Chapter 13 Effector Mechanisms of Humoral Immunity Effector











- Slides: 33

Chapter 13 Effector Mechanisms of Humoral Immunity



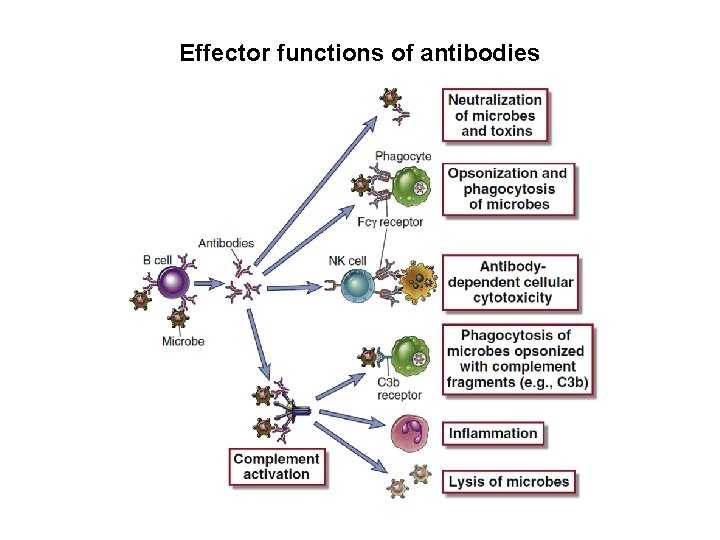
Effector functions of antibodies


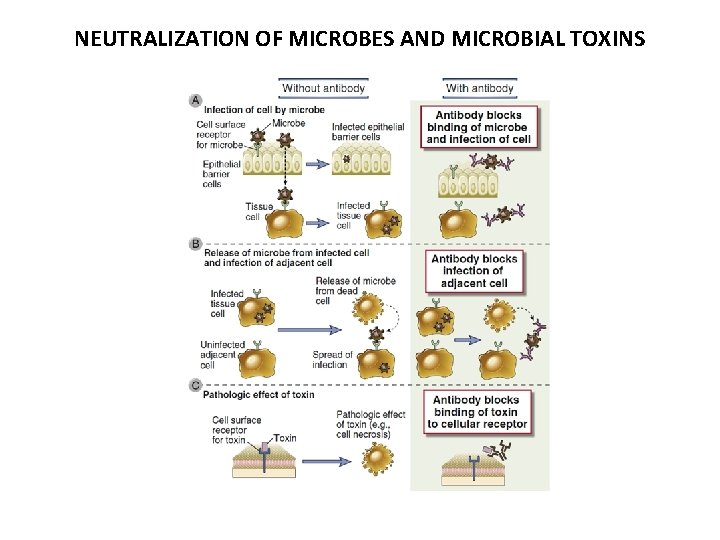
NEUTRALIZATION OF MICROBES AND MICROBIAL TOXINS

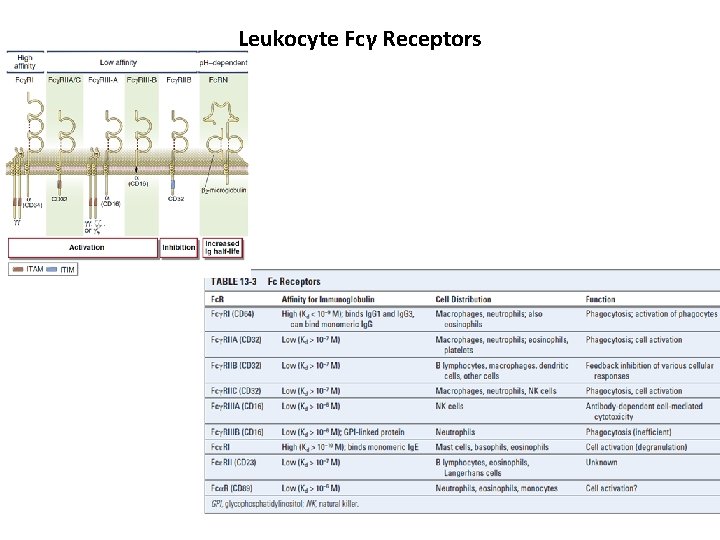
Leukocyte Fcγ Receptors

Inhibitory Signaling by the FcγRIIB Receptor • Immune complex–mediated cross-linking of the inhibitory FcγRIIB leads to tyrosine phosphorylation of the ITIM in the cytoplasmic tail • One somewhat empirical but often useful treatment of many autoimmune diseases is the intravenous administration of pooled human Ig. G (IVIG). IVIG may engage FcγRIIB to deliver inhibitory signals to B lymphocytes and other cells, thus reducing antibody production and dampening inflammation

