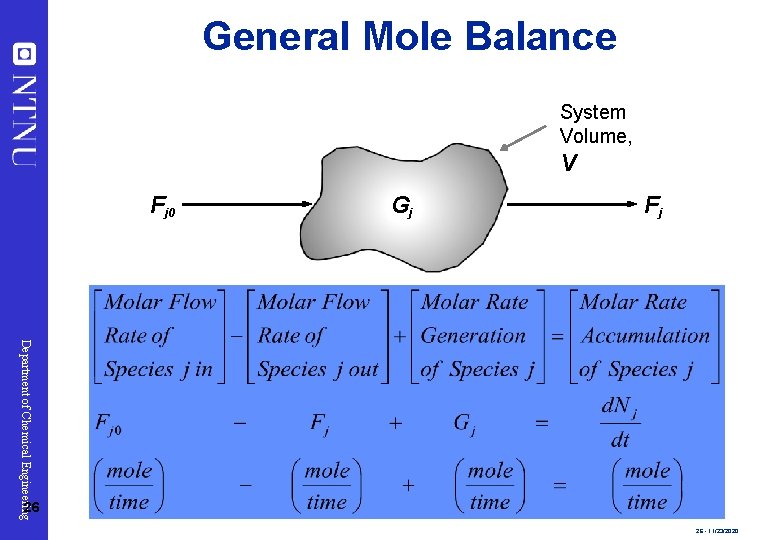
Reaction Rate Department of Chemical Engineering Consider species j: 1. rj is the rate of formation of species j per unit volume [e. g. mol/dm 3 s] 2. rj is a function of concentration, temperature, pressure, and the type of catalyst (if any) 3. rj is independent of the type of reaction system (batch, plug flow, etc. ) 4. rj is an algebraic equation, not a differential equation (e. g. = -r. A = k. CA or -r. A = k. CA 2) 25 25 - 11/23/2020

General Mole Balance System Volume, V Fj 0 Gj Fj Department of Chemical Engineering 26 26 - 11/23/2020

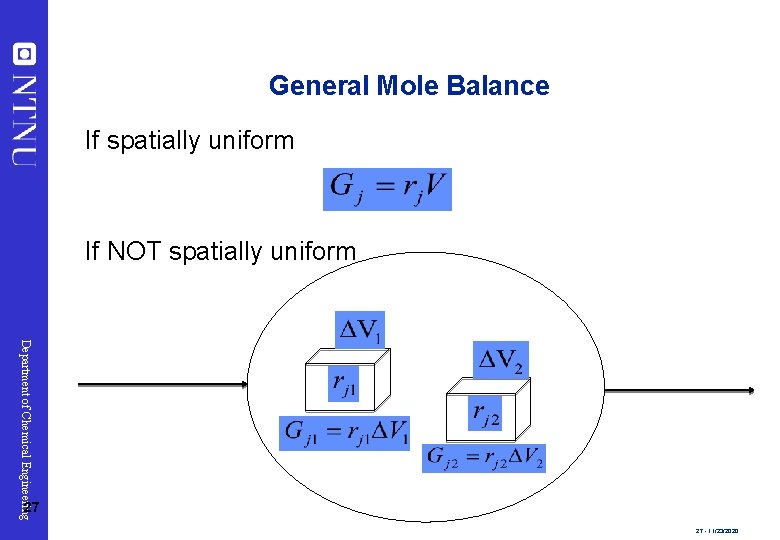
General Mole Balance If spatially uniform If NOT spatially uniform Department of Chemical Engineering 27 27 - 11/23/2020

General Mole Balance Take limit Department of Chemical Engineering 28 28 - 11/23/2020

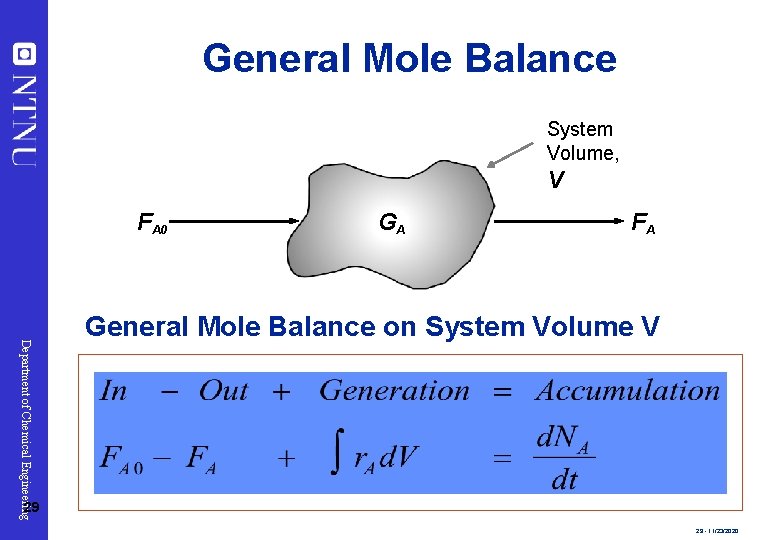
General Mole Balance System Volume, V FA 0 GA FA Department of Chemical Engineering General Mole Balance on System Volume V 29 29 - 11/23/2020

