What is Ferroelectric Ferroelectrics are materials which possess
















- Slides: 28

What is Ferroelectric? Ferroelectrics are materials which possess a “spontaneous” electric polarization Ps which can be reversed by applying a suitable electric field E. This process is known as “switching”, and is followed by “hysteresis”. Ferroelectrics are electrical analogues of “ferromagnetics” (P-E and M-H relations).

Ferroelectric Characteristics Three important characteristics of ferroelectrics: • Reversible polarization • “Anomalous” properties (i. e. ferroelectric disappears above a temperature Tc known as “Curie Point” • Non-linearities

Ferroelectric Characteristics • “Anomalous” properties (i. e. ferroelectric disappears above a temperature Tc known as “Curie Point” • Above Tc, the anomaly is frequently of the “ Curie-Weiss” form: (Curie-Weiss Relation) e = C / (T-T 0) C ~ Curie-Weiss constant T 0 is called “Curie-Weiss Temperature” T 0 < Tc in materials with first-order transitions T 0 = Tc in materials with second-order transitions !! Some materials do not follow Curie-Weiss Relation !!

Ferroelectric Characteristics • Dielectric non-linearities Measured dielectric permittivity changes with change of applied (bias field)

What is Piezoelectricity? Piezoelectrics are § materials which acquire electric polarization under external mechanical stresses (Direct Effect), OR § materials that change size or shape when subject to external electric field E (Converse Effect). ! (Piezo ~ Pressure or Stress) ! Ø Many piezoelectric materials are NOT ferroelectric Ø All ferroelectrics are piezoelectric ØAbove T 0, some ferroelectrics are STILL piezoelectric







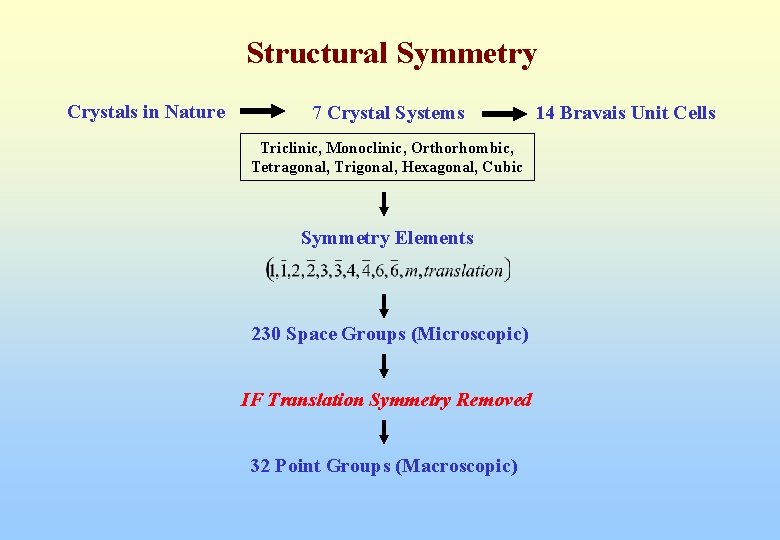
Structural Symmetry Crystals in Nature 7 Crystal Systems Triclinic, Monoclinic, Orthorhombic, Tetragonal, Trigonal, Hexagonal, Cubic Symmetry Elements 230 Space Groups (Microscopic) IF Translation Symmetry Removed 32 Point Groups (Macroscopic) 14 Bravais Unit Cells

