Chemistry The Central Science 10 th edition Theodore






![p. H = −log [H+] or p. H = −log [H 3 O+] H p. H = −log [H+] or p. H = −log [H 3 O+] H](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-14.jpg)
![[H 3 O+] & [OH–] = Acidic Neutral Basic [H 3 O+] & [OH–] = Acidic Neutral Basic](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-15.jpg)

![“base 10” scale Neutral [H+] p. H p. OH [OH–] 1 x 10– 0 “base 10” scale Neutral [H+] p. H p. OH [OH–] 1 x 10– 0](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-17.jpg)
![[H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14. [H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14.](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-18.jpg)
![[H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14. [H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14.](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-19.jpg)
![p. H p. OH [H+] [OH–] Calculations on equation sheet p. H = –log[H+] p. H p. OH [H+] [OH–] Calculations on equation sheet p. H = –log[H+]](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-20.jpg)
![p. H & p. OH Calculations p. H = –log[H+] = 1 x 10– p. H & p. OH Calculations p. H = –log[H+] = 1 x 10–](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-21.jpg)


![I Do Know This Stuff! 2) The [H 3 O+] for four solutions is I Do Know This Stuff! 2) The [H 3 O+] for four solutions is](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-25.jpg)





![Calculating Percent Ionization NOT on [H 3 O+]eq % ionization = [HA]in 100% equation Calculating Percent Ionization NOT on [H 3 O+]eq % ionization = [HA]in 100% equation](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-33.jpg)
![Calculating p. H from Ka [H 3 O+] [C 2 H 3 O 2−] Calculating p. H from Ka [H 3 O+] [C 2 H 3 O 2−]](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-35.jpg)
![Kb can be used to find [OH−], then p. OH, & p. H. Weak Kb can be used to find [OH−], then p. OH, & p. H. Weak](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-38.jpg)

![Calculating p. H of Basic Solutions [NH 4+] [OH−] Kb = [NH 3] Kb Calculating p. H of Basic Solutions [NH 4+] [OH−] Kb = [NH 3] Kb](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-42.jpg)
![Calculating p. H of Basic Solutions [OH−] = 1. 6 10− 3 M Therefore, Calculating p. H of Basic Solutions [OH−] = 1. 6 10− 3 M Therefore,](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-43.jpg)




- Slides: 48

Chemistry, The Central Science, 10 th edition Theodore L. Brown; H. Eugene Le. May, Jr. ; and Bruce E. Bursten Unit 9 (Chp 16): Acid-Base Equilibria (Ka, Kb, Kw, p. H, p. OH) John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall, Inc.

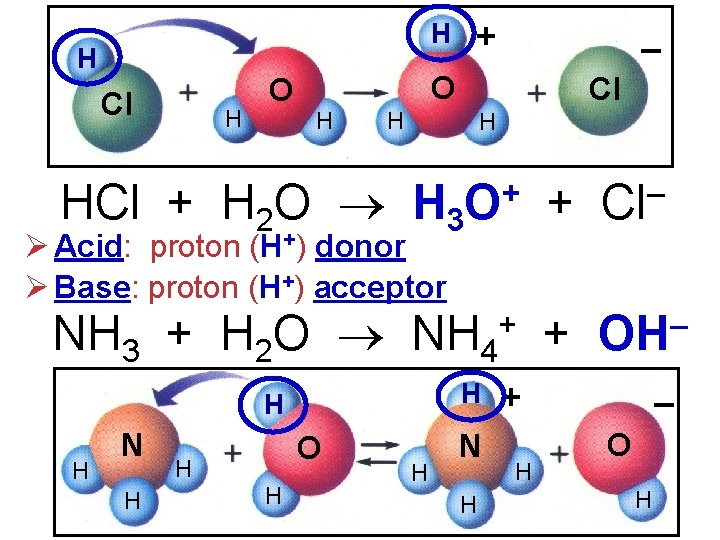
H H Cl H O + – O H Cl H H HCl + H 2 O H 3 O+ + Cl– Ø Acid: proton (H+) donor Ø Base: proton (H+) acceptor NH 3 + H 2 O NH 4 H H H N H O H H H N H + + OH– + H – O H

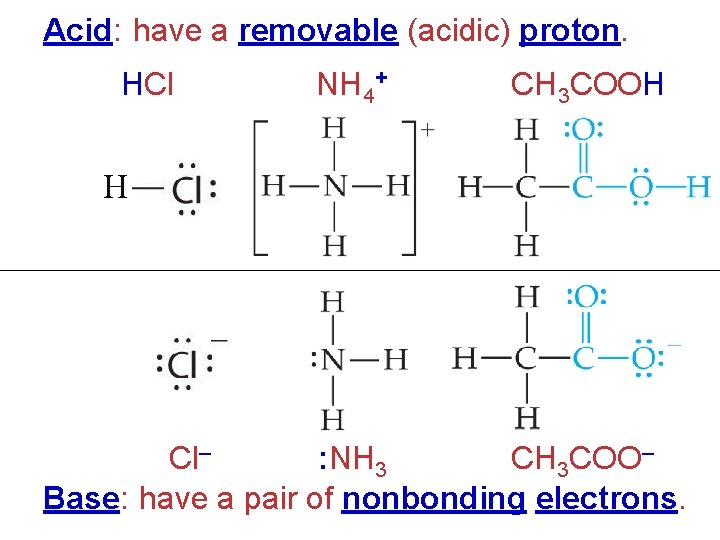
Acid: have a removable (acidic) proton. HCl NH 4+ CH 3 COOH H Cl– : NH 3 CH 3 COO– Base: have a pair of nonbonding electrons.