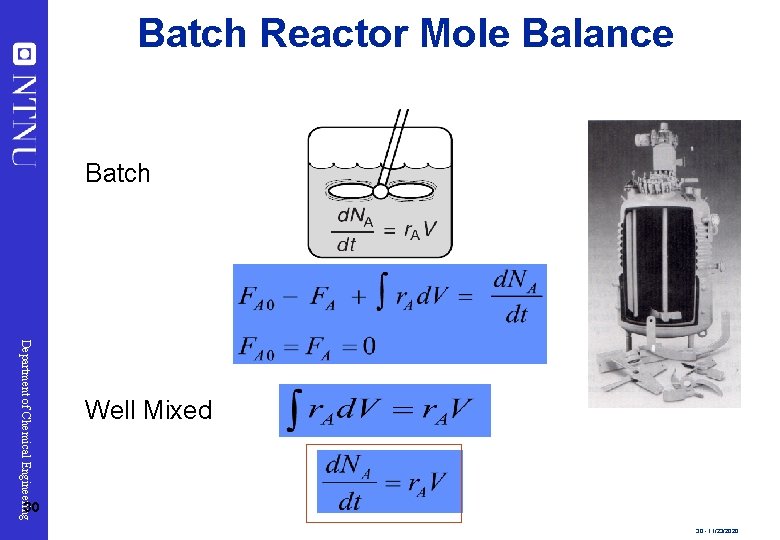
Batch Reactor Mole Balance Batch Department of Chemical Engineering Well Mixed 30 30 - 11/23/2020

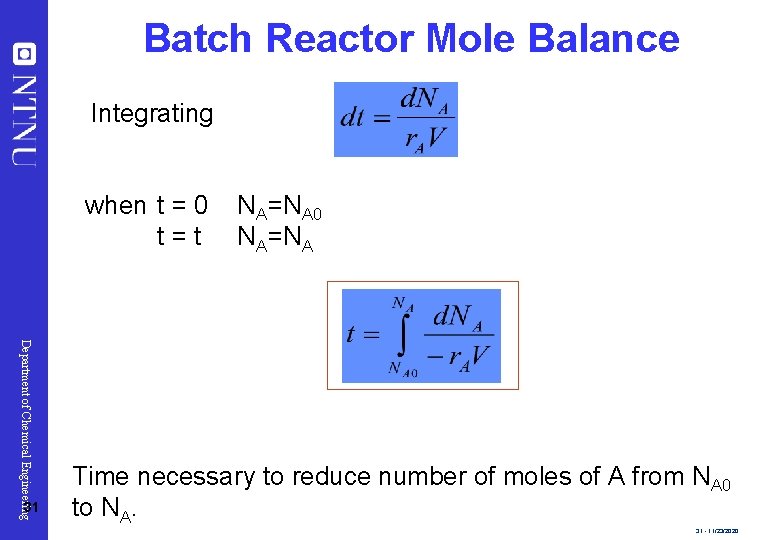
Batch Reactor Mole Balance Integrating when t = 0 t=t Department of Chemical Engineering 31 NA=NA 0 NA=NA Time necessary to reduce number of moles of A from NA 0 to NA. 31 - 11/23/2020

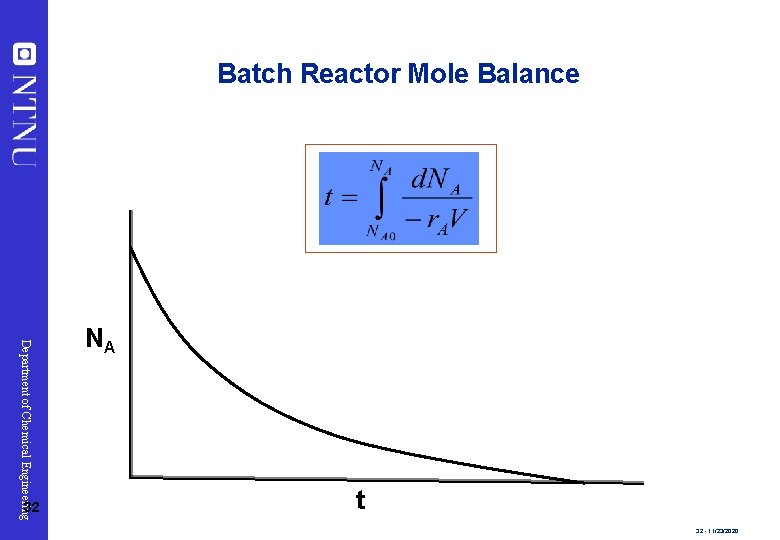
Batch Reactor Mole Balance Department of Chemical Engineering 32 NA t 32 - 11/23/2020