Role of Fcγ Receptors in Phagocytosis and Activation of Phagocytes

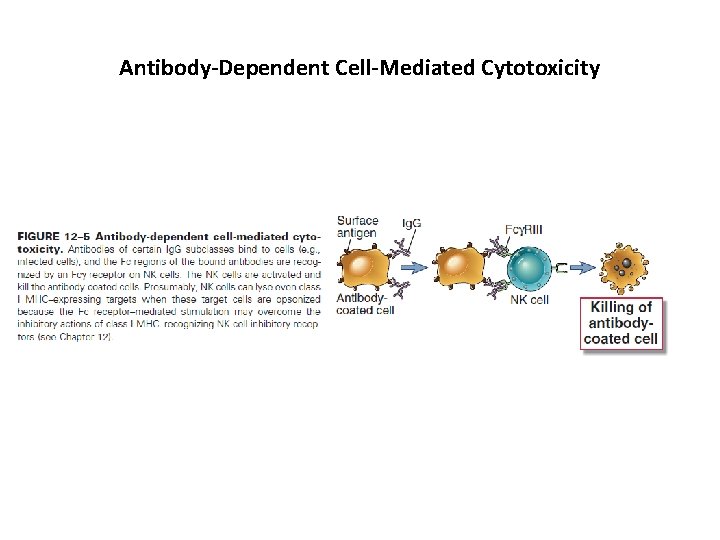
Antibody-Dependent Cell-Mediated Cytotoxicity

Antibody-Mediated Clearance of Helminths • Antibodies, mast cells, and eosinophils function with antibodies to mediate the expulsion and killing of some helminthic parasites • Ig. E, Ig. G, and Ig. A antibodies that coat helminths can bind to Fc receptors on eosinophils and cause the degranulation of these cells, releasing the basic protein and other eosinophil granule contents that kill the parasites • The high affinity Fcε receptor of eosinophils (FcεRI) lacks the signaling β chain and can only signal relatively weakly through the associated γ chain

THE COMPLEMENT SYSTEM

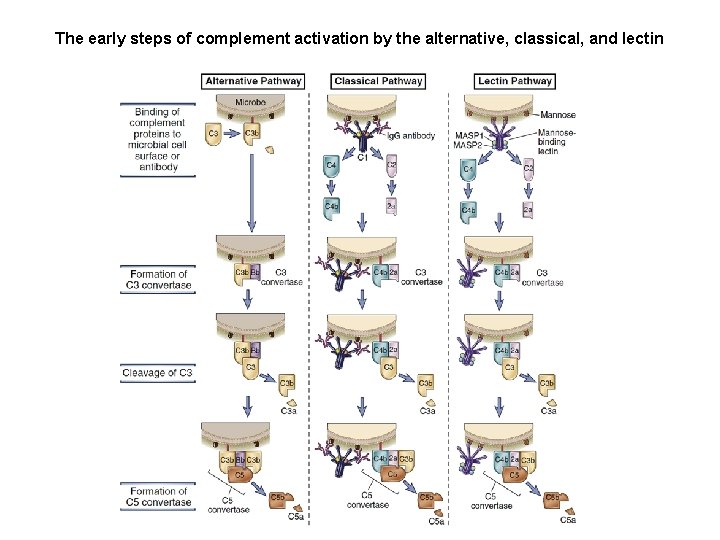
The early steps of complement activation by the alternative, classical, and lectin pathways