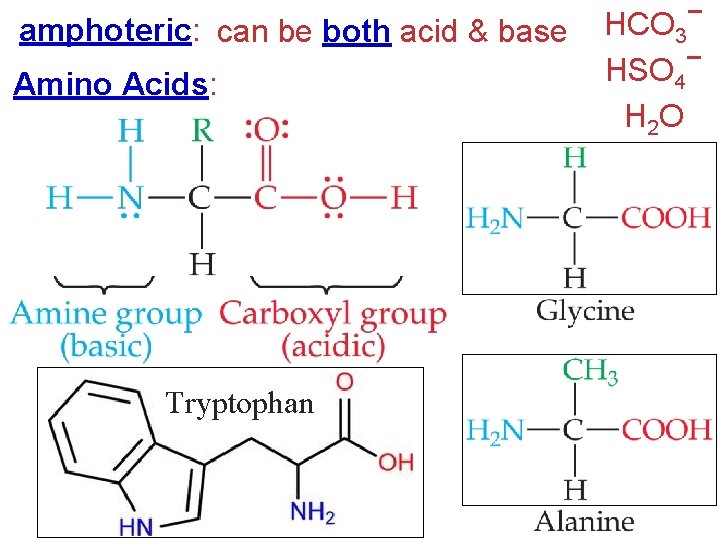
amphoteric: can be both acid & base Amino Acids: Tryptophan HCO 3− − HSO 4 H 2 O

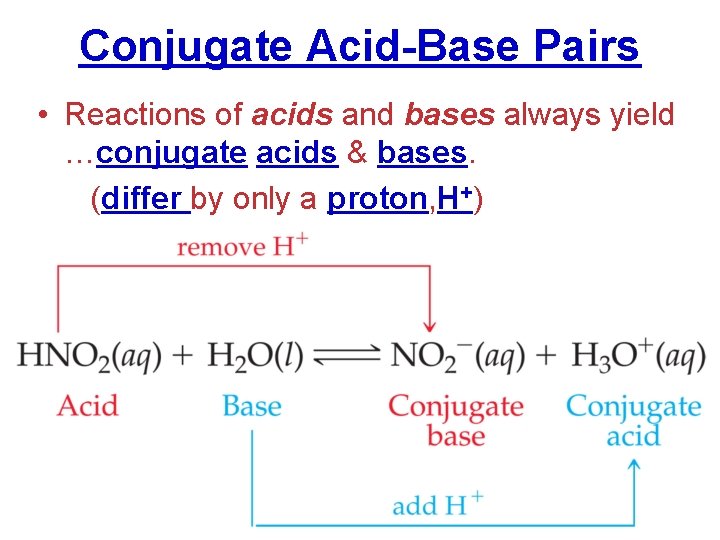
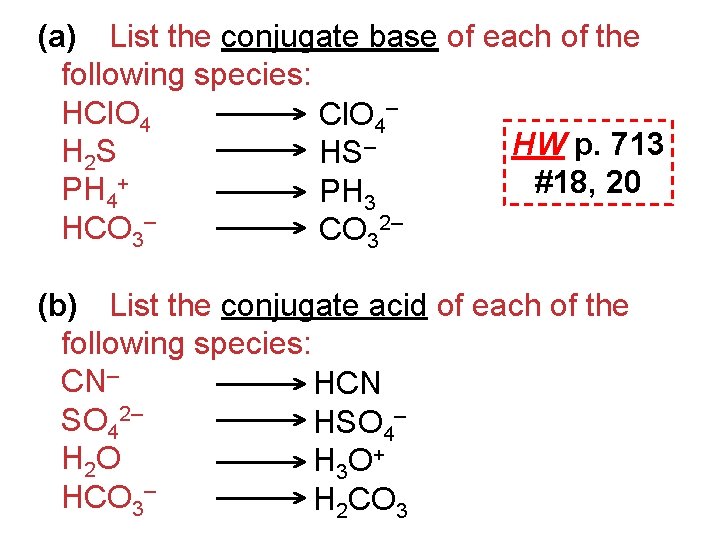
Conjugate Acid-Base Pairs • Reactions of acids and bases always yield …conjugate acids & bases. (differ by only a proton, H+)

(a) List the conjugate base of each of the following species: HCl. O 4– HW p. 713 H 2 S HS– #18, 20 PH 4+ PH 3 HCO 3– CO 32– (b) List the conjugate acid of each of the following species: CN– HCN SO 42– HSO 4– H 2 O H 3 O + HCO 3– H 2 CO 3

Strong Acids • Recall the six Strong Acids are… HNO 3 , H 2 SO 4 , HCl, HBr, HI, and HCl. O 4. • strong electrolytes (completely ionized) (exist totally as ions in aqueous solution) HNO 3 + H 2 O H 3 O+ + NO 3– • For the monoprotic strong acids, [acid] = [H 3 O+] 0. 50 M HCl means… [H 3 O+] = 0. 50 M

