Chemistry The Central Science 10 th edition Theodore



















![4 Types of Bonding in Solids Diamond ØCovalent [nonmetals] • sharing e–’s in mutual 4 Types of Bonding in Solids Diamond ØCovalent [nonmetals] • sharing e–’s in mutual](https://slidetodoc.com/presentation_image/932cf791ab8b2c86bc029dc6dd2152af/image-64.jpg)







- Slides: 74

Chemistry, The Central Science, 10 th edition Theodore L. Brown; H. Eugene Le. May, Jr. ; and Bruce E. Bursten Unit 5 (Chp 11, 13) Bonds & IMAFs in Solids, Liquids, and Solutions John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall, Inc.

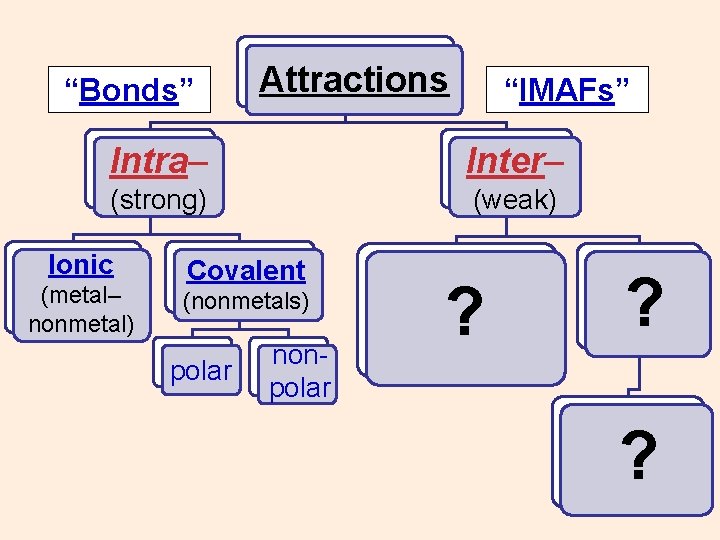
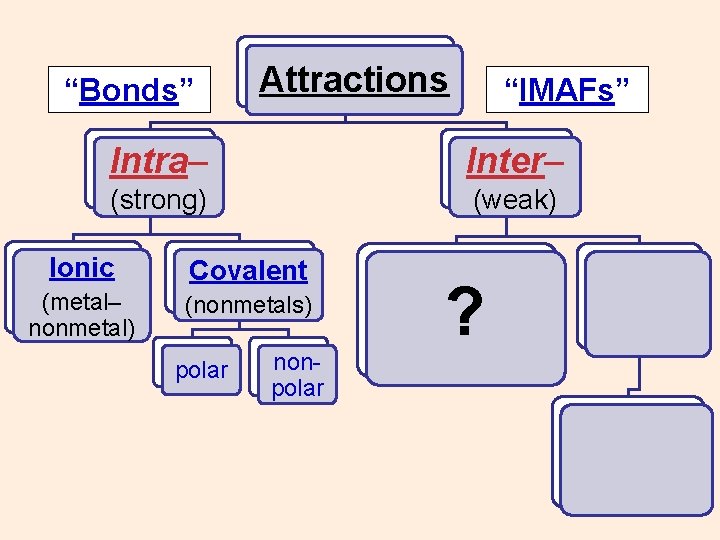
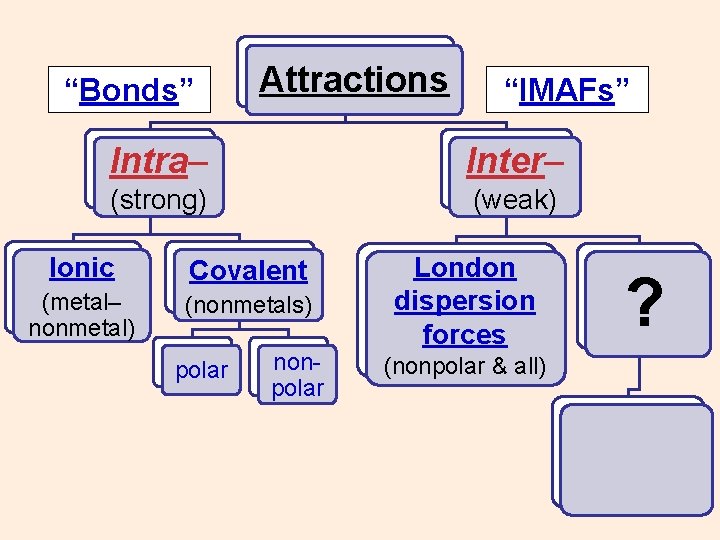
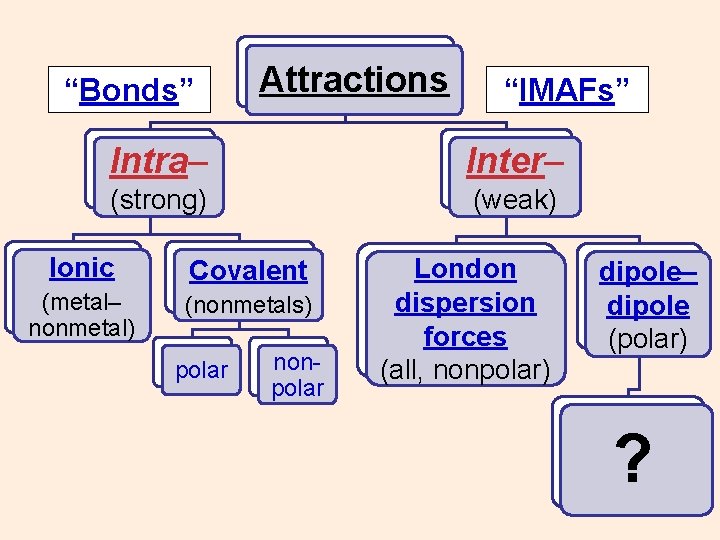
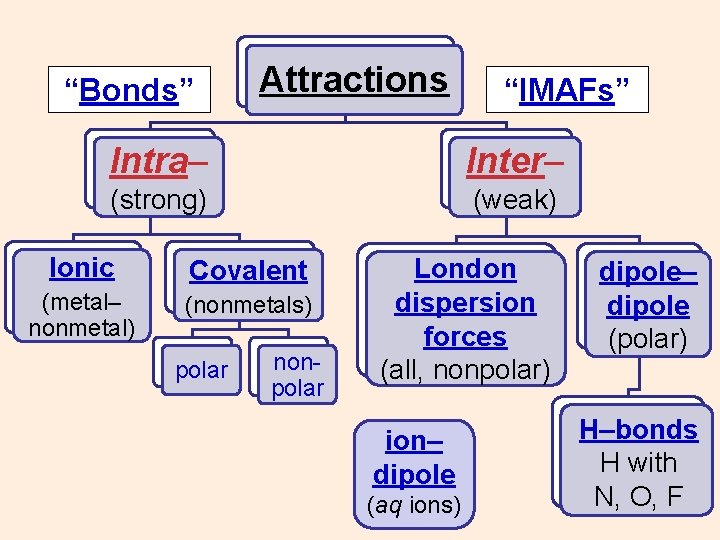
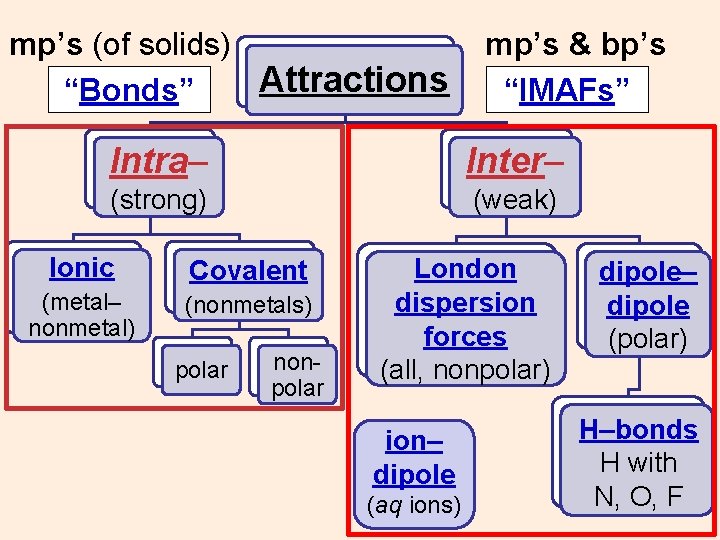
“Bonds” Attractions “IMAFs” Intra– Inter– (strong) (weak) Ionic (metal– nonmetal) Covalent (nonmetals) polar nonpolar ? ? ?

