Chapter 3 Water Copyright 2005 Pearson Education Inc






















![H 2 O H+ + OH- At equilibrium [H+] = [OH-] = 10 -7 H 2 O H+ + OH- At equilibrium [H+] = [OH-] = 10 -7](https://slidetodoc.com/presentation_image_h2/698755588b1bd5fb983157eea4327152/image-27.jpg)


![LE 3 -8 p. H Scale 0 Increasingly Acidic [H+] > [OH–] 1 Neutral LE 3 -8 p. H Scale 0 Increasingly Acidic [H+] > [OH–] 1 Neutral](https://slidetodoc.com/presentation_image_h2/698755588b1bd5fb983157eea4327152/image-30.jpg)

- Slides: 31

Chapter 3 Water Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Overview: The Molecule That Supports All of Life • Water is the biological medium on Earth • All living organisms require water more than any other substance • Most cells are surrounded by water, and cells themselves are about 70 -95% water • The abundance of water is the main reason the Earth is habitable Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

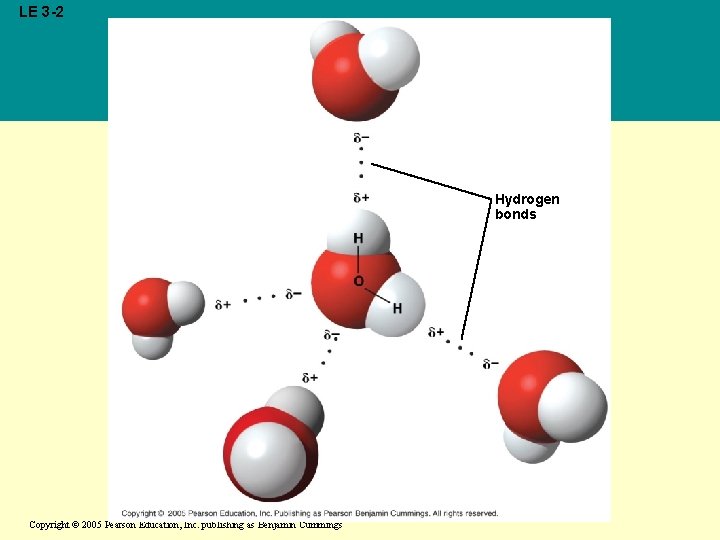
Concept 3. 1: The polarity of water molecules results in hydrogen bonding • The water molecule is a polar molecule: The opposite ends have opposite charges • Polarity allows water molecules to form hydrogen bonds with each other Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

LE 3 -2 Hydrogen bonds Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Concept 3. 2: Four emergent properties of water contribute to Earth’s fitness for life • Four of water’s properties that facilitate an environment for life: Cohesive behavior Ability to moderate temperature Expansion upon freezing Versatility as a solvent Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Cohesion • Collectively, hydrogen bonds hold water molecules together, a phenomenon called cohesion • Cohesion helps the transport of water against gravity in plants • Adhesion of water to plant cell walls also helps to counter gravity Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

LE 3 -3 Water-conducting cells 100 µm Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Surface tension is a measure of how hard it is to break the surface of a liquid • Surface tension is related to cohesion Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Water has High Specific Heat • The specific heat of a substance is the amount of heat that must be absorbed or lost for 1 gram of that substance to change its temperature by 1ºC • Water’s high specific heat minimizes temperature fluctuations –Heat is absorbed when hydrogen bonds break –Heat is released when hydrogen bonds form Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Moderation of Temperature • Water absorbs heat from warmer air and releases stored heat to cooler air • Because of its high specific heat, water can absorb or release a large amount of heat with only a slight change in its own temperature Thus water moderates climate Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Evaporative Cooling • Evaporation is transformation of a substance from liquid to gas • Heat of vaporization is the heat a liquid must absorb for 1 gram to be converted to gas • As a liquid evaporates, its remaining surface cools, a process called evaporative cooling • Because of the H-bonds, water has high heat of vaporization, making it a very efficient cooler of organisms and bodies of water Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Insulation of Bodies of Water by Floating Ice Most substances contract when cooled, and so does water, down to 4 C; then it expands again. So the bottom of the ocean is never below 4 C Upon freezing, water expands further, and ice is less dense than water • Ice floats in liquid water because hydrogen bonds in ice are more “ordered, ” making ice less dense • If ice sank, all bodies of water would eventually freeze solid, making life impossible on Earth Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Solvent of Life • A solution is a liquid that is a homogeneous mixture of substances • A solvent is the dissolving agent of a solution • The solute is the substance that is dissolved • Water is a versatile solvent due to its polarity • An aqueous solution is one in which water is the solvent Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

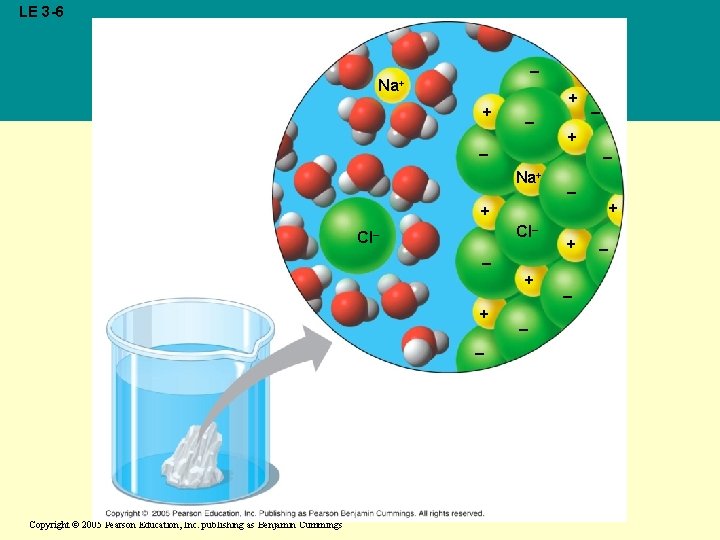
• Water is an effective solvent because it readily forms hydrogen bonds • When an ionic compound is dissolved in water, each ion is surrounded by a sphere of water molecules, a hydration shell Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