Strong Bases • Strong Bases are the soluble hydroxides (OH–) of… Group 1 (Li, Na, K, Rb, Cs) and Ca 2+, Ba 2+, Sr 2+. • strong electrolytes (completely dissociated into ions in aqueous solution). Na. OH Na+ + OH– • and accept H’s easily in aqueous solution. OH– + HA H 2 O + A–

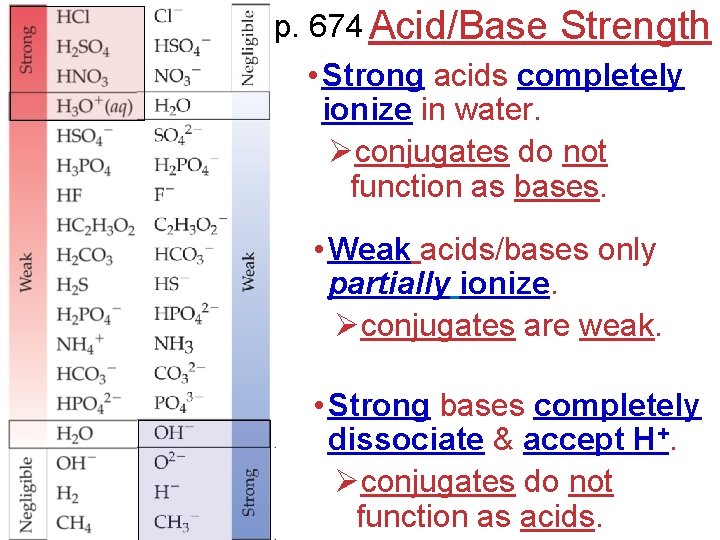
p. 674 Acid/Base Strength • Strong acids completely ionize in water. Øconjugates do not function as bases. • Weak acids/bases only partially ionize. Øconjugates are weak. • Strong bases completely dissociate & accept H+. Øconjugates do not function as acids.

Acid/Base Strength The equilibrium will favor the direction that moves the proton away from stronger acid. (OR…the stronger base gets the proton) HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl−(aq) HCl is stronger acid than H 3 O+, and equilibrium lies so far to the right K is not measured (K>>>1). Which side is favored? HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2−(aq) H 3 O+ is a stronger acid than HC 2 H 3 O 2, HW p. 714 equilibrium favors left side reactants (K<1). #26

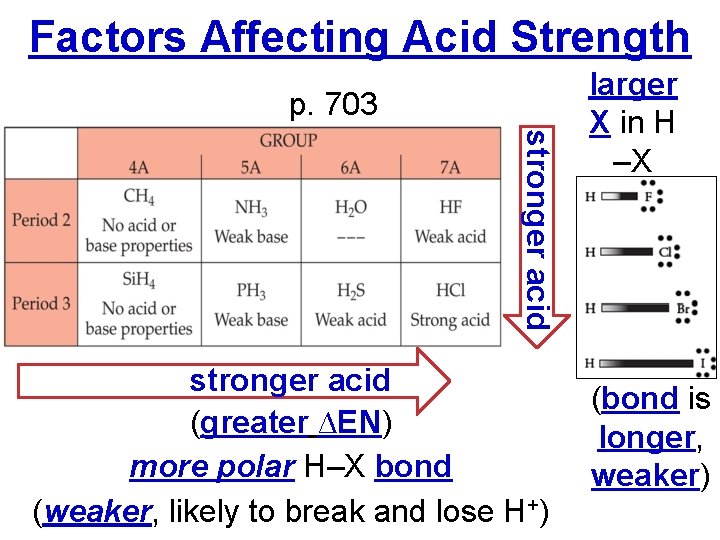
Factors Affecting Acid Strength p. 703 stronger acid (greater ∆EN) more polar H–X bond (weaker, likely to break and lose H+) larger X in H –X (bond is longer, weaker)

Factors Affecting Acid Strength p. 705 • The more O’s the more polar (weaker) the H-O bond the more acidic (more likely to lose H+) HW p. 716 #93

Autoionization of Water • Recall, water is amphoteric. • In a sample of pure water, a few molecules act as bases and a few act as acids. Write the K exp. for the autoionization of water: H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH−(aq) Kw = [H 3 O+] [OH−] = 1. 0 10– 14 on equation sheet (at 25 o. C) • This special equilibrium constant is called the ion-product constant for water, Kw.
![p H log H or p H log H 3 O H p. H = −log [H+] or p. H = −log [H 3 O+] H](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-14.jpg)
p. H = −log [H+] or p. H = −log [H 3 O+] H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH−(aq) In pure water, [H 3 O+] = [OH−] Kw = [H 3 O+] [OH−] = 1. 0 10− 14 x 2 = 1. 0 10− 14 [H 3 O+] = 1. 0 10− 7 M (in pure water) p. H = −log(1. 0 10− 7) = 7. 00 • Acids: higher [H 3 O+], 1 x 10–(<7) , so p. H <7 • Bases: lower [H 3 O+], 1 x 10–(>7) , so p. H >7
![H 3 O OH Acidic Neutral Basic [H 3 O+] & [OH–] = Acidic Neutral Basic](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-15.jpg)
[H 3 O+] & [OH–] = Acidic Neutral Basic