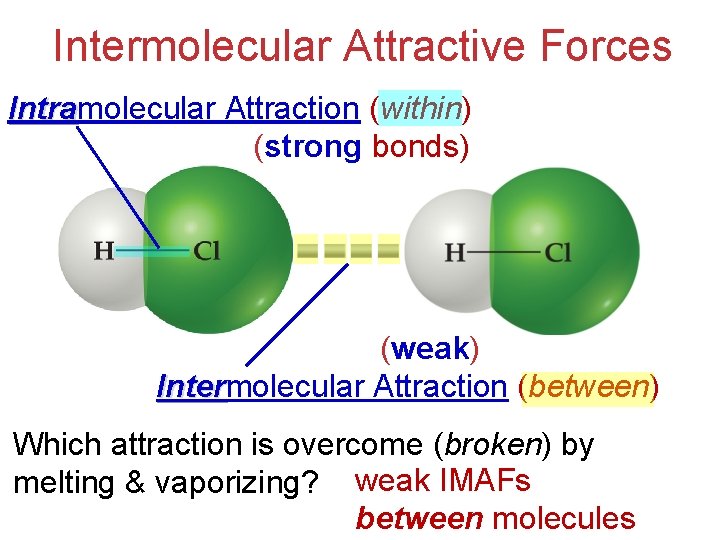
Intermolecular Attractive Forces Intramolecular Attraction (within) Intra (strong bonds) (weak) Intermolecular Attraction (between) Inter Which attraction is overcome (broken) by melting & vaporizing? weak IMAFs between molecules

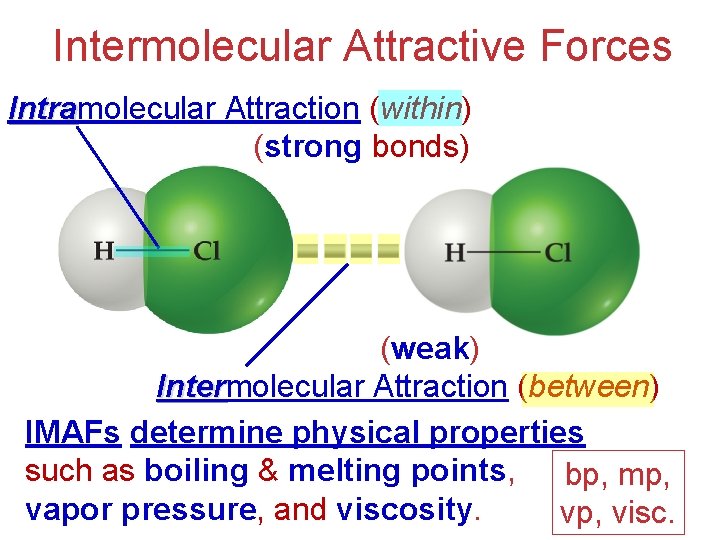
Intermolecular Attractive Forces Intramolecular Attraction (within) Intra (strong bonds) (weak) Intermolecular Attraction (between) Inter IMAFs determine physical properties such as boiling & melting points, bp, mp, vapor pressure, and viscosity. vp, visc.

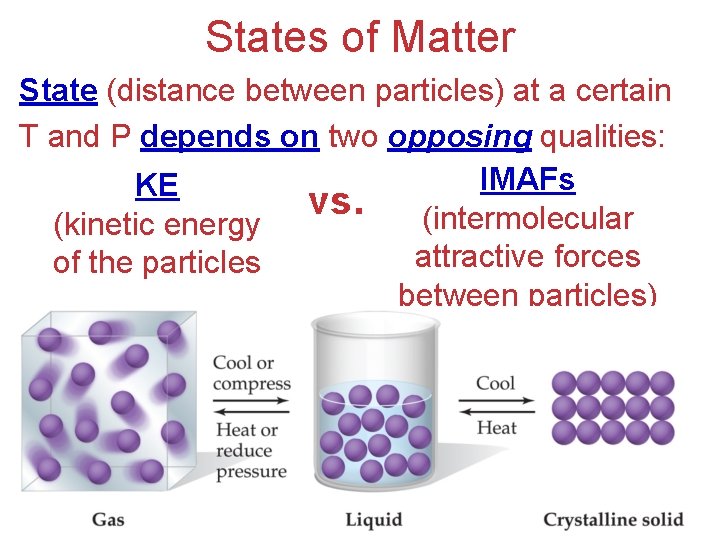
States of Matter The main difference between phases of matter is the distance between particles.

States of Matter State (distance between particles) at a certain T and P depends on two opposing qualities: IMAFs KE vs. (intermolecular (kinetic energy attractive forces of the particles between particles)

“Bonds” Attractions “IMAFs” Intra– Inter– (strong) (weak) Ionic Covalent (metal– nonmetal) (nonmetals) polar nonpolar ?

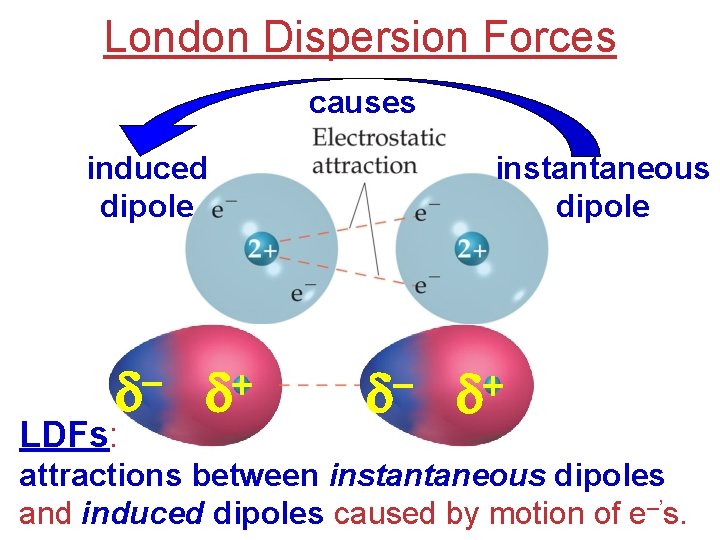
London Dispersion Forces Electrons in the 1 s orbital of He repel each other, BUT… they occasionally wind up on the same side of the atom. d + d At that instant, the He atom is polar (instantaneous dipole). (excess of e–’s on one side, and shortage on the other)

London Dispersion Forces causes induced dipole d LDFs: + d instantaneous dipole d + d attractions between instantaneous dipoles and induced dipoles caused by motion of e–’s.

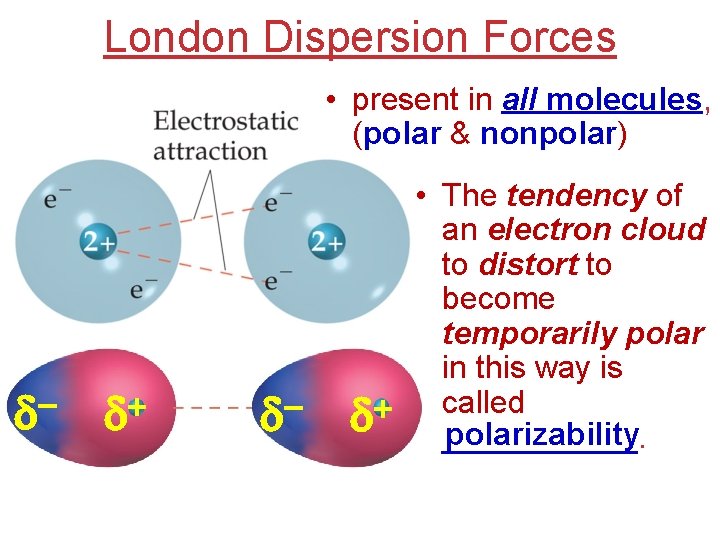
London Dispersion Forces • present in all molecules, (polar & nonpolar) d + d d • The tendency of an electron cloud to distort to become temporarily polar in this way is called + d ______. polarizability

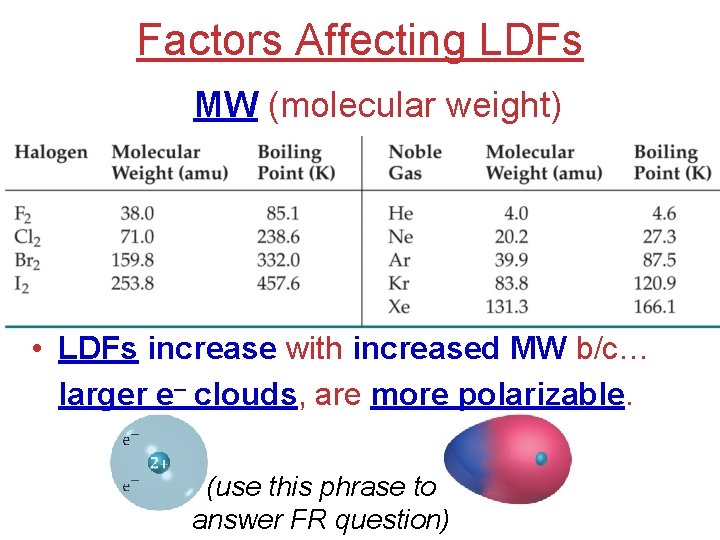
Factors Affecting LDFs MW (molecular weight) • LDFs increase with increased MW b/c… larger e– clouds, are more polarizable. (use this phrase to answer FR question)