LE 3 -6 – Na+ + + – – – + – Na+ – + + Cl– – + – Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings + – –

• Water can also dissolve compounds made of nonionic polar molecules • Even large polar molecules such as proteins can dissolve in water if they have ionic and polar regions Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

LE 3 -7 a Lysozyme molecule in a nonaqueous environment. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

LE 3 -7 b Lysozyme molecule in a aqueous environment. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Hydrophilic and Hydrophobic Substances • A hydrophilic substance is one that has an affinity for water • A hydrophobic substance is one that does not have an affinity for water Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Solute Concentration in Aqueous Solutions • Most biochemical reactions occur in water • Chemical reactions depend on collisions of molecules and therefore on the concentration of solutes in an aqueous solution Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Molecular mass is the sum of all masses of all atoms in a molecule • Mole: amount of compound where grams = molecular mass • One mole has 6. 02 x 1023 molecules • Molarity is the number of moles of solute per liter of solution Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

What is the molarity of water? Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

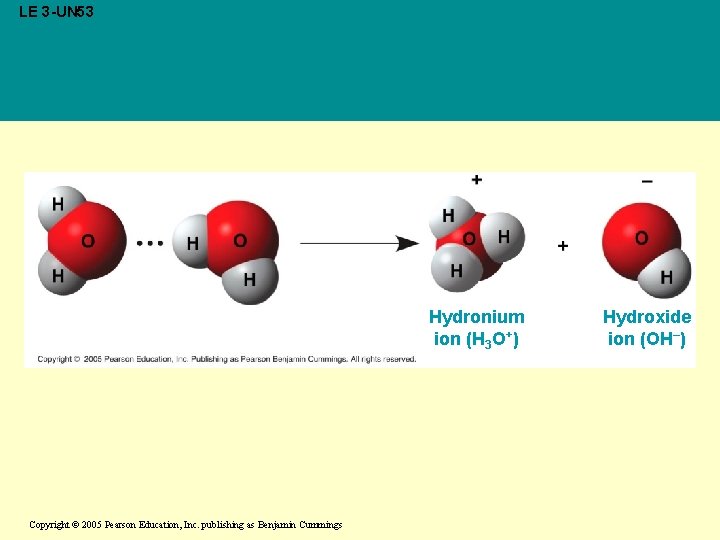
Concept 3. 3: Dissociation of water molecules leads to acidic and basic conditions that affect living organisms • A hydrogen atom in a hydrogen bond between two water molecules can shift from one to the other: – The hydrogen atom leaves its electron behind and is transferred as a proton, or hydrogen ion (H+) – The molecule with the extra proton is now a hydronium ion (H 3 O+) – The molecule that lost the proton is now a hydroxide ion (OH-) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

LE 3 -UN 53 Hydronium ion (H 3 O+) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Hydroxide ion (OH–)
![H 2 O H OH At equilibrium H OH 10 7 H 2 O H+ + OH- At equilibrium [H+] = [OH-] = 10 -7](https://slidetodoc.com/presentation_image_h2/698755588b1bd5fb983157eea4327152/image-27.jpg)
H 2 O H+ + OH- At equilibrium [H+] = [OH-] = 10 -7 M (neutral) Most of H 2 O is not dissociated p. H = -log[H+] Neutral p. H = ? ? Acidic p. H: [H+]>[OH-]; basic p. H: [H+]<[OH-]; (>10 -7); p. H ? (<10 -7); p. H ? p. H<7 p. H>7 Life likes p. H 6 -8 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Effects of changes in p. H • Concentrations of H+ and OH- are equal in pure water • Adding certain solutes, called acids and bases, modifies the concentrations of H+ and OH • Biologists use something called the p. H scale to describe how acidic or basic (the opposite of acidic) a solution is Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Acids and Bases • An acid is any substance that increases the H+ concentration of a solution • A base is any substance that reduces the H+ concentration of a solution Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
![LE 3 8 p H Scale 0 Increasingly Acidic H OH 1 Neutral LE 3 -8 p. H Scale 0 Increasingly Acidic [H+] > [OH–] 1 Neutral](https://slidetodoc.com/presentation_image_h2/698755588b1bd5fb983157eea4327152/image-30.jpg)
LE 3 -8 p. H Scale 0 Increasingly Acidic [H+] > [OH–] 1 Neutral [H+] = [OH–] Battery acid 2 Digestive (stomach) juice, lemon juice 3 Vinegar, beer, wine, cola 4 Tomato juice 5 Black coffee Rainwater 6 Urine 7 Pure water Human blood 8 Increasingly Basic [H+] < [OH–] Seawater 9 10 Milk of magnesia 11 Household ammonia 12 13 14 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Household bleach Oven cleaner

Buffers • The internal p. H of most living cells must remain close to p. H 7 • Buffers are substances that minimize changes in concentrations of H+ and OH- in a solution • Most buffers consist of an acid-base pair that reversibly combines with H+ Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings