STED Nanoscale 3 D Optical Imaging Digvijay Raorane











- Slides: 19

STED: Nanoscale 3 D Optical Imaging Digvijay Raorane & Arun Majumdar Department of Mechanical Engineering Department of Materials Science University of California, Berkeley Materials Sciences Division Lawrence Berkeley National Laboratory

Outline • • Motivation Introduction: Conventional Optics Near Field Imaging STED - Theory - Previous experimental work • On-going experiments

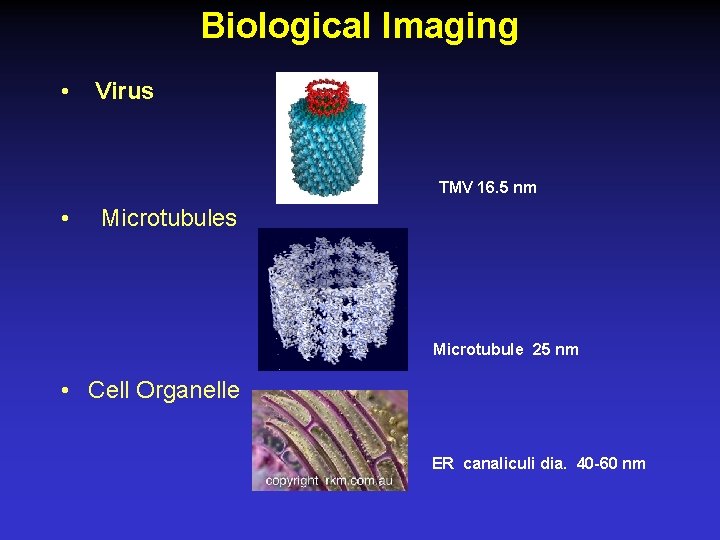
Biological Imaging • Virus TMV 16. 5 nm • Microtubules Microtubule 25 nm • Cell Organelle ER canaliculi dia. 40 -60 nm

Need for High Resolution Optical Technique • Biomolecules that require imaging are typically 1 - 50 nm in size • Far-field optics (e. g. confocal) limited to resolution > 200 nm, which cannot directly resolve molecular-scale phenomena • Atomic Force Microscopy cannot be used inside a cell • Optical/fluorescence imaging is most-widely used approach for real-time intracellular visualization • NSOM (Near-field Scanning Optical Microscope)

Optical Imaging at a Glance Far-Field Optics Near-Field Scanning Optical Microscope (NSOM) d Diffraction-limited spatial resolution ~ λ/2*NA Aperture-limited spatial resolution d ~ 50 nm

NSOM Limitations • Single fiber is limited to the field of view to ~ 50 nm. • It is difficult to maintain the tip at the constant distance from the sample within few nms. • Tip can get damaged by thermal stress due to the light. • Scanning a whole cell area (10 mm x 10 mm) takes time. • Multi-location imaging and dynamics cannot be observed. • Fiber-drawing and aperture fabrication is not repeatable, producing different imaging conditions each time. • Tip may get clogged when biological sample is in its buffer medium. Tip Profile http: //micro. magnet. fsu. edu Tip Damage Rosa et al. , Appl. Phys. Lett. 67, (18), 2597 -2599 (1995)

What is Stimulated Emission Depletion (STED) Microscopy?

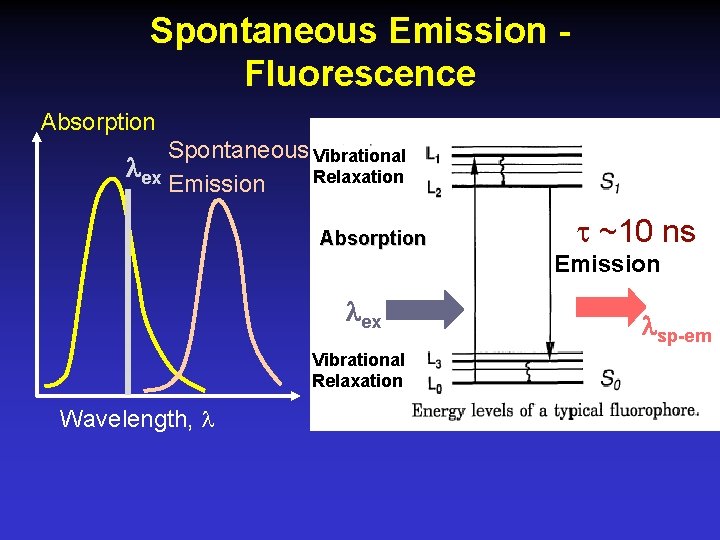
Spontaneous Emission Fluorescence Absorption Spontaneous Vibrational ex Relaxation Emission Absorption ~10 ns Emission ex Vibrational Relaxation Wavelength, sp-em