“p” Scales • The “p” in p. H means to take the negative log of the quantity (in this case, H+ ions). • Some similar examples are p. OH = −log [OH–] on equation sheet Kw = 1. 0 10− 14 = [H 3 O+] [OH−] −log Kw = 14. 00 = −log [H 3 O+] + −log [OH−] p. Kw = 14 = p. H on equation sheet + p. OH
![base 10 scale Neutral H p H p OH OH 1 x 10 0 “base 10” scale Neutral [H+] p. H p. OH [OH–] 1 x 10– 0](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-17.jpg)
“base 10” scale Neutral [H+] p. H p. OH [OH–] 1 x 10– 0 0. 0 14. 0 1 x 10– 14 1 x 10– 1 1. 0 13. 0 1 x 10– 13 1 x 10– 2 2. 0 1 x 10– 12 1 x 10– 3 3. 0 11. 0 1 x 10– 11 1 x 10– 4 4. 0 10. 0 1 x 10– 10 1 x 10– 5 5. 0 9. 0 1 x 10– 9 1 x 10– 6 6. 0 8. 0 1 x 10– 8 1 x 10– 7 7. 0 1 x 10– 7 1 x 10– 8 8. 0 6. 0 1 x 10– 6 1 x 10– 9 9. 0 5. 0 1 x 10– 5 1 x 10– 10 10. 0 4. 0 1 x 10– 4 1 x 10– 11 11. 0 3. 0 1 x 10– 3 1 x 10– 12 12. 0 1 x 10– 2 1 x 10– 13 13. 0 1 x 10– 14 14. 0 0. 0 1 x 10– 0 Kw = [H+]x[OH–] 1 x 10– 14 1 x 10– 14 1 x 10– 14 1 x 10– 14
![H Neutral p H p OH OH 1 x 10 0 0 0 14 [H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14.](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-18.jpg)
[H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14. 0 1 x 10– 14 1 x 10– 1 1. 0 13. 0 1 x 10– 13 1 x 10– 2 2. 0 1 x 10– 12 1 x 10– 3 3. 0 11. 0 1 x 10– 11 1 x 10– 4 4. 0 10. 0 1 x 10– 10 1 x 10– 5 5. 0 9. 0 1 x 10– 9 1 x 10– 6 6. 0 8. 0 1 x 10– 8 1 x 10– 7 7. 0 1 x 10– 7 1 x 10– 8 8. 0 6. 0 1 x 10– 6 1 x 10– 9 9. 0? 5. 0? 1 x 10 ? – 5 1 x 10– 10 10. 0 4. 0 1 x 10– 4 1 x 10– 11 11. 0 3. 0 1 x 10– 3 1 x 10– 12 12. 0 1 x 10– 2 1 x 10– 13 13. 0 1 x 10– 14 14. 0 0. 0 1 x 10– 0 Kw = [H+]x[OH–] 1 x 10– 14 1 x 10– 14 1 x 10– 14 1 x 10– 14
![H Neutral p H p OH OH 1 x 10 0 0 0 14 [H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14.](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-19.jpg)
[H+] Neutral p. H p. OH [OH–] 1 x 10– 0 0. 0 14. 0 1 x 10– 14 1 x 10– 1 1. 0 13. 0 1 x 10– 13 1 x 10– 2 2. 0 1 x 10– 12 1 x 10 ? – 3 3. 0? 11. 0 ? 1 x 10– 11 1 x 10– 4 4. 0 10. 0 1 x 10– 10 1 x 10– 5 5. 0 9. 0 1 x 10– 9 1 x 10– 6 6. 0 8. 0 1 x 10– 8 1 x 10– 7 7. 0 1 x 10– 7 1 x 10– 8 8. 0 6. 0 1 x 10– 6 1 x 10– 9 9. 0 5. 0 1 x 10– 5 1 x 10– 10 10. 0 4. 0 1 x 10– 4 1 x 10– 11 11. 0 3. 0 1 x 10– 3 1 x 10– 12 12. 0 1 x 10– 2 1 x 10– 13 13. 0 1 x 10– 14 14. 0 0. 0 1 x 10– 0 Kw = [H+]x[OH–] 1 x 10– 14 1 x 10– 14 1 x 10– 14 1 x 10– 14
![p H p OH H OH Calculations on equation sheet p H logH p. H p. OH [H+] [OH–] Calculations on equation sheet p. H = –log[H+]](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-20.jpg)
p. H p. OH [H+] [OH–] Calculations on equation sheet p. H = –log[H+] p. OH = –log[OH–] 10 –p. H = [H+] NOT 10 –p. OH = [OH–] on equation sheet Kw – 14 + – = [H ] [OH ] = 1. 0 x 10 14 = p. H + p. OH on equation sheet NOT
![p H p OH Calculations p H logH 1 x 10 p. H & p. OH Calculations p. H = –log[H+] = 1 x 10–](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-21.jpg)
p. H & p. OH Calculations p. H = –log[H+] = 1 x 10– 14 [OH–] ALL on the Equation Sheet! p. OH = 14 – p. H = 14 – p. OH – OH p. OH = –log[OH–] [H+] = 10–p. H + H [OH–] = 10–p. OH [OH–] = 1 x 10– 14 [H+] p. OH