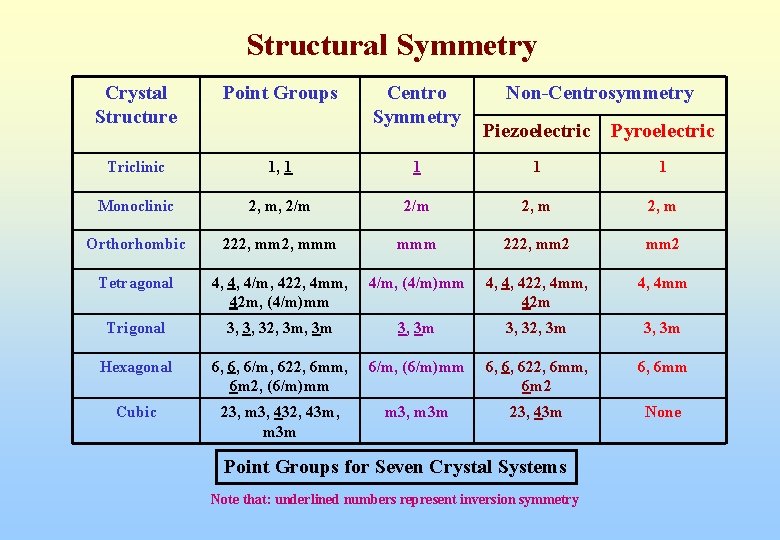
Structural Symmetry Crystal Structure Point Groups Triclinic 1, 1 Monoclinic Centro Symmetry Non-Centrosymmetry Piezoelectric Pyroelectric 1 1 1 2, m, 2/m 2, m Orthorhombic 222, mmm 222, mm 2 Tetragonal 4, 4, 4/m, 422, 4 mm, 42 m, (4/m)mm 4/m, (4/m)mm 4, 4, 422, 4 mm, 42 m 4, 4 mm Trigonal 3, 3, 32, 3 m 3, 3 m Hexagonal 6, 6, 6/m, 622, 6 mm, 6 m 2, (6/m)mm 6/m, (6/m)mm 6, 6, 622, 6 mm, 6 m 2 6, 6 mm Cubic 23, m 3, 432, 43 m, m 3 m m 3, m 3 m 23, 43 m None Point Groups for Seven Crystal Systems Note that: underlined numbers represent inversion symmetry

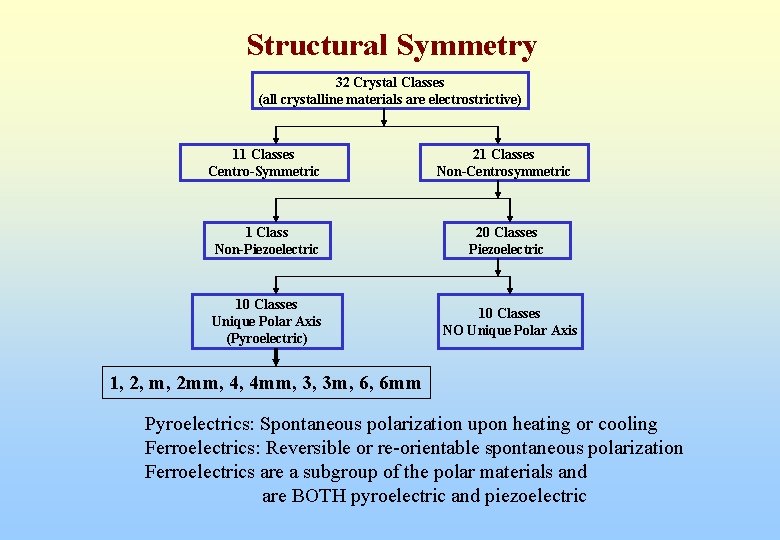
Structural Symmetry 32 Crystal Classes (all crystalline materials are electrostrictive) 11 Classes Centro-Symmetric 21 Classes Non-Centrosymmetric 1 Class Non-Piezoelectric 20 Classes Piezoelectric 10 Classes Unique Polar Axis (Pyroelectric) 10 Classes NO Unique Polar Axis 1, 2, m, 2 mm, 4, 4 mm, 3, 3 m, 6, 6 mm Pyroelectrics: Spontaneous polarization upon heating or cooling Ferroelectrics: Reversible or re-orientable spontaneous polarization Ferroelectrics are a subgroup of the polar materials and are BOTH pyroelectric and piezoelectric