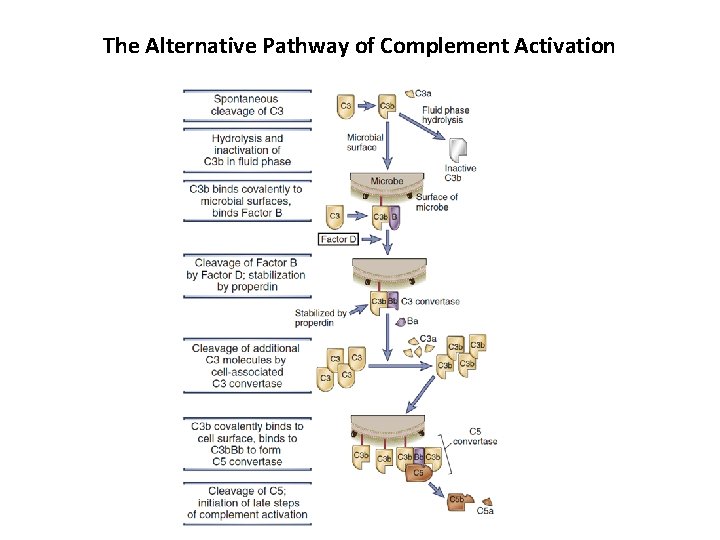
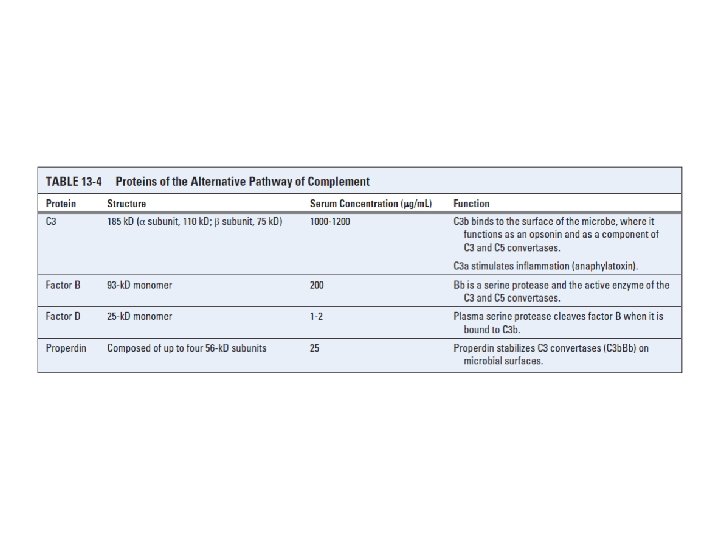
The Alternative Pathway of Complement Activation

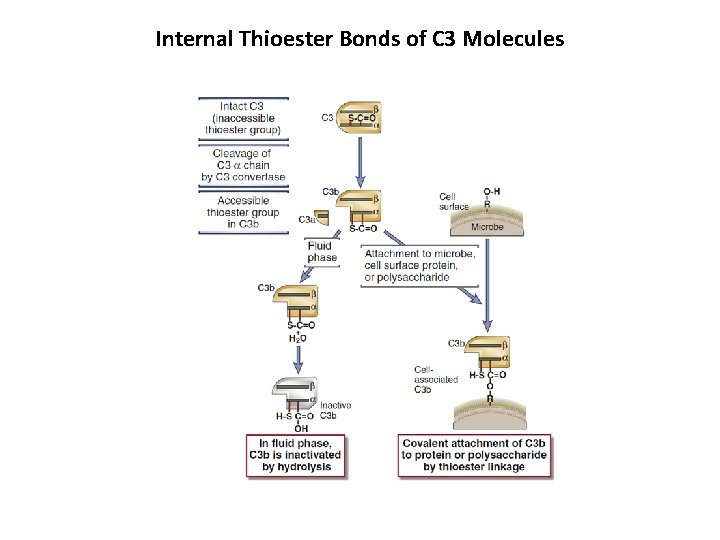
Internal Thioester Bonds of C 3 Molecules


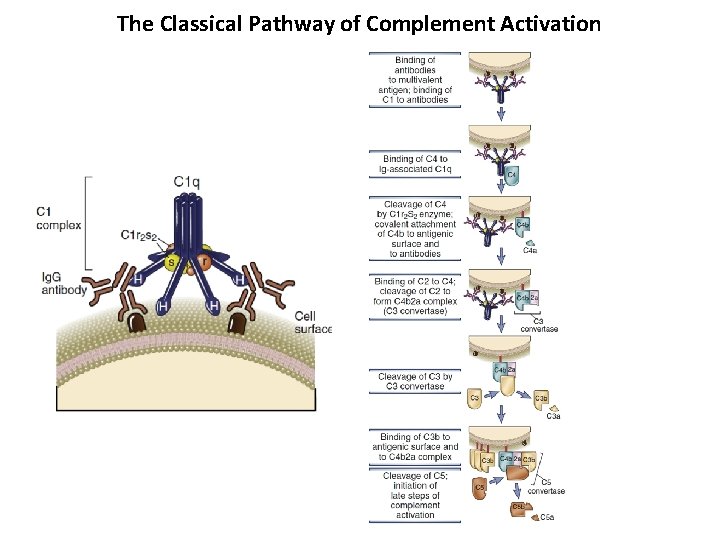
The Classical Pathway of Complement Activation