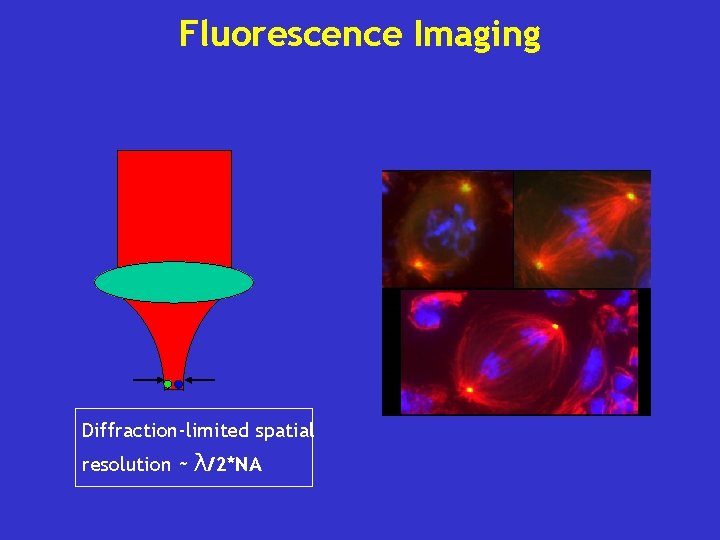
Fluorescence Imaging Diffraction-limited spatial resolution ~ λ/2*NA

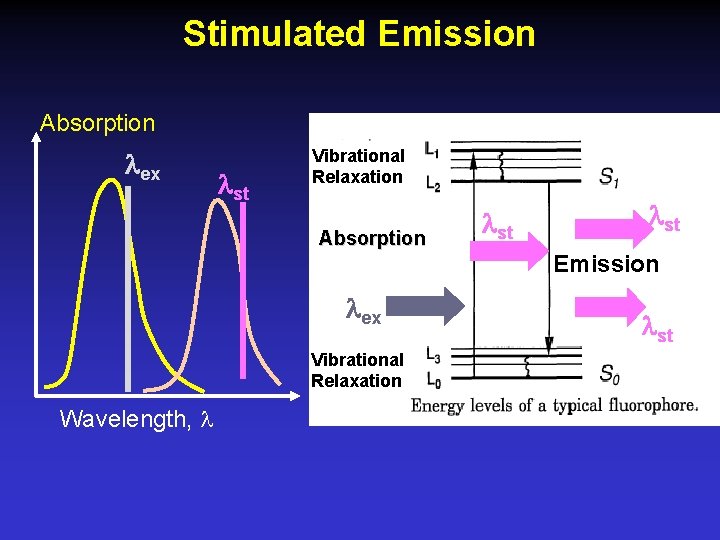
Stimulated Emission Absorption ex st Vibrational Relaxation Absorption st Emission ex Vibrational Relaxation Wavelength, st