Polarization (P) = Values of the dipole moment per unit volume = Values of the charge per unit surface area P = Nm / V = Nqd/Ad = Nq/A N = number of dipole moment per unit volume m = dipole moment = qd q = charge d = distance between positive and negative charges V = volume AND A = surface area

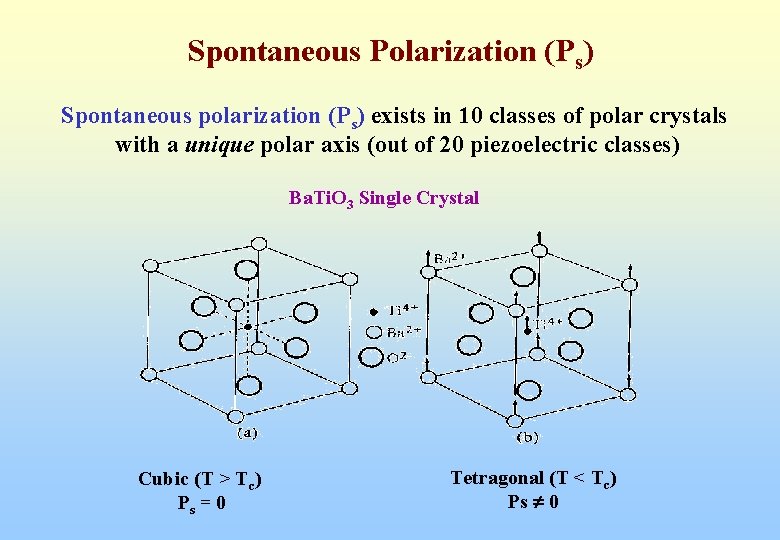
Spontaneous Polarization (Ps) Spontaneous polarization (Ps) exists in 10 classes of polar crystals with a unique polar axis (out of 20 piezoelectric classes) Ba. Ti. O 3 Single Crystal Cubic (T > Tc) Ps = 0 Tetragonal (T < Tc) Ps 0

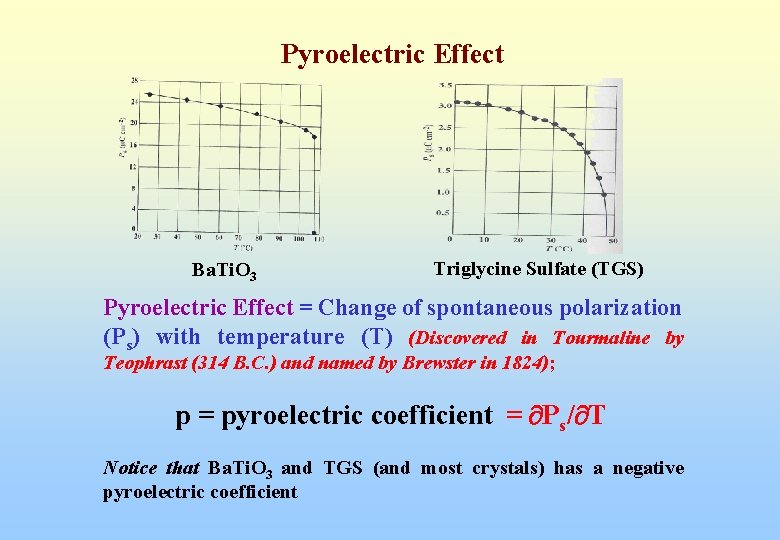
Pyroelectric Effect Ba. Ti. O 3 Triglycine Sulfate (TGS) Pyroelectric Effect = Change of spontaneous polarization (Ps) with temperature (T) (Discovered in Tourmaline by Teophrast (314 B. C. ) and named by Brewster in 1824); p = pyroelectric coefficient = Ps/ T Notice that Ba. Ti. O 3 and TGS (and most crystals) has a negative pyroelectric coefficient