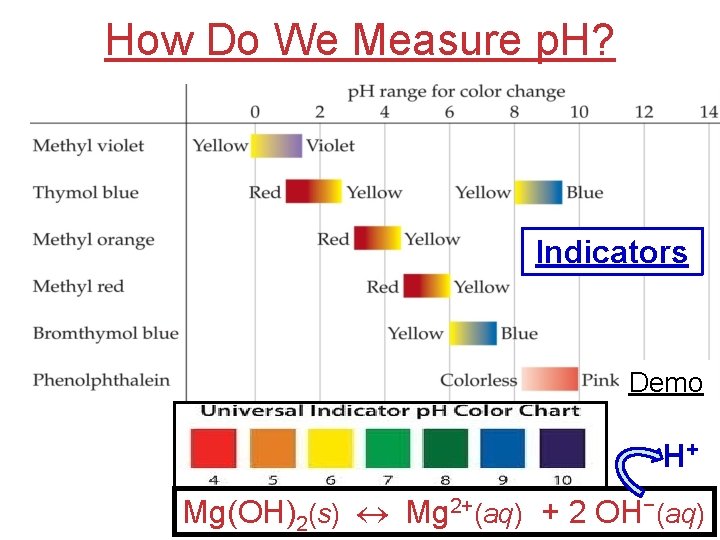
How Do We Measure p. H? Indicators Demo H+ Mg(OH)2(s) Mg 2+(aq) + 2 OH−(aq)

How Do We Measure p. H? For accurate measurements, use a p. H meter, which measures voltage in solution by tracking the number of H+ particles that contact the device.

I Think I Know This Stuff. 1) Which one of the following solutions is the most basic? A) soap p. H ≈ 10 B) milk p. H ≈ 6. 5 C) vinegar p. H ≈ 2. 5 D) ammonia p. H ≈ 11 E) tomato juice F) blood p. H ≈ 4. 0 p. H ≈ 7. 4
![I Do Know This Stuff 2 The H 3 O for four solutions is I Do Know This Stuff! 2) The [H 3 O+] for four solutions is](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-25.jpg)
I Do Know This Stuff! 2) The [H 3 O+] for four solutions is given below. Which one of the solutions is the most acidic? A) 1 x 10– 3 M B) 1 x 10– 7 M C) 1 x 10– 9 M D) 1 x 10– 14 M

Can I Calculate This Stuff? 3) What is the p. H of a solution with a [H+] concentration of 2. 50 x 10– 11 M ? A) 12. 93 B) 10. 60 C) 8. 92 D) 5. 50

I Can Calculate This Stuff! 4) What is the p. H of a solution with a [OH–] concentration of 1. 50 x 10– 3 M ? A) B) C) 2. 82 11. 18 6. 67 x 10– 12 D) 1. 50 x 10– 12 WS Acids & Bases 1 #1 -3 p. OH = –log[OH–] p. H = 14 – p. OH OR [H+] = 1 x 10– 14 [OH–] p. H = –log[H+]

Dissociation Constants (Ka , Kb) • For a generalized acid dissociation, HA(aq) + H 2 O(l) H 3 O+(aq) + A−(aq) the equilibrium expression would be [H 3 O+] [A−] on equation Ka = sheet [HA] • called the acid-dissociation constant, Ka. p. Ka = −log Ka on equation sheet

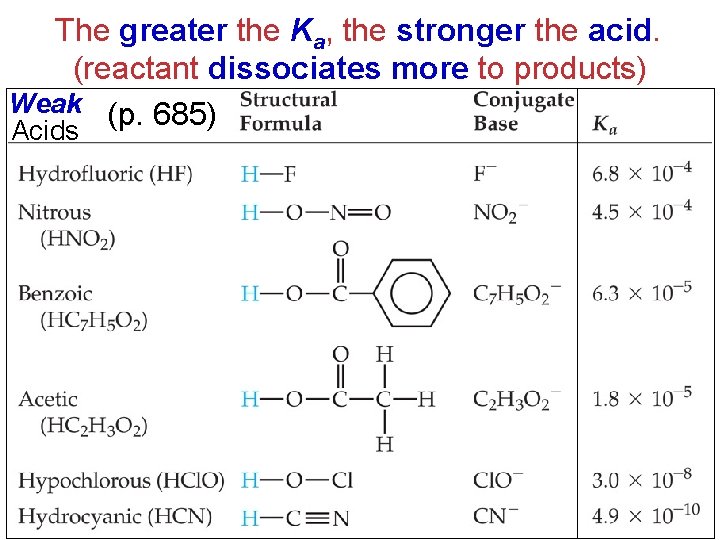
The greater the Ka, the stronger the acid. (reactant dissociates more to products) Weak (p. 685) Acids