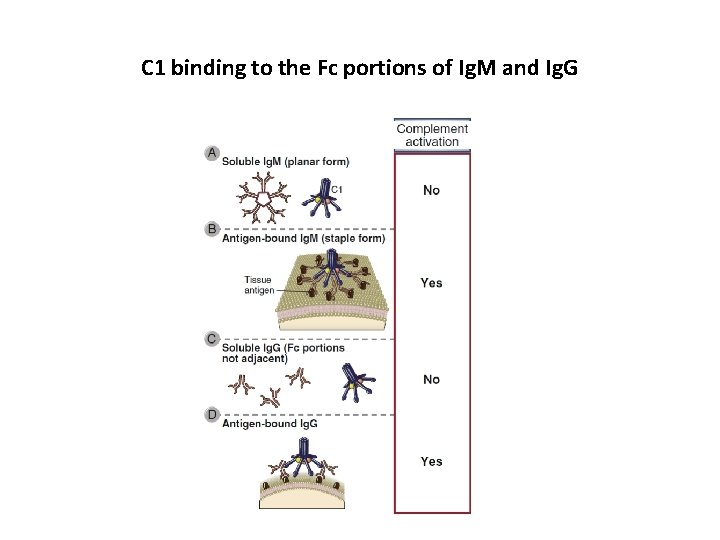
C 1 binding to the Fc portions of Ig. M and Ig. G

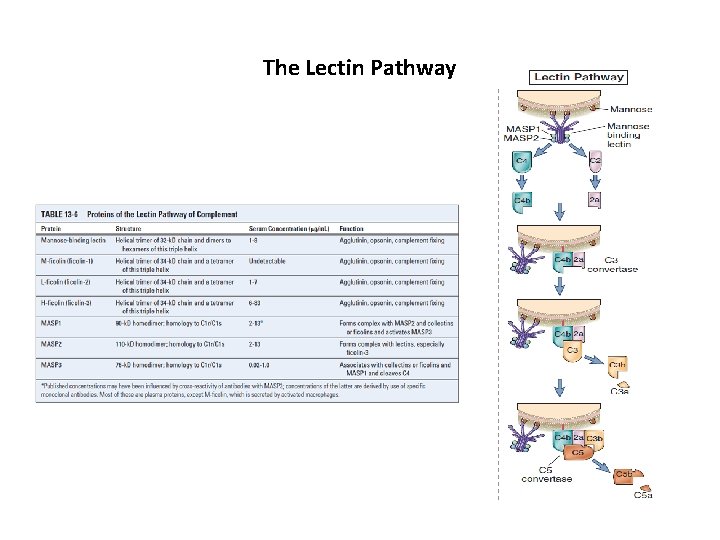
The Lectin Pathway

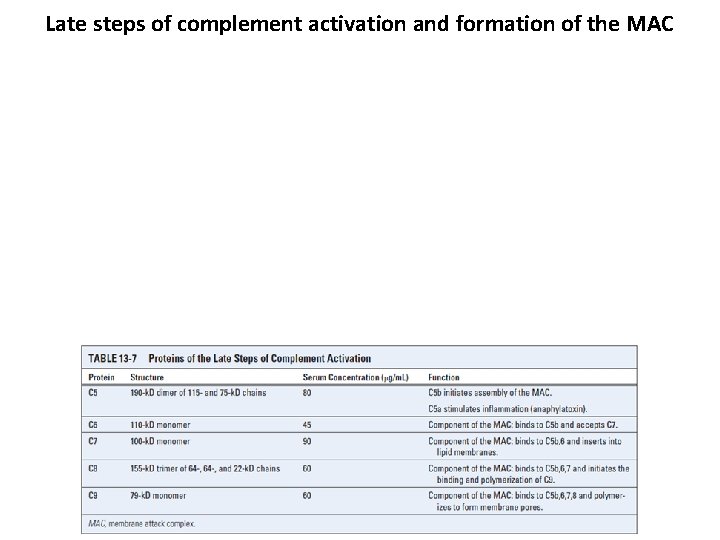
Late steps of complement activation and formation of the MAC