Spontaneous Polarization (Ps) Re-Orientation Ceramics a large number of randomly oriented crystallites polarization re-orientation “Poling Process” E Unpoled Poled Changes in Ps-directions require small ionic movements Larger number of possible directions of polar axes Closer to poling direction Easily poled Tetragonal 4 mm 6 possible polar axes Rhombohedral 3 m 8 possible polar axes better alignment (poled)

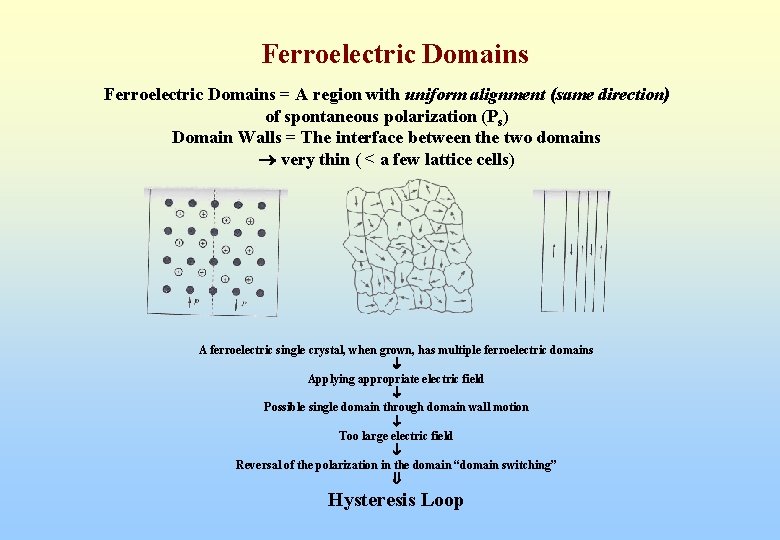
Ferroelectric Domains = A region with uniform alignment (same direction) of spontaneous polarization (Ps) Domain Walls = The interface between the two domains very thin ( < a few lattice cells) A ferroelectric single crystal, when grown, has multiple ferroelectric domains Applying appropriate electric field Possible single domain through domain wall motion Too large electric field Reversal of the polarization in the domain “domain switching” Hysteresis Loop

Ferroelectric Hysteresis Loop D Hysteresis Loop Starting from very small E-field Linear P-E relationship (OA) E leads to domain re-alignment in the positive direction along E rapid increase in P (OB) until it reaches the saturation value (Psat) E results in P, but NOT all to Zero P as E = 0 (BD) because some domains remain aligned in positive direction Remnant OR Remanent Polarization (Pr) Certain opposite E is needed to completely depolarize the domain Coercive Field (Ec) As E in negative direction of domains flip Þ Hysteresis Loop Þ Spontaneous Polarization (Ps) is obtained through extrapolation Þ Hysteresis Loop is observed by a Sawyer-Tower Circuit

Ferroelectric Curie Point and Phase Transitions Curie Point (Tc) = Phase transition temperature between non-ferroelectric and ferroelectric phases T < Tc = Ferroelectric Phase T > Tc = Paraelectric (Non-ferroelectric) Phase Transition Temperature = Other phase transition temperature between one ferroelectric phase to another