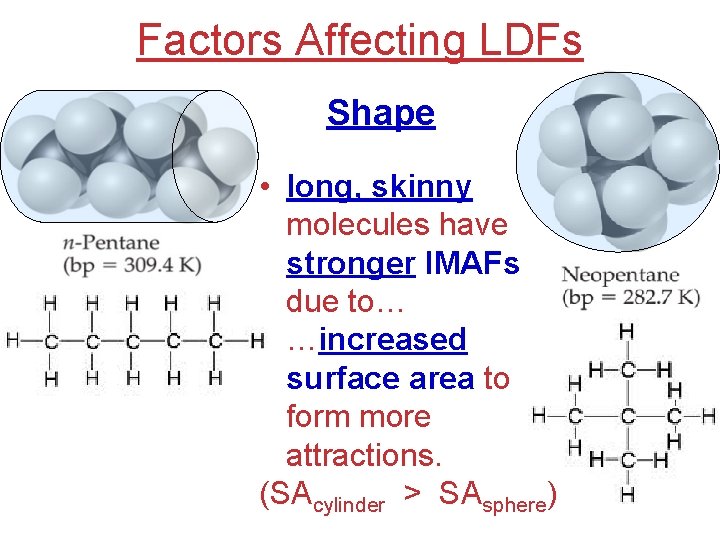
Factors Affecting LDFs Shape • long, skinny molecules have stronger IMAFs due to… …increased surface area to form more attractions. (SAcylinder > SAsphere)

“Bonds” Attractions “IMAFs” Intra– Inter– (strong) (weak) Ionic Covalent (metal– nonmetal) (nonmetals) polar nonpolar London dispersion forces (nonpolar & all) ?

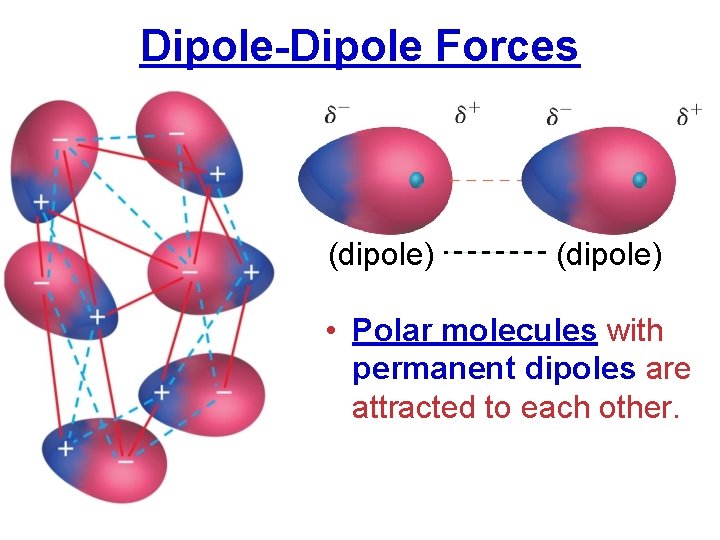
Dipole-Dipole Forces (dipole) • Polar molecules with permanent dipoles are attracted to each other.

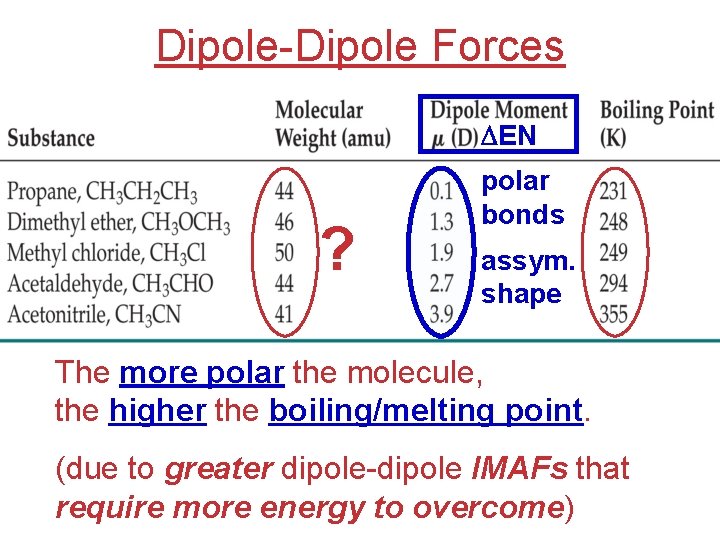
Dipole-Dipole Forces DEN ? polar bonds assym. shape The more polar the molecule, the higher the boiling/melting point. (due to greater dipole-dipole IMAFs that require more energy to overcome)

Which Have a Greater Effect: Dipole-Dipole Interactions or Dispersion Forces? • dipole–dipole forces (permanent dipoles) are typically stronger than LDFs (temporary dipoles as instantaneous–induced). • But much larger molecules could have LDFs that are stronger than dipole-dipole interactions. (larger e– clouds, are more polarizable)

“Bonds” Attractions “IMAFs” Intra– Inter– (strong) (weak) Ionic Covalent (metal– nonmetal) (nonmetals) polar nonpolar London dispersion forces (all, nonpolar) dipole– dipole (polar) ?

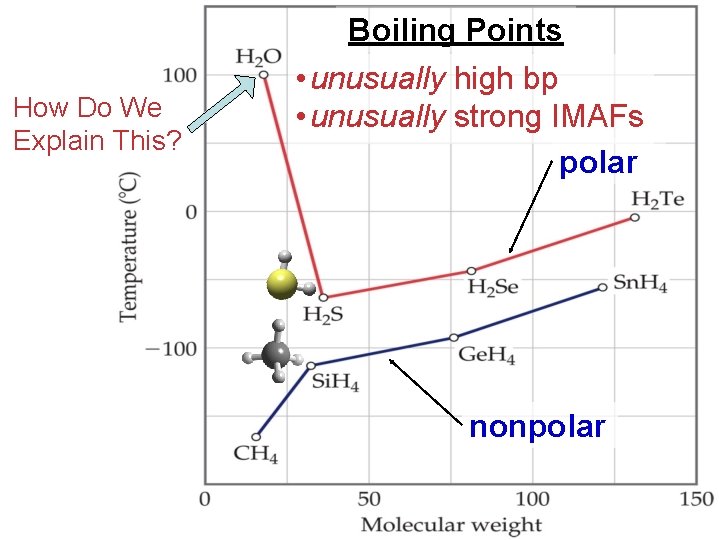
How Do We Explain This? Boiling Points • unusually high bp • unusually strong IMAFs polar nonpolar

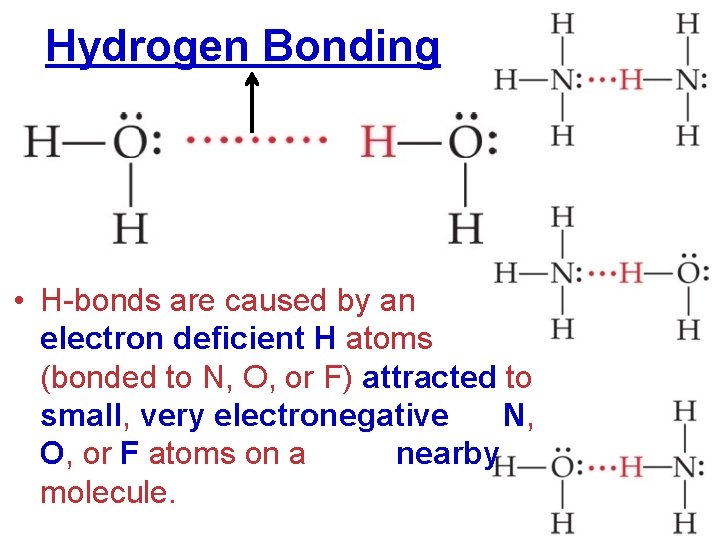
Hydrogen Bonding • H-bonds are caused by an electron deficient H atoms (bonded to N, O, or F) attracted to small, very electronegative N, O, or F atoms on a nearby molecule.