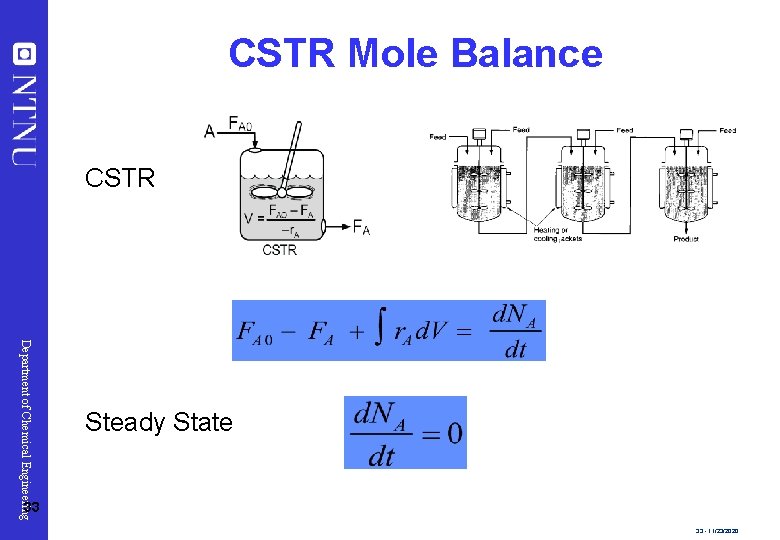
CSTR Mole Balance CSTR Department of Chemical Engineering Steady State 33 33 - 11/23/2020

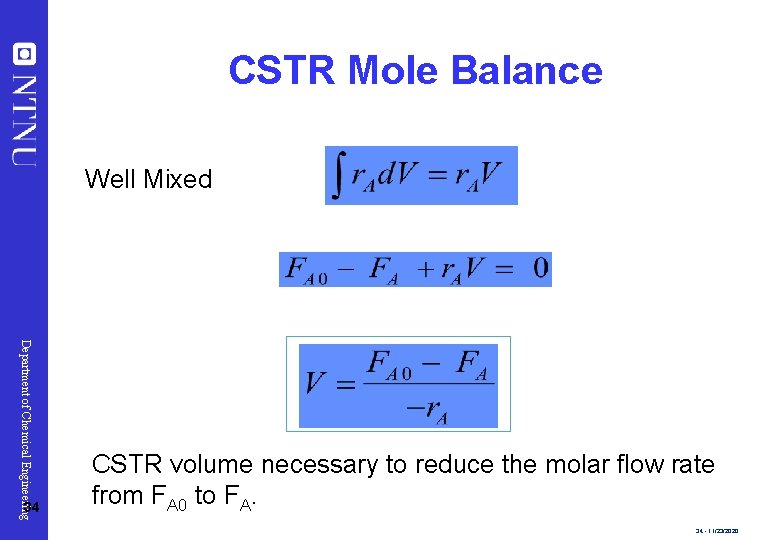
CSTR Mole Balance Well Mixed Department of Chemical Engineering 34 CSTR volume necessary to reduce the molar flow rate from FA 0 to FA. 34 - 11/23/2020

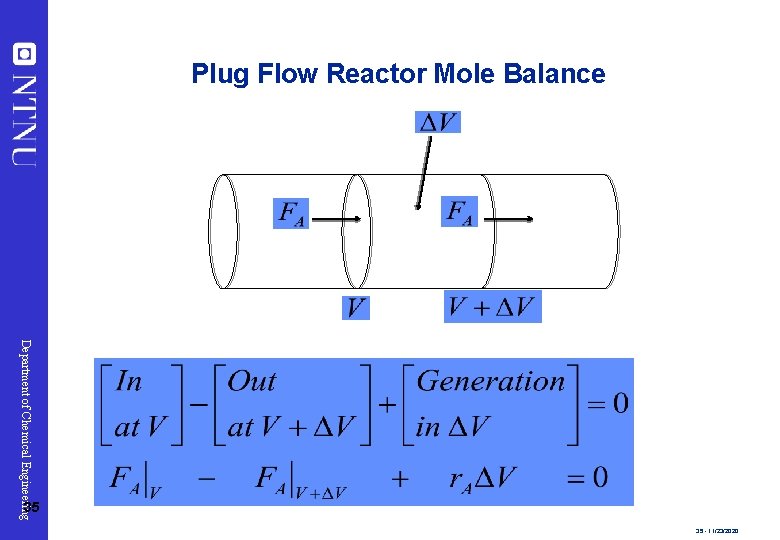
Plug Flow Reactor Mole Balance Department of Chemical Engineering 35 35 - 11/23/2020