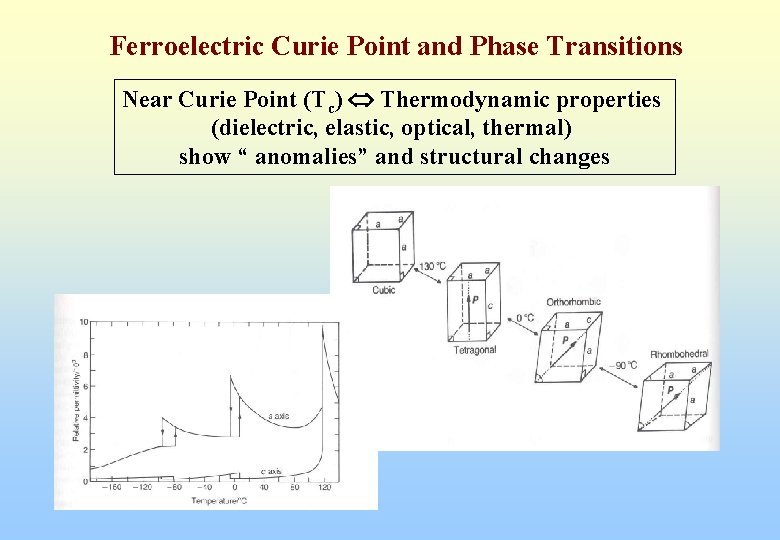
Ferroelectric Curie Point and Phase Transitions Near Curie Point (Tc) Thermodynamic properties (dielectric, elastic, optical, thermal) show “ anomalies” and structural changes

Ferroelectric Curie Point and Phase Transitions In most ferroelectrics, er above Curie Point (Tc) obeys Curie-Weiss Relation e = e 0 + C/(T-T 0) C = Curie-Weiss constant T 0 = Curie-Weiss Temperature (different from Curie Point Tc) T 0 < Tc for first-order phase transition T 0 = Tc for second-order phase transition Tc = actual temperature when crystal structure changes T 0 = formula constant obtained by extrapolation (Usually e 0 term is neglected because e 0 << e near T 0)

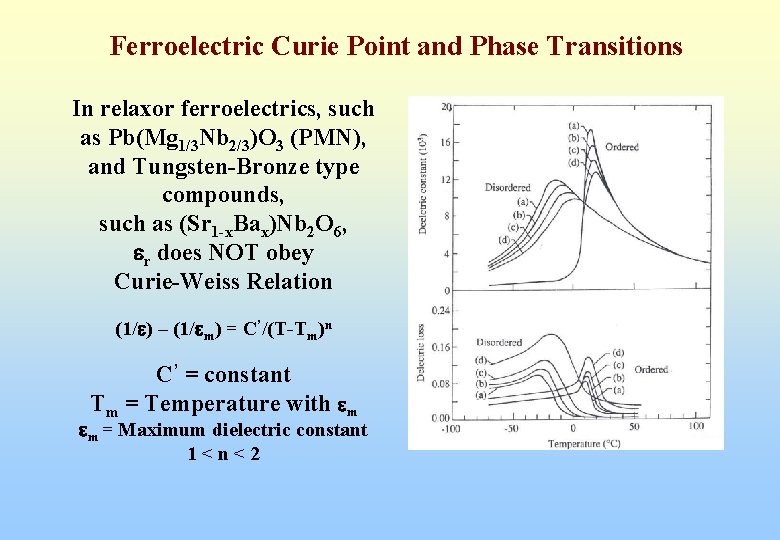
Ferroelectric Curie Point and Phase Transitions In relaxor ferroelectrics, such as Pb(Mg 1/3 Nb 2/3)O 3 (PMN), and Tungsten-Bronze type compounds, such as (Sr 1 -x. Bax)Nb 2 O 6, er does NOT obey Curie-Weiss Relation (1/e) – (1/em) = C’/(T-Tm)n C’ = constant Tm = Temperature with em em = Maximum dielectric constant 1<n<2