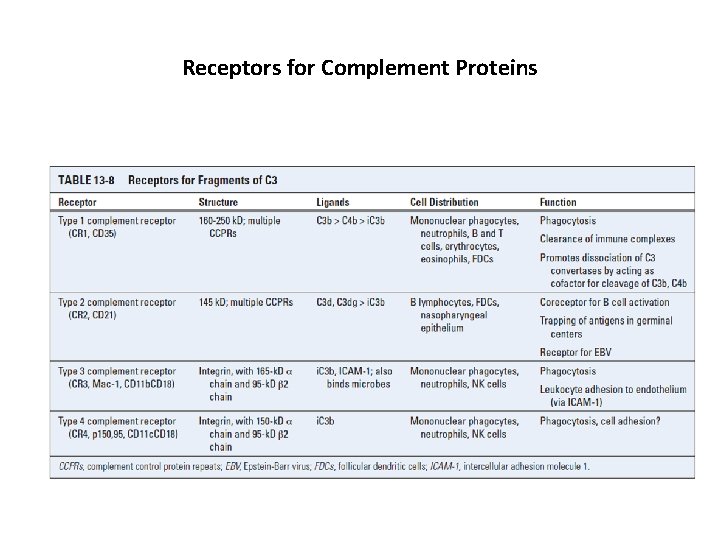
Receptors for Complement Proteins

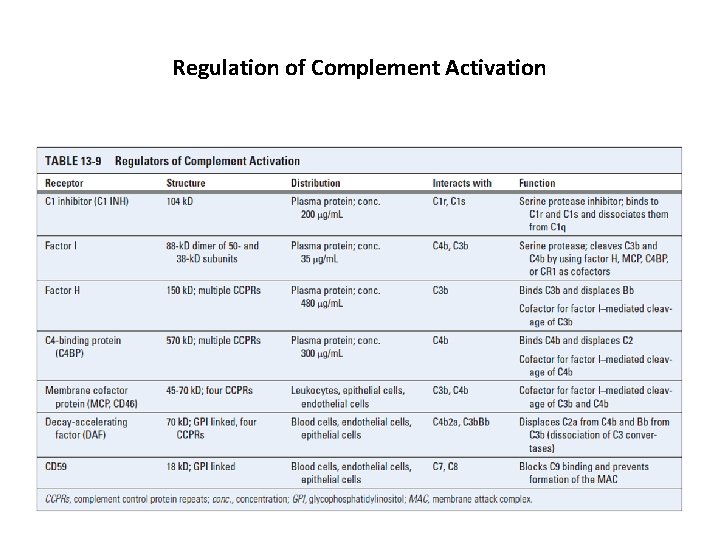
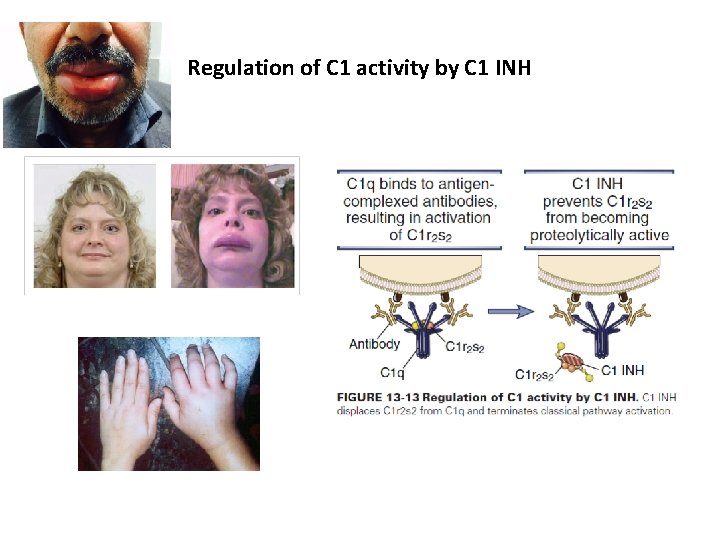