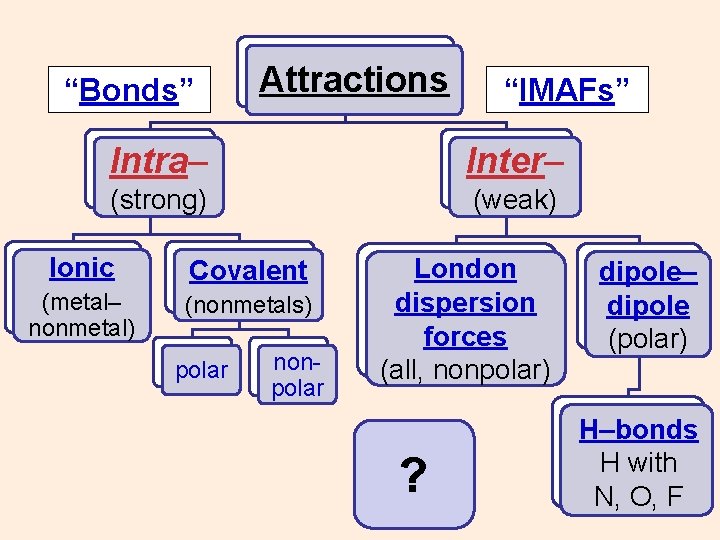
“Bonds” Attractions “IMAFs” Intra– Inter– (strong) (weak) Ionic Covalent (metal– nonmetal) (nonmetals) polar nonpolar London dispersion forces (all, nonpolar) ? dipole– dipole (polar) H–bonds H with N, O, F

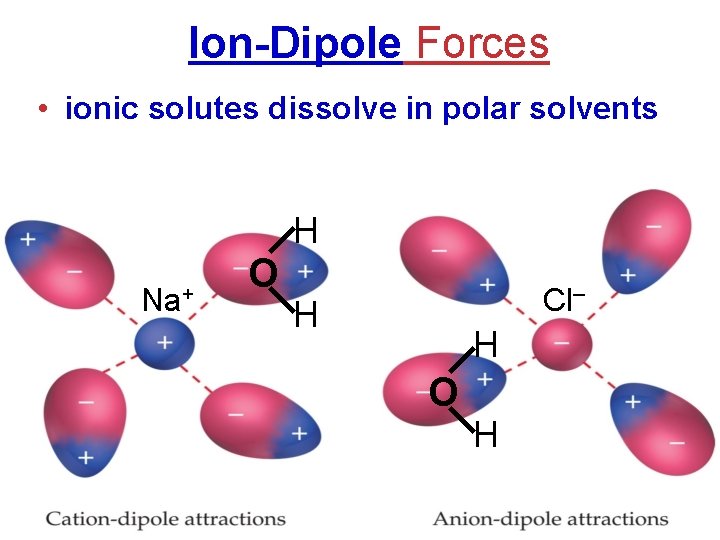
Ion-Dipole Forces • ionic solutes dissolve in polar solvents H Na+ O Cl– H H O H

“Bonds” Attractions “IMAFs” Intra– Inter– (strong) (weak) Ionic Covalent (metal– nonmetal) (nonmetals) polar nonpolar London dispersion forces (all, nonpolar) ion– dipole (aq ions) dipole– dipole (polar) H–bonds H with N, O, F

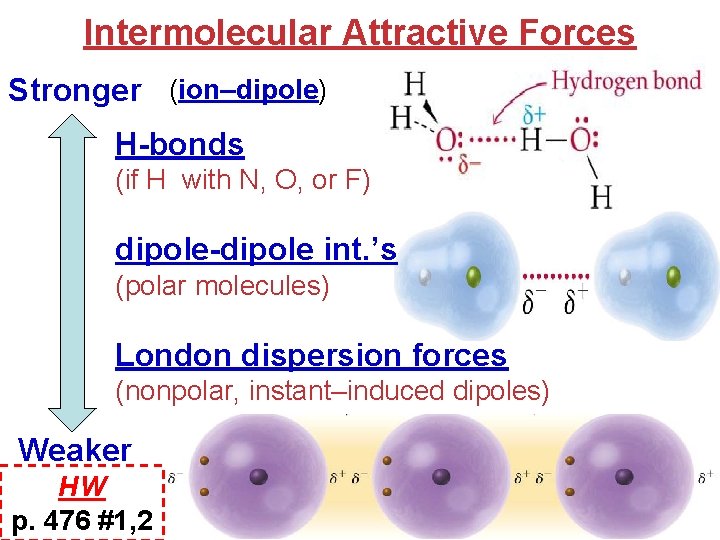
Intermolecular Attractive Forces Stronger (ion–dipole) H-bonds (if H with N, O, or F) dipole-dipole int. ’s (polar molecules) London dispersion forces (nonpolar, instant–induced dipoles) Weaker HW p. 476 #1, 2

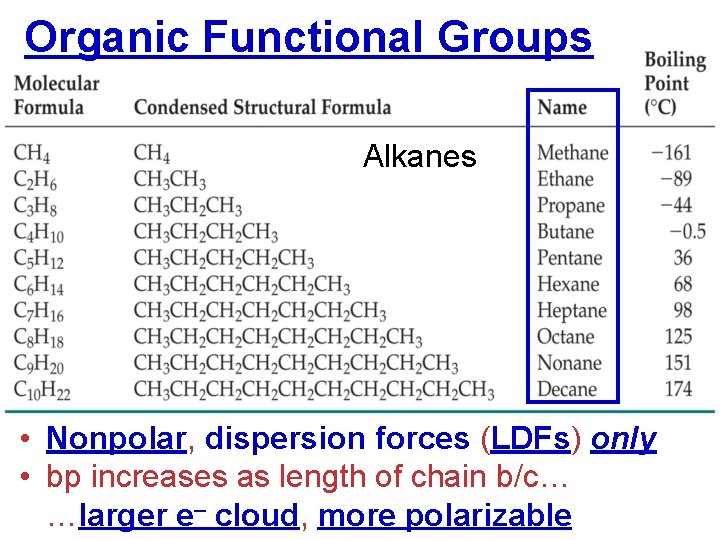
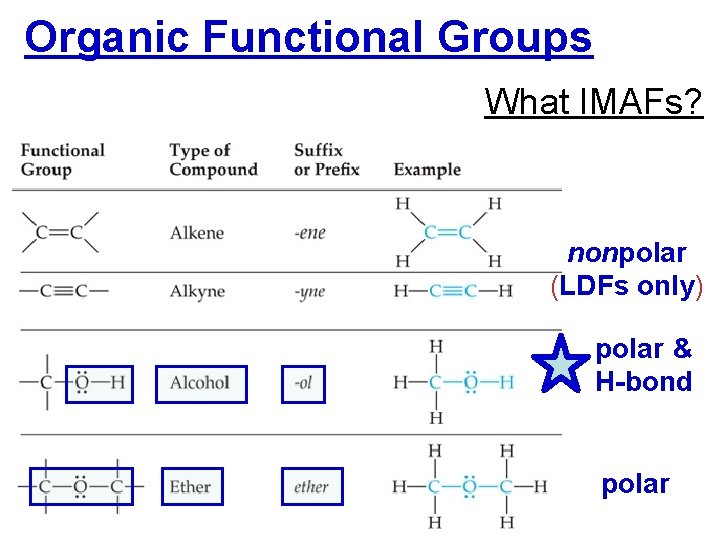
Organic Functional Groups Alkanes • Nonpolar, dispersion forces (LDFs) only • bp increases as length of chain b/c… …larger e– cloud, more polarizable

Organic Functional Groups What IMAFs? nonpolar (LDFs only) polar & H-bond polar

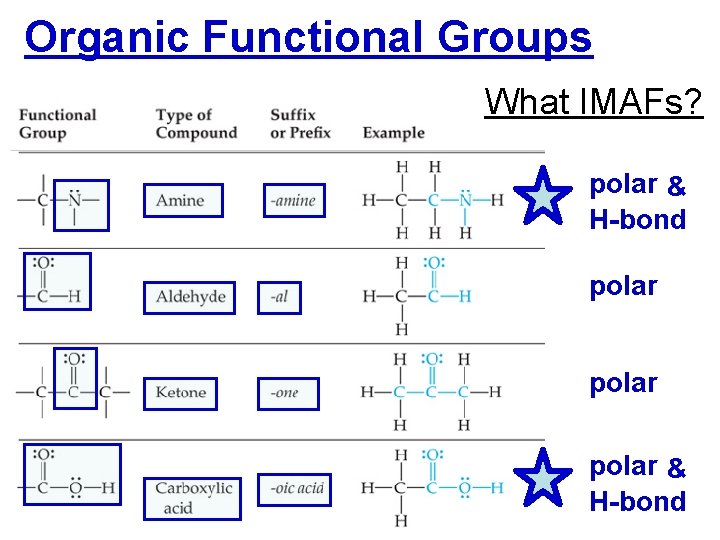
Organic Functional Groups What IMAFs? polar & H-bond polar & H-bond