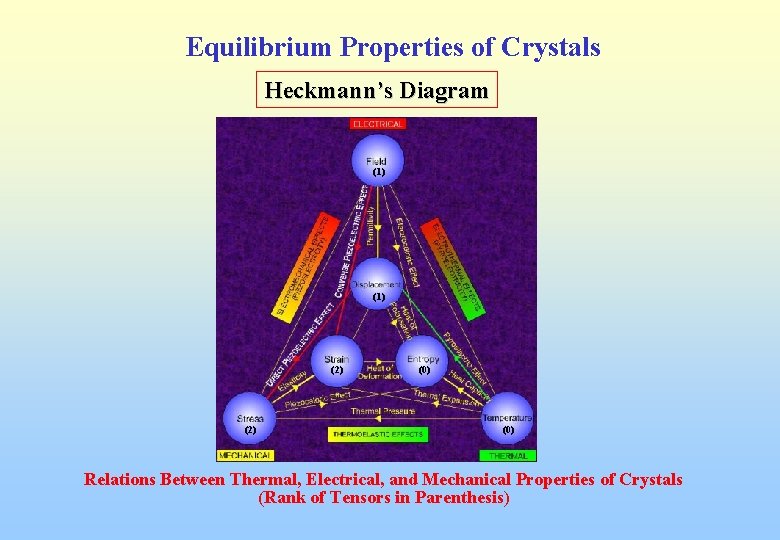
Equilibrium Properties of Crystals Heckmann’s Diagram (1) (2) (0) Relations Between Thermal, Electrical, and Mechanical Properties of Crystals (Rank of Tensors in Parenthesis)

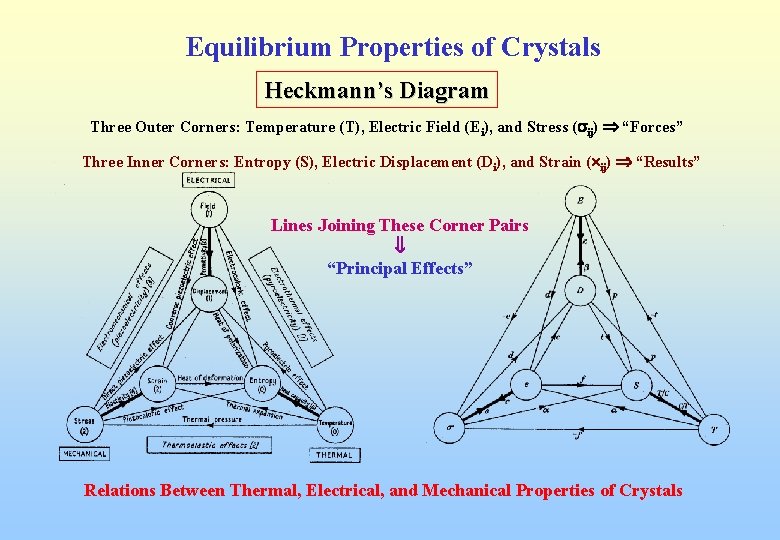
Equilibrium Properties of Crystals Heckmann’s Diagram Three Outer Corners: Temperature (T), Electric Field (Ei), and Stress ( ij) “Forces” Three Inner Corners: Entropy (S), Electric Displacement (Di), and Strain ( ij) “Results” Lines Joining These Corner Pairs “Principal Effects” Relations Between Thermal, Electrical, and Mechanical Properties of Crystals

Equilibrium Properties of Crystals I. An increase of temperature produces a change of entropy d. S: d. S = (C/T)d. T where C ( a scalar) is the “heat capacity per unit volume” T is the absolute temperature II. A small change of electric field d. Ei produces a change of electric displacement d. Di = ijd. Ej where ij is the “permittivity” tensor III. A small change of stress d kl produces a change of strain dxij = sijkl d kl where sijkl is the “elastic compliance”

Equilibrium Properties of Crystals Coupled Effects : Lines joining pairs not on the same corner Bottom : Thermoelastic Effects Right : Electrothermal Effects (Pyroelectric Effects) Left : Electromechanical Effects (Piezoelectric Effects) Direct and Converse Piezoelectric Effects (Third-Rank Tensors) d. Di = dijkd jk “Direct Effect” dxij = dijkd. Ek “Converse Effect” where dijk is the “piezoelectric coefficient ” Thermoelastic Effects : dxij = jjd. T Pyroelectric Effects : d. Di = pid. T