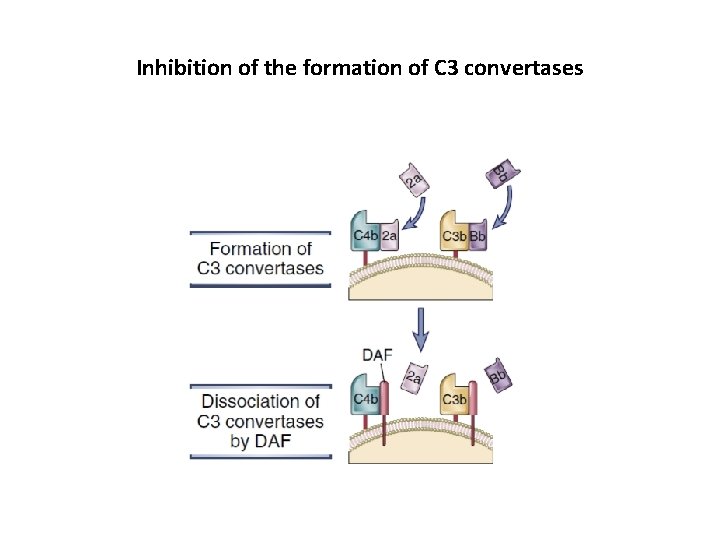
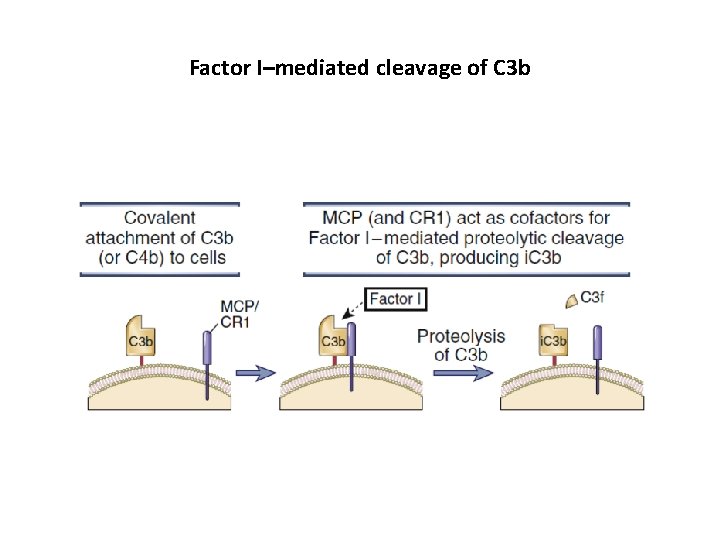
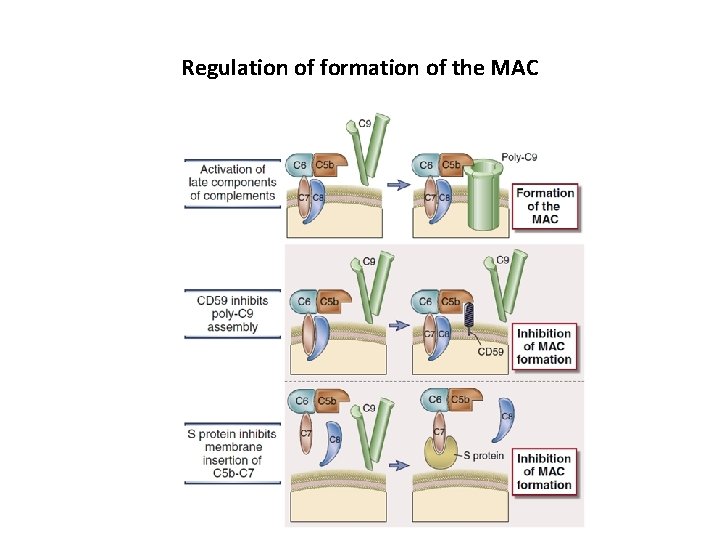
Regulation of Complement Activation