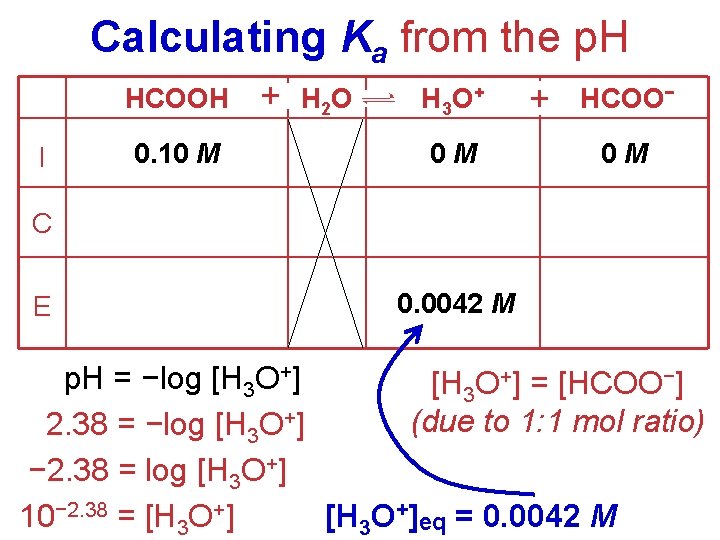
Calculating Ka from the p. H • The p. H of a 0. 10 M solution of formic acid, HCOOH, at 25 o. C is 2. 38. Calculate Ka formic acid at 25 o. C. HCOOH(aq) + H 2 O(l) H 3 O+(aq) + HCOO–(aq) [H 3 O+] [HCOO−] • We know: Ka = [HCOOH] • We need: all 3 concentrations at equilibrium. • We are given: • [HCOOH]initial = 0. 10 M • get [H 3 O+]eq from p. H.

Calculating Ka from the p. H HCOOH I 0. 10 M + H 2 O H 3 O + 0 M + HCOO− 0 M C E 0. 0042 M p. H = −log [H 3 O+] = [HCOO−] (due to 1: 1 mol ratio) 2. 38 = −log [H 3 O+] − 2. 38 = log [H 3 O+]eq = 0. 0042 M 10− 2. 38 = [H 3 O+]

Calculating Ka from the p. H HCOOH + H 2 O H 3 O + + HCOO− I 0. 10 M 0 M 0 M C − 0. 0042 +0. 0042 M E 0. 10 − 0. 0042 = 0. 0958 = 0. 10 M Ka = [H 3 O+] [HCOO−] [HCOOH] 2 (0. 0042) Ka = (0. 10) Ka = 1. 8 10− 4
![Calculating Percent Ionization NOT on H 3 Oeq ionization HAin 100 equation Calculating Percent Ionization NOT on [H 3 O+]eq % ionization = [HA]in 100% equation](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-33.jpg)
Calculating Percent Ionization NOT on [H 3 O+]eq % ionization = [HA]in 100% equation sheet HCOOH(aq) + H 2 O(l) H 3 O+(aq) + HCOO–(aq) HCOOH + H 2 O I 0. 10 M C − 0. 0042 E 0. 10 − 0. 0042 = 0. 0958 = 0. 10 M H 3 O + 0 M +0. 0042 M + HCOO− 0 M +0. 0042 M 0. 0042 % ionization = 100% = 4. 2% 0. 10

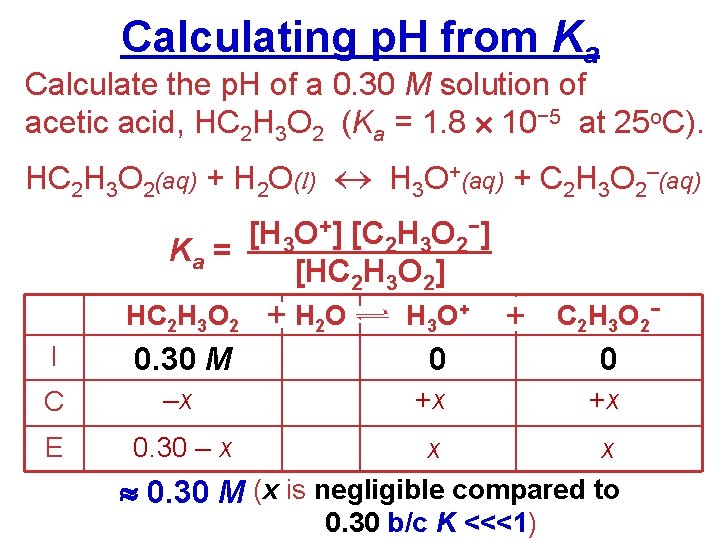
Calculating p. H from Ka Calculate the p. H of a 0. 30 M solution of acetic acid, HC 2 H 3 O 2 (Ka = 1. 8 10− 5 at 25 o. C). HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2–(aq) [H 3 O+] [C 2 H 3 O 2−] Ka = [HC 2 H 3 O 2] HC 2 H 3 O 2 + H 2 O H 3 O + + C 2 H 3 O 2− I 0. 30 M 0 0 C –x +x +x E 0. 30 – x x x 0. 30 M (x is negligible compared to 0. 30 b/c K <<<1)
![Calculating p H from Ka H 3 O C 2 H 3 O 2 Calculating p. H from Ka [H 3 O+] [C 2 H 3 O 2−]](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-35.jpg)
Calculating p. H from Ka [H 3 O+] [C 2 H 3 O 2−] Ka = [HC 2 H 3 O 2] 2 (x) 1. 8 10− 5 = (0. 30) (1. 8 10− 5) (0. 30) = x 2 5. 4 10− 6 = x 2 − 3 = x 2. 3 10 HW p. 715 #56, 62 ab p. H = −log [H 3 O+] − 3) p. H = −log (2. 3 10 WS Acids & Bases 1 #7 -9 WS Acids & Bases 2 #1 -2 p. H = 2. 64