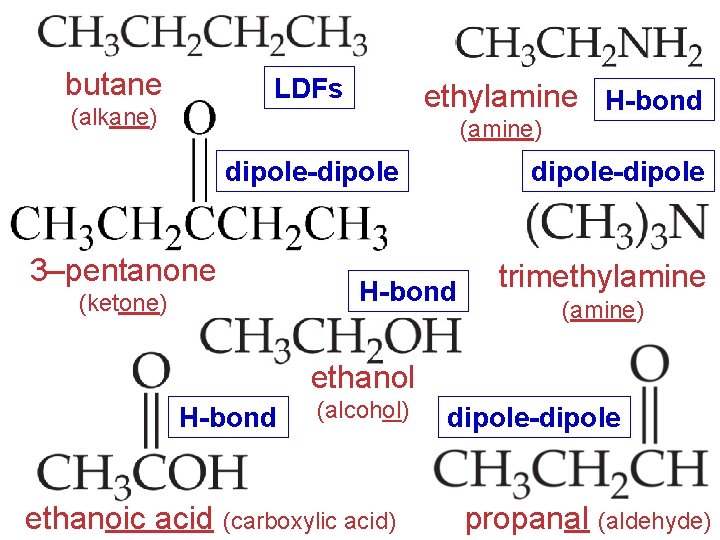
butane LDFs ethylamine H-bond (alkane) (amine) dipole-dipole 3–pentanone (ketone) dipole-dipole H-bond trimethylamine (amine) ethanol H-bond (alcohol) ethanoic acid (carboxylic acid) dipole-dipole propanal (aldehyde)

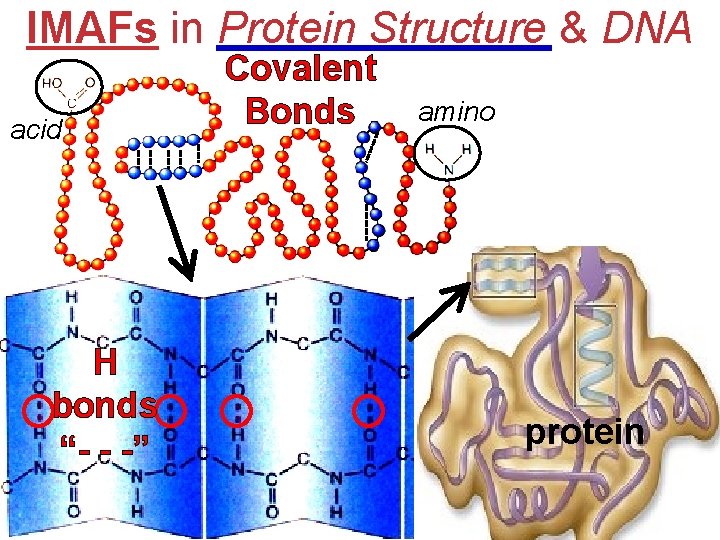
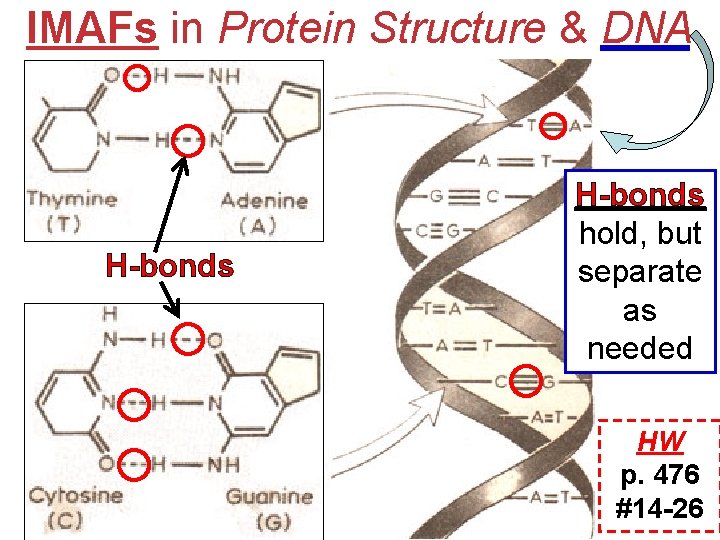
IMAFs in Protein Structure & DNA acid H bonds “- - -” Covalent Bonds amino protein

IMAFs in Protein Structure & DNA H-bonds hold, but separate as needed HW p. 476 #14 -26

Intermolecular Forces Affect Physical Properties The strength of the attractions between particles can greatly affect the physical properties of a substance or solution.

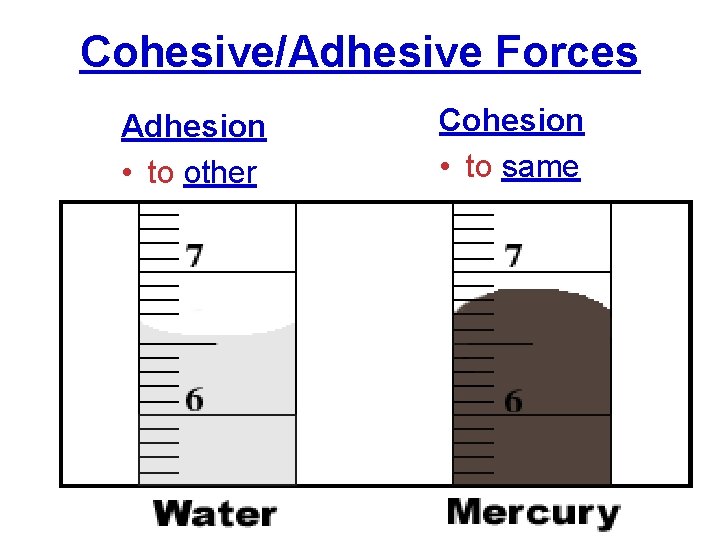
Cohesive/Adhesive Forces Adhesion • to other Cohesion • to same

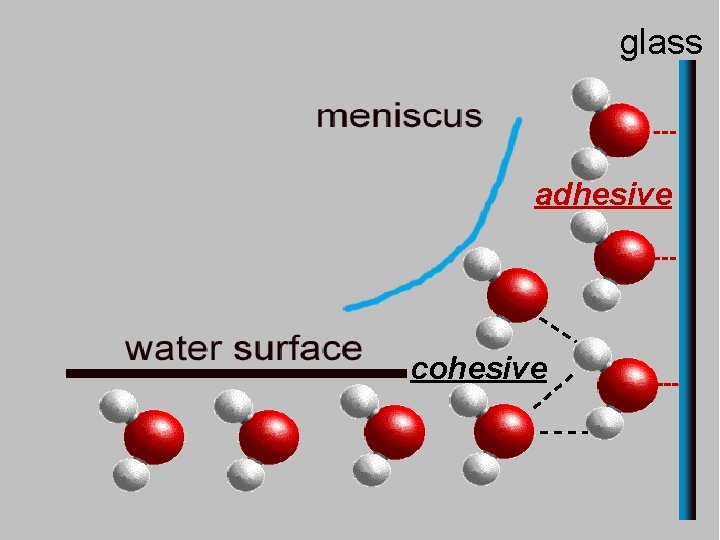
glass adhesive cohesive

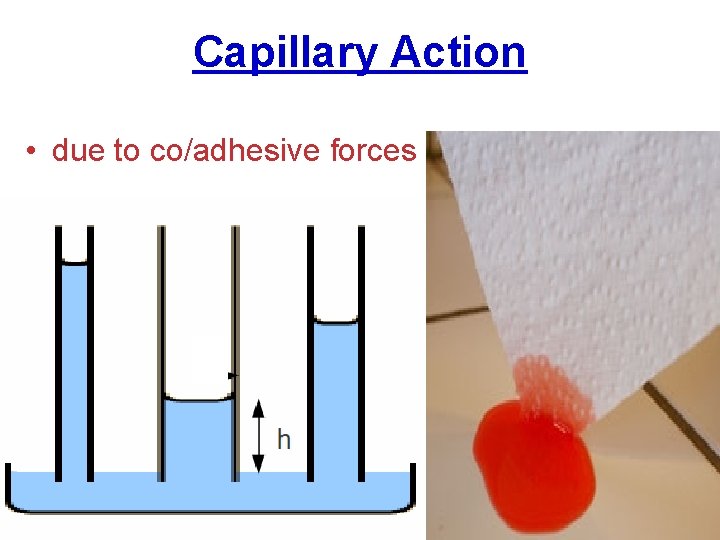
Capillary Action • due to co/adhesive forces

Surface Tension results from the net inward force experienced by the molecules on the surface of a liquid.

Viscosity • resistance of a liquid to flow • increases with IMAF’s and decreases with higher temp. HW p. 479 #29

Phase Changes HW p. 479 #34

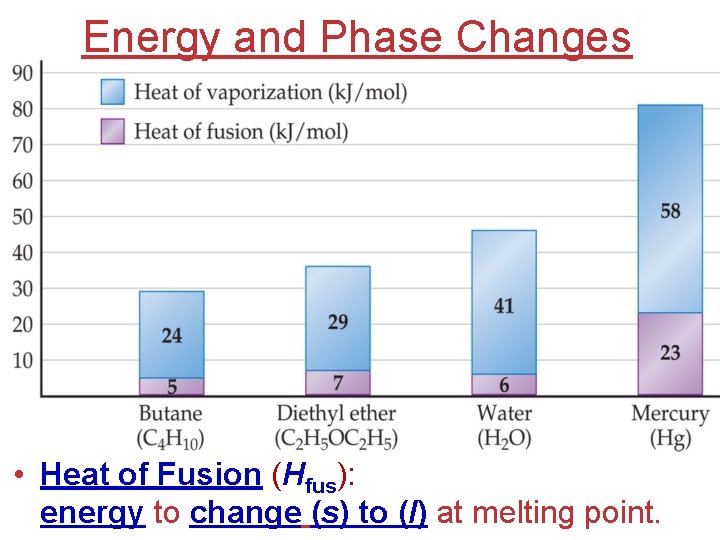
Energy and Phase Changes • Heat of Fusion (Hfus): energy to change (s) to (l) at melting point.