Plug Flow Reactor Mole Balance Rearrange and take limit as ΔV 0 Department of Chemical Engineering 36 This is the volume necessary to reduce the entering molar flow rate (mol/s) from FA 0 to the exit molar flow 36 - 11/23/2020

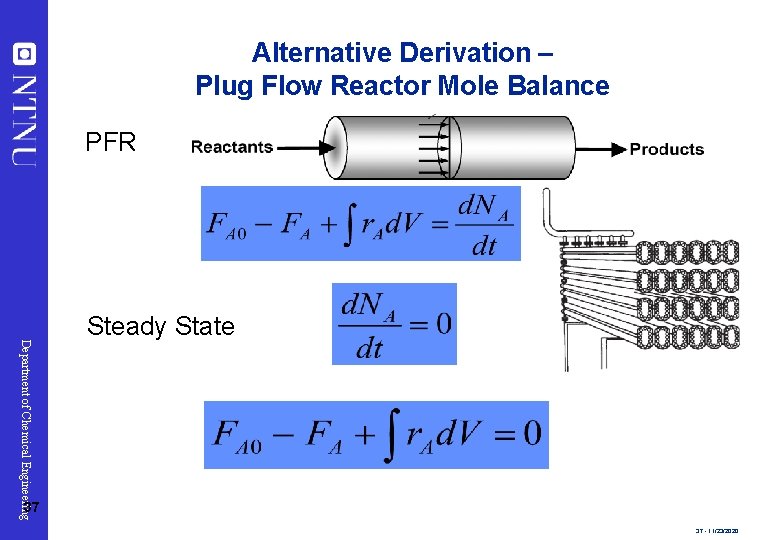
Alternative Derivation – Plug Flow Reactor Mole Balance PFR Department of Chemical Engineering Steady State 37 37 - 11/23/2020

Alternative Derivation – Plug Flow Reactor Mole Balance Differientiate with respect to V Department of Chemical Engineering 38 The integral form is: This is the volume necessary to reduce the entering molar flow rate (mol/s) from FA 0 to the exit molar flow rate of FA. 38 - 11/23/2020

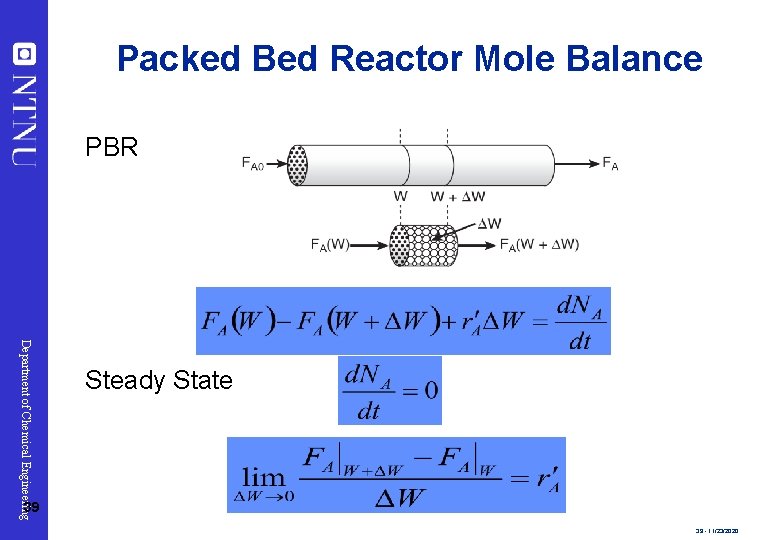
Packed Bed Reactor Mole Balance PBR Department of Chemical Engineering Steady State 39 39 - 11/23/2020

Packed Bed Reactor Mole Balance Rearrange: The integral form to find the catalyst weight is: Department of Chemical Engineering 40 PBR catalyst weight necessary to reduce the entering molar flow rate FA 0 to molar flow rate FA. 40 - 11/23/2020

Reactor Mole Balance Summary Reactor Differential Algebraic Integral NA Batch t Department of Chemical Engineering CSTR FA PFR V 41 41 - 11/23/2020