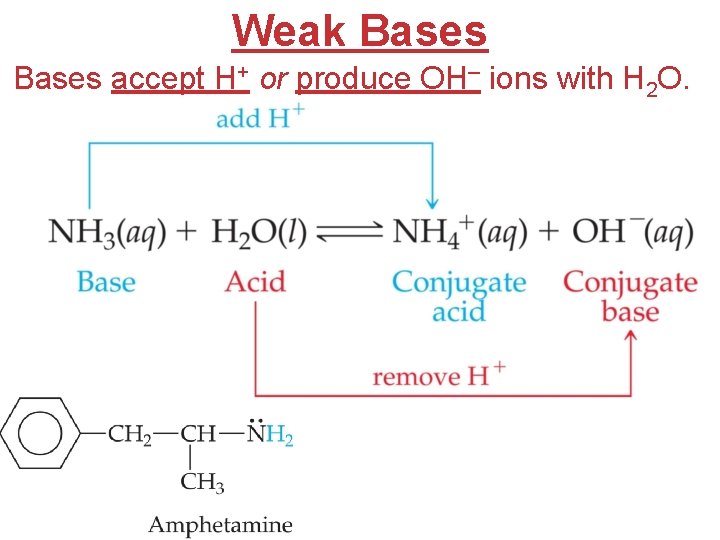
Weak Bases accept H+ or produce OH– ions with H 2 O.

Dissociation Constants • For a generalized base dissociation, B(aq) + H 2 O(l) BH+(aq) + OH−(aq) the equilibrium expression would be [BH+] [OH−] Kb = [B] where Kb is the base-dissociation constant. OR B–(aq) + H 2 O(l) HB(aq) + OH−(aq)
![Kb can be used to find OH then p OH p H Weak Kb can be used to find [OH−], then p. OH, & p. H. Weak](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-38.jpg)
Kb can be used to find [OH−], then p. OH, & p. H. Weak (p. 694) Bases

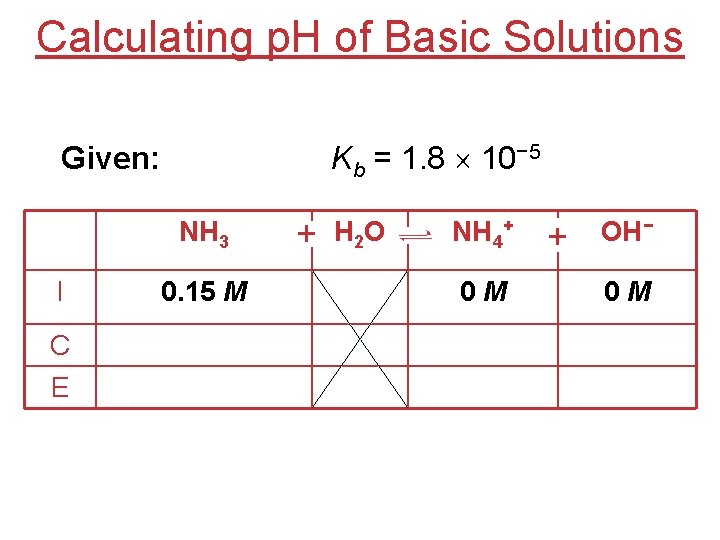
Calculating p. H of Basic Solutions What is the p. H of a 0. 15 M solution of NH 3? NH 3(aq) + H 2 O(l) NH 4+(aq) + OH−(aq) [NH 4+] [OH−] Kb = [NH 3] Kb = 1. 8 10− 5

Calculating p. H of Basic Solutions Given: Kb = 1. 8 10− 5 NH 3 I C E 0. 15 M + H 2 O NH 4+ 0 M + OH− 0 M