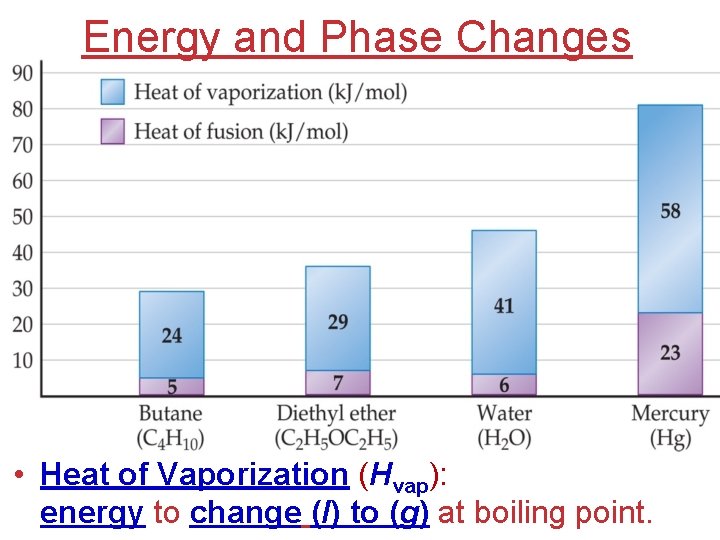
Energy and Phase Changes • Heat of Vaporization (Hvap): energy to change (l) to (g) at boiling point.

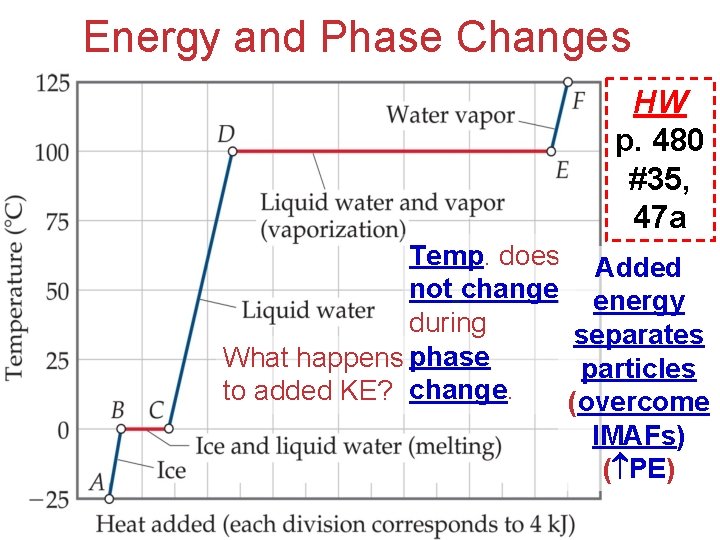
Energy and Phase Changes HW p. 480 #35, 47 a Temp. does Added not change energy during separates What happens phase particles to added KE? change. (overcome IMAFs) ( PE)

Vapor Pressure pressure above liquid more volatile = more vapor = _____ weak IMAFS liquid molecules escape to vapor gas molecules condense to liquid (dynamic equilibrium)

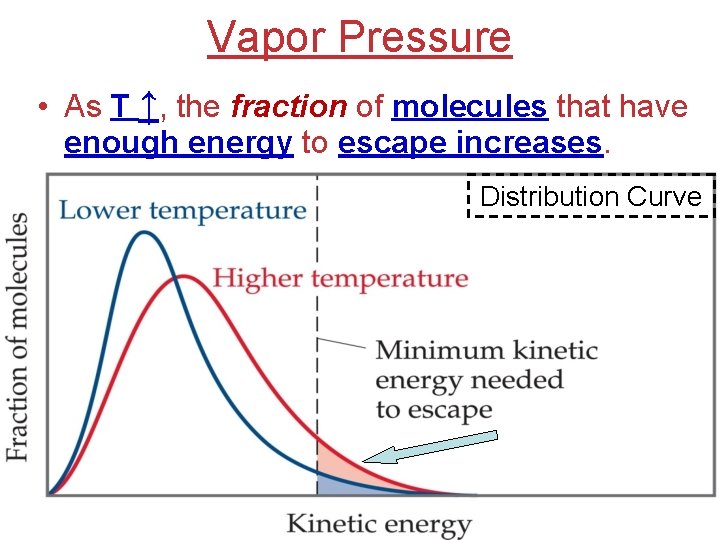
Vapor Pressure • As T ↑, the fraction of molecules that have enough energy to escape increases. Distribution Curve

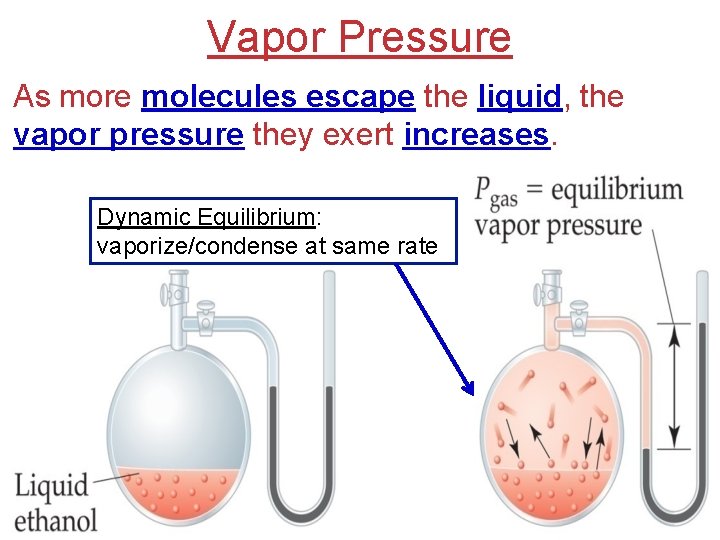
Vapor Pressure As more molecules escape the liquid, the vapor pressure they exert increases. Dynamic Equilibrium: vaporize/condense at same rate

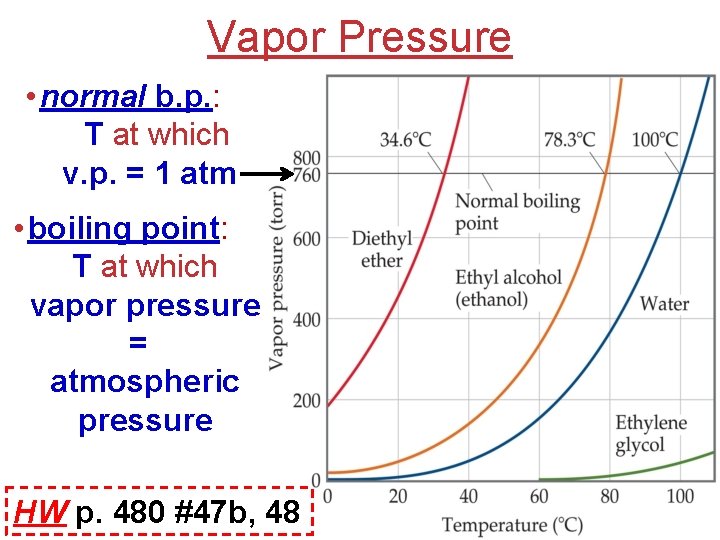
Vapor Pressure • normal b. p. : T at which v. p. = 1 atm • boiling point: T at which vapor pressure = atmospheric pressure HW p. 480 #47 b, 48

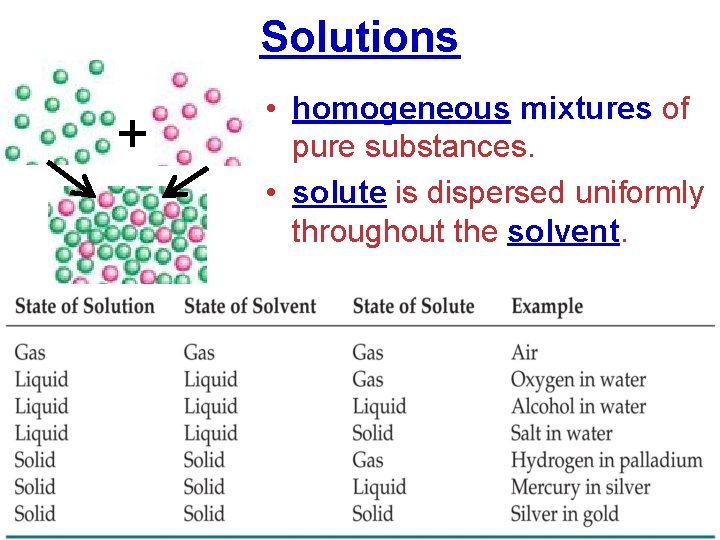
Solutions + • homogeneous mixtures of pure substances. • solute is dispersed uniformly throughout the solvent.