Regulation of C 1 activity by C 1 INH

Inhibition of the formation of C 3 convertases

Factor I–mediated cleavage of C 3 b

Regulation of formation of the MAC

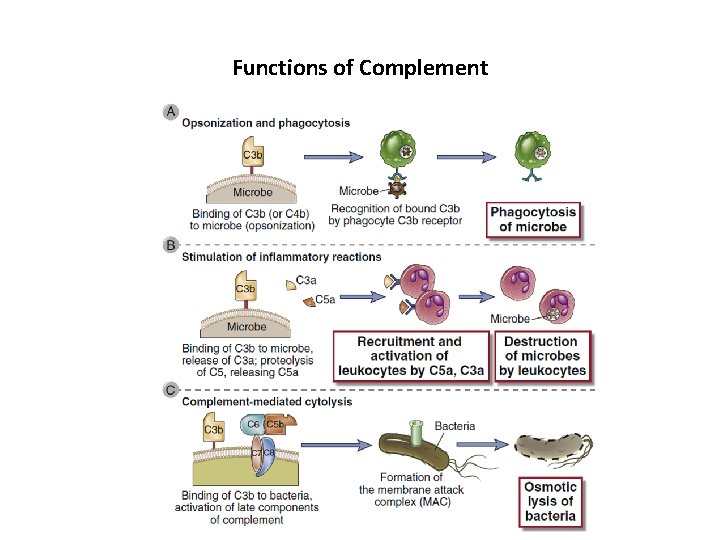
Functions of Complement

Complement Deficiencies • Genetic deficiencies in classical pathway components, including C 1 q, C 1 r, C 4, C 2, and C 3; C 2 deficiency is the most common human complement deficiency • More than 50% of patients with C 2 and C 4 deficiencies develop systemic lupus erythematosus • Deficiency of C 3 is associated with frequent serious pyogenic bacterial infections that may be fatal, illustrating the central role of C 3 in opsonization, enhanced phagocytosis, and destruction of these organisms • Deficiencies in components of the alternative pathway, including properdin and factor D, result in increased susceptibility to infection with pyogenic bacteria

• Deficiencies in the terminal complement components, including C 5, C 6, C 7, C 8, and C 9 disseminated infections by Neisseria bacteria, including Neisseria meningitidis and Neisseria gonorrhoeae • Deficiencies in complement regulatory proteins are associated with abnormal complement activation and a variety of related clinical abnormalities • Deficiencies in complement receptors include the absence of CR 3 and CR 4, both resulting from rare mutations in the β chain (CD 18) gene common to the CD 11 CD 18 family of integrin molecules is characterized by recurrent pyogenic infections and is caused by inadequate adherence of neutrophils to endothelium

Pathologic Effects of a Normal Complement System • Evasion of Complement by Microbes • Microbes can evade the complement system by recruiting host complement regulatory proteins • Some pathogens, like schistosomes, Neisseria gonorrhoeae, and certain Haemophilus species, scavenge sialic acids from the host and enzymatically transfer the sugar to their cell surfaces • GP 41 on human immunodeficiency virus (HIV) can bind to factor H, and this property of the virus is believed to contribute to virion protection

NEONATAL IMMUNITY • Neonatal mammals are protected from infection by maternally produced antibodies transported across the placenta into the fetal circulation and by antibodies in ingested milk transported across the gut epithelium of newborns by a specialized process known as transcytosis • Maternal Ig. G is transported across the placenta, and maternal Ig. A and Ig. G in breast milk are ingested by the nursing infant. The transepithelial transport of maternal Ig. A into breast milk depends on the poly Ig receptor

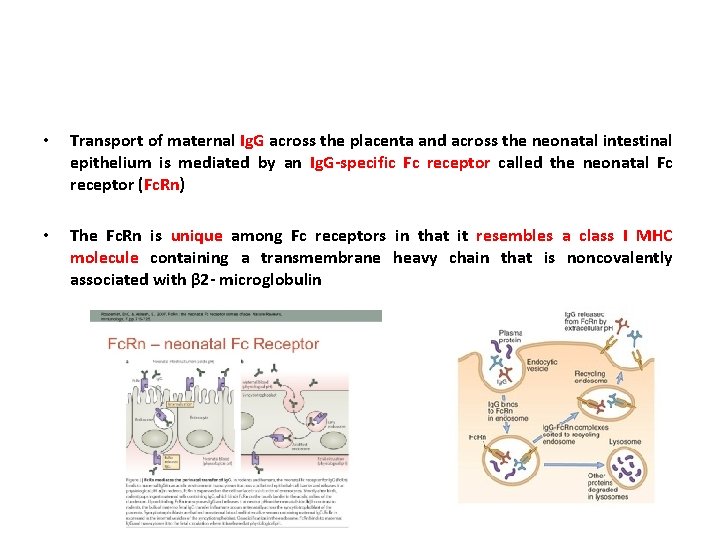
• Transport of maternal Ig. G across the placenta and across the neonatal intestinal epithelium is mediated by an Ig. G-specific Fc receptor called the neonatal Fc receptor (Fc. Rn) • The Fc. Rn is unique among Fc receptors in that it resembles a class I MHC molecule containing a transmembrane heavy chain that is noncovalently associated with β 2 - microglobulin