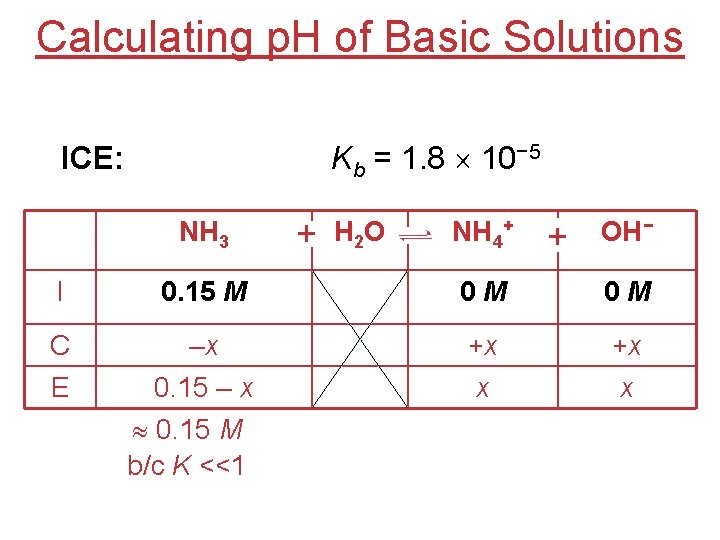
Calculating p. H of Basic Solutions ICE: Kb = 1. 8 10− 5 NH 3 + H 2 O NH 4+ + OH− I 0. 15 M 0 M 0 M C –x +x +x x x E 0. 15 – x 0. 15 M b/c K <<1
![Calculating p H of Basic Solutions NH 4 OH Kb NH 3 Kb Calculating p. H of Basic Solutions [NH 4+] [OH−] Kb = [NH 3] Kb](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-42.jpg)
Calculating p. H of Basic Solutions [NH 4+] [OH−] Kb = [NH 3] Kb = 1. 8 10− 5 2 (x) 1. 8 10− 5 = (0. 15) (1. 8 10− 5) (0. 15) = x 2 2. 7 10− 6 = x 2 1. 6 10− 3 = x 2 [OH−] = 1. 6 10− 3 M
![Calculating p H of Basic Solutions OH 1 6 10 3 M Therefore Calculating p. H of Basic Solutions [OH−] = 1. 6 10− 3 M Therefore,](https://slidetodoc.com/presentation_image_h/1c56b6f41cdb46efdcd0a91a78f0a2c5/image-43.jpg)
Calculating p. H of Basic Solutions [OH−] = 1. 6 10− 3 M Therefore, p. OH = −log [OH−] p. OH = −log (1. 6 10− 3) p. OH = 2. 80 p. H + p. OH = 14 p. H + 2. 80 = 14. 00 p. H = 11. 20 HW p. 716 #76, 78

Ka and Kb are related in this way: Ka Kb = on equation Kw sheet Therefore: If you only know one of them, HW p. 716 #82, 84 a you can calculate the other.

Acidic/Basic Ions with Water • ANIONS are bases (raise p. H). • react with water in hydrolysis reaction to form OH− and the conjugate acid: X−(aq) + H 2 O(l) HX(aq) + OH−(aq) anion base conj. acid • CATIONS with acidic protons (H+) (like NH 4+, CH 3 NH 3+, etc. ) will lower p. H. NH 4+(aq) + H 2 O(l) NH 3(aq) + H 3 O+(aq)

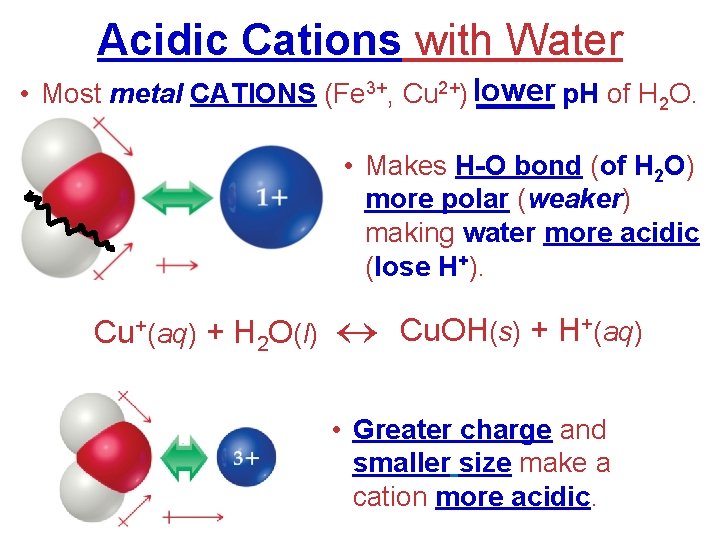
Acidic Cations with Water lower • Most metal CATIONS (Fe 3+, Cu 2+) _____ p. H of H 2 O. • Makes H-O bond (of H 2 O) more polar (weaker) making water more acidic (lose H+). Cu+(aq) + H 2 O(l) Cu. OH(s) + H+(aq) • Greater charge and smaller size make a cation more acidic.

Effect of Cations on p. H Na+ Ca 2+ Zn 2+ Al 3+ M+(aq) + H 2 O(l) MOH(s) + H+(aq) Na. OH(s) Ca(OH)2(s) Zn(OH)2(s) Al(OH)3(s) Consider the size and charge of each metal ion and its effect on the p. H of solution.

(donates H+) Cations: NH 4+(aq) + H 2 O(l) NH 3(aq) + H 3 O+(aq) (acidic, ↓p. H) Cu+(aq) + H O(l) Cu. OH(s) + H+(aq) 2 1. Most metal cations will lower p. H (acidic). (Fe 3+, Zn 2+, …) (take OH– from H 2 O to lose H+) 2. But, metal cations of strong base CAN’T affect p. H. (Li+, Na+, K+, Ca 2+, Ba 2+) (can’t take OH– from H 2 O) Anions: (basic, ↑p. H) F−(aq) + H 2 O(l) HF(aq) + OH−(aq) 3. Most anions will increase p. H (basic). (F–, HCO 3–, NO 2–) (take H+ from H 2 O) 4. But, anions from conj. base of a strong acid CAN’T affect p. H. (NO 3–, HSO 4–, Cl–) (can’t take H+ from H 2 O)