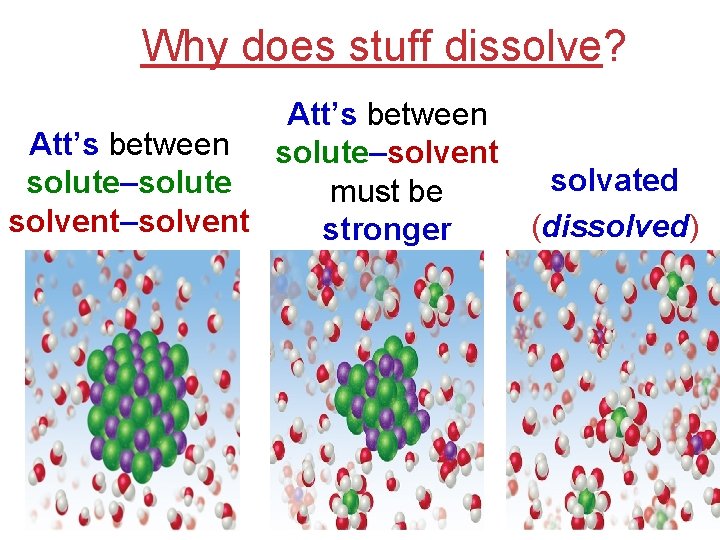
Why does stuff dissolve? Att’s between solute–solvent solvated solute–solute must be solvent–solvent (dissolved) stronger

For Ionic Solutes… Ions are soluble in water because ion-dipole attractions are strong enough to overcome the crystal lattice energy of the ionic solid salt.

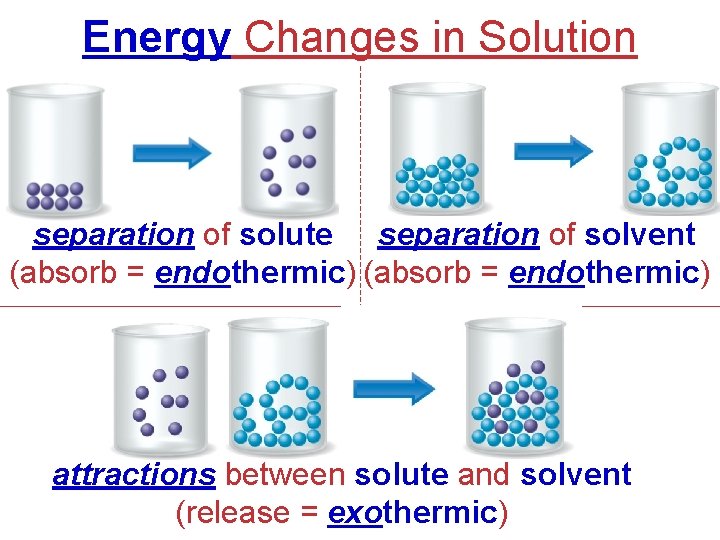
Energy Changes in Solution separation of solute separation of solvent (absorb = endothermic) attractions between solute and solvent (release = exothermic)

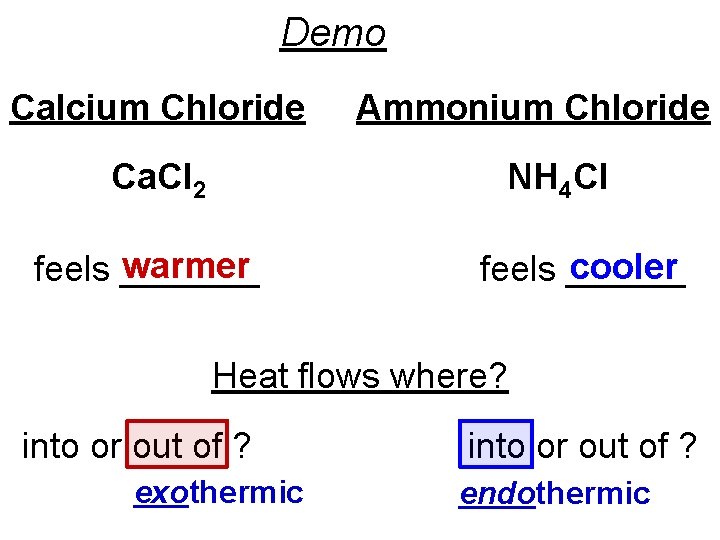
Demo Calcium Chloride Ca. Cl 2 Ammonium Chloride NH 4 Cl warmer feels _______ cooler feels ______ Heat flows where? into or out of ? exothermic into or out of ? endothermic

+ + H + – enthalpy – + (heat) – ∆H (final – initial) +

Why Do Endothermic Processes Occur? Usually favorable processes tend to lower energy. ∆E = – (exo or release) But in some processes, heat is absorbed, not released. How? + – + +

Why Endothermic Processes? Entropy (S): dispersal of matter & energy • increasing entropy (dispersal) (∆S = +) by mixing lowers the energy (∆E = –) of a system (even if ∆H = +). ∆S = (final – initial) (less dispersal) ∆S = (more – less) (more dispersal) ∆S = + 25 o. C +∆H (absorb heat)(+∆E) +∆S (gain entropy)(–∆E) 25 o. C –∆E +∆E(lowered) (raised) 10 o. C

Types of Solutions • Unsaturated ØLess than the maximum dissolved at that temperature.

Types of Solutions • Saturated Ødissolved maximum solute possible (at that temperature) ØDissolved solute in dynamic equilibrium with solid solute particles.

Types of Solutions • Supersaturated ØMore solute than is normally possible at that temperature. Øunstable; crystallization is stimulated by a “seed crystal” or scratching.

Factors Affecting Solubility • “like dissolves like”: (similar IMAFs) ØPolar substances dissolve in polar solvents. H 2 O CH 3 Cl CH 3 CH 2 OH NH 3 ØNonpolar dissolve in nonpolar solvents. C 6 H 14 CCl 4 I 2 Øsimilar IMAFs are more soluble.

HW p. 566 #12, 14, 18 Which is more soluble in water (H 2 O) and which is more soluble in hexane (C 6 H 14)

Chromatography? ? ?

Gases in Solution • The solubility of gases in water increases with increasing size. …WHY? • Larger e– clouds are more polarizable with stronger dispersion forces. • Stronger solute-solvent attractions = greater solubility

Gases in Solution Gases are more soluble at: High Pressure Low Temp Why keep sodas with a cap on and in the fridge?

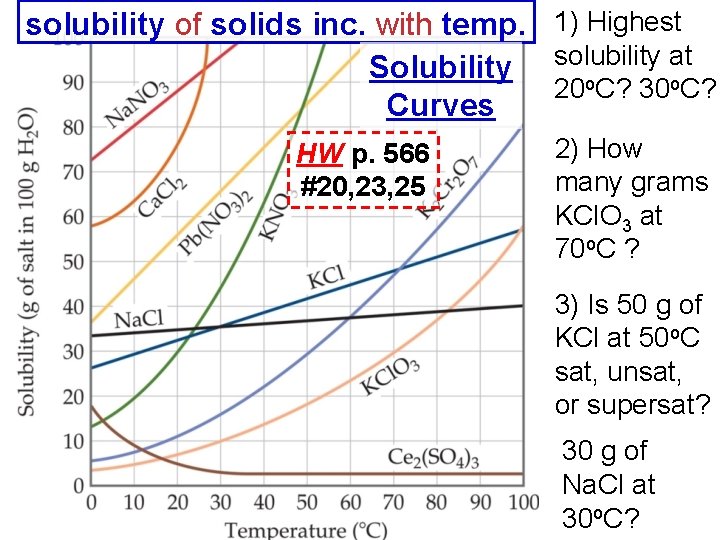
solubility of solids inc. with temp. 1) Highest at Solubility solubility 20 o. C? 30 o. C? Curves HW p. 566 #20, 23, 25 2) How many grams KCl. O 3 at 70 o. C ? 3) Is 50 g of KCl at 50 o. C sat, unsat, or supersat? 30 g of Na. Cl at 30 o. C?

Intermolecular Forces Affect Physical Properties The greater the IMAFs, … greater the bp and mp. the _______ greater the cohesion/adhesion. the _______ greater the surface tension. the _______ greater the viscosity. the _______ lower the vapor pressure the _______ (less volatile)

Intermolecular Forces Affect Physical Properties Which has a higher boiling point? Explain. CF 4 vs. CH 3 OCH 3 CF 4 has London dispersion forces(LDFs) and +1 CH 3 OCH 3 has dipole-dipole interactions(DDIs). The DDI’s in CH 3 OCH 3 are stronger and +1 require more energy to overcome resulting in a +1 higher bp. 3 -part answer for full credit

mp’s (of solids) Attractions “Bonds” mp’s & bp’s “IMAFs” Intra– Inter– (strong) (weak) Ionic Covalent (metal– nonmetal) (nonmetals) polar nonpolar London dispersion forces (all, nonpolar) ion– dipole (aq ions) dipole– dipole (polar) H–bonds H with N, O, F
![4 Types of Bonding in Solids Diamond ØCovalent nonmetals sharing es in mutual 4 Types of Bonding in Solids Diamond ØCovalent [nonmetals] • sharing e–’s in mutual](https://slidetodoc.com/presentation_image/932cf791ab8b2c86bc029dc6dd2152af/image-64.jpg)
4 Types of Bonding in Solids Diamond ØCovalent [nonmetals] • sharing e–’s in mutual nuclear attraction ØCovalent Network [C(d)] • shares e–’s throughout Quartz ØIonic [metal–nonmetal] • transfer e–’s forming attractive charged ions ØMetallic [metals] • metal atoms bonded by delocalized val. e–’s

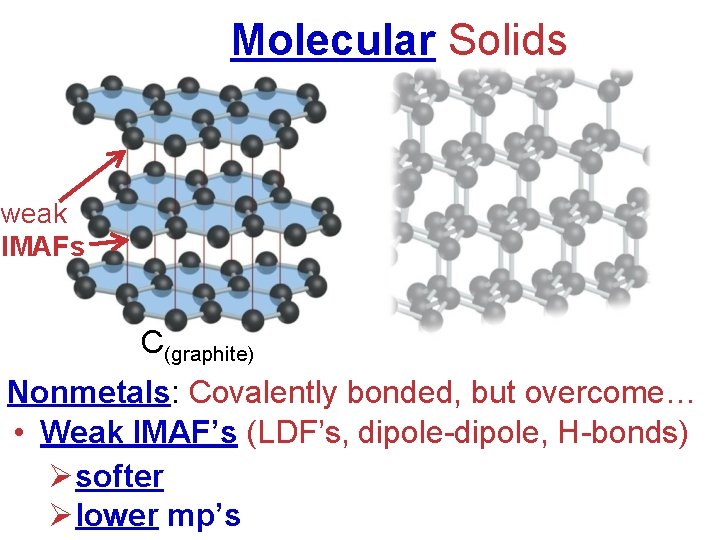
Molecular Solids weak IMAFs C(graphite) Nonmetals: Covalently bonded, but overcome… • Weak IMAF’s (LDF’s, dipole-dipole, H-bonds) Øsofter Ølower mp’s

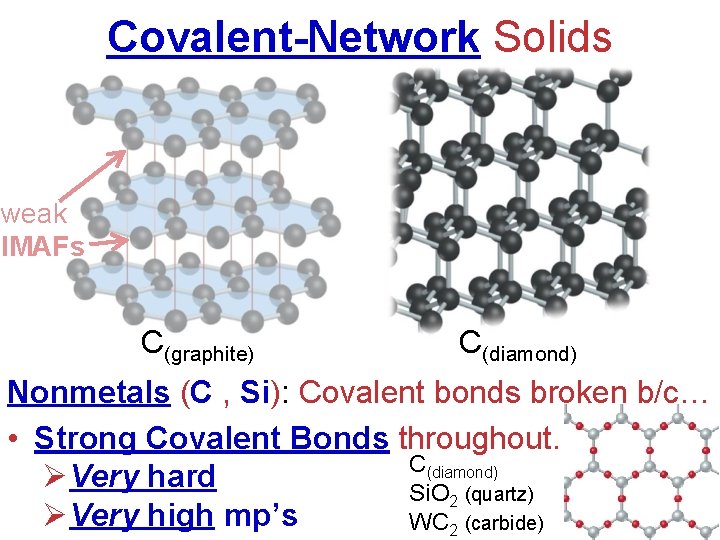
Covalent-Network Solids weak IMAFs C(graphite) C(diamond) Nonmetals (C , Si): Covalent bonds broken b/c… • Strong Covalent Bonds throughout. C(diamond) ØVery hard Si. O 2 (quartz) ØVery high mp’s WC 2 (carbide)

Ionic Solids more q less d stronger attraction more energy to break Metal-Nonmetal: Ionic bonds broken. • Strong Ionic Bonds in a crystal lattice of +/– ions bonded by Coulombic attraction. Øhard and brittle q 1 q 2 E= Øhigh mp’s d Øconduct only in solution(aq) or molten(l)

Metallic Solids • Metals are not covalently bonded, but attractions are too strong to be IMAFs. • Metallic Bonds Ø delocalized “sea” of valence electrons. Ø Excellent conductors Ø Malleable and Ductile (“smooshable”) Ø soft to very hard Ø low to very high m. p. ’s

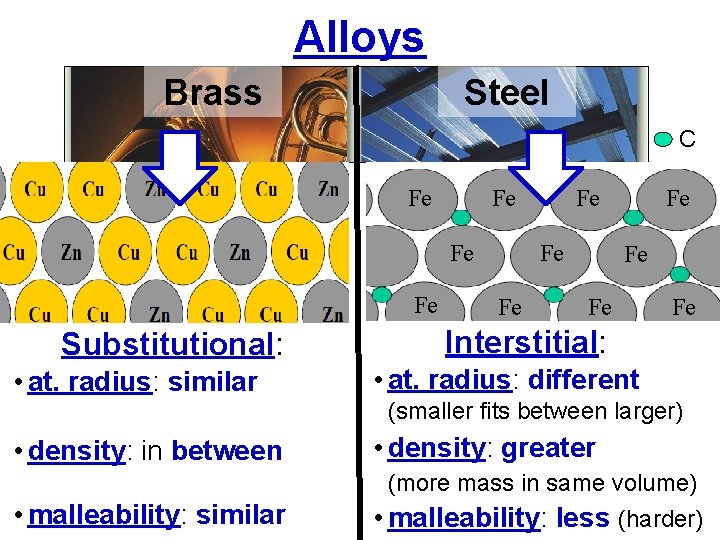
Alloys Brass Steel 67%Cu, 33%Zn 80%Fe, 0. 4%C, 18%Cr, 1%Ni Alloys: Homogeneous metallic mixtures (solutions). Attractions: metallic bonding (delocalized sea of valence e–’s)

Alloys Brass Steel C Fe Fe Substitutional: • at. radius: similar Fe Fe Interstitial: • at. radius: different (smaller fits between larger) • density: in between • density: greater (more mass in same volume) • malleability: similar • malleability: less (harder)

Attractions in Solids, Liquids, & Solutions Type Forces Between Particles Properties Intermolecular Attractions (IMAFs) Molecular London dispersion forces (nonpolar) Ar I 2 C 2 H 5 OH C(diamond) Si. O 2 WC 2 CO 2 H 2 O Dipole-dipole interactions (polar) C 11 H 22 O 11 Hydrogen bonds(H with N, O, F) d- d Covalent- Covalent Bonds (network) Network C(diamond) , Si. O 2 (quartz) , WC 2 (tungsten carbide) , etc… Ionic Bonds + qq Soft + Low mp & bp Poor conductor Very hard Very high mp Poor conductor Hard and brittle Ionic crystal lattice of High mp 1 2 charged ions Conducts as (aq) or (l) Soft to very hard Metallic Bonds All metals Metallic –’s Low to very high mp delocalized “sea” of val. e & alloys: Cu, Fe, K, Al, … Malleable, Ductile, Conductor Na. Cl Cu. SO 4 (l) + (l), Solutions (s) + (l) = (aq) d Solute-Solvent Attractions similar IMAFs or ion–dipole

IMAF or Bond Stronger with: Molecular (IMAFs) (nonmetals) Hydrogen bonds (if H with N , O , or F) greater ∆EN N<O<F Dipole-dipole interactions (polar molecules) greater ∆EN London dispersion forces (all molecules & nonpolar) instant/induced dipoles) Ionic Bonds (metal-nonmetal) (attractions between +/– ions) greater dipole moment larger e– cloud, more polarizable greater q, less d (Coulombic attraction)

Attractions Affect Physical Properties The Stronger the Attractions, the… higher mp of solid (and higher DHfus) harder solid higher bp of liquid (and higher DHvap) higher viscosity of liquid lower vapor pressure of liquid (less volatile) more soluble (similar solute-solvent IMAFs)

Reasoning with Concepts 1) Identify attractions (of both) (Bonds? IMAFs? ) 2) Compare strength + – 3) Connect to energy (endo/exo) and/or physical property