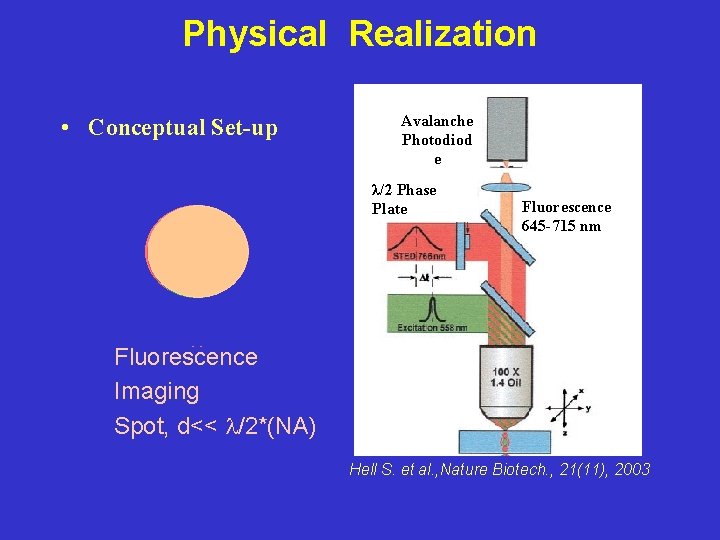
Physical Realization • Conceptual Set-up STED Spot Avalanche Photodiod e λ/2 Phase Plate Fluorescence 645 -715 nm Excitation Spot Fluorescence Imaging Spot, d<< /2*(NA) Hell S. et al. , Nature Biotech. , 21(11), 2003

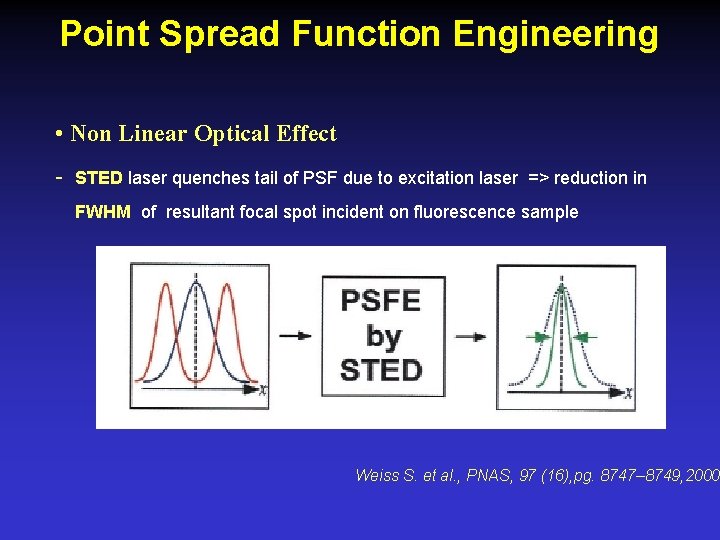
Point Spread Function Engineering • Non Linear Optical Effect - STED laser quenches tail of PSF due to excitation laser => reduction in FWHM of resultant focal spot incident on fluorescence sample Weiss S. et al. , PNAS, 97 (16), pg. 8747– 8749, 2000

Resolution • Resolution (FWHM) dependence on Intensity FWHM (Δr) • For typical experiment,

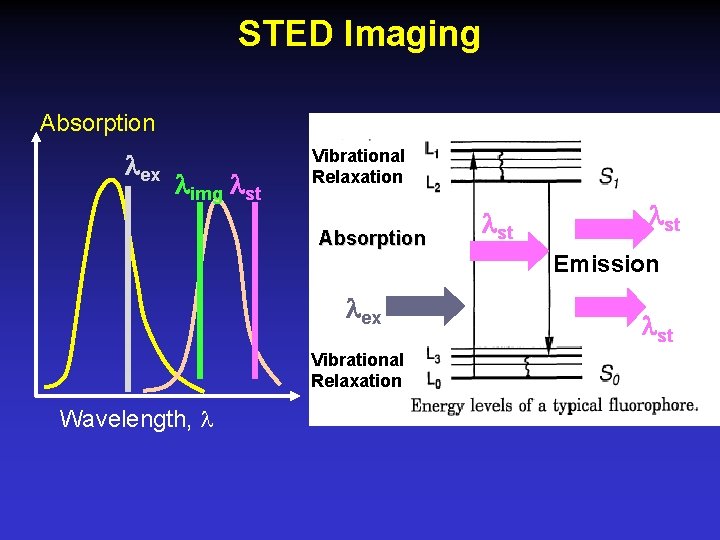
STED Imaging Absorption ex img st Vibrational Relaxation Absorption st Emission ex Vibrational Relaxation Wavelength, st

Proof of Concept Al 2 O 3 matrix wetted by Polymethyl Methacrylate Westphal et al, APL, 82(18), 3125 - 3127 (2003) Westphal et al, PRL 94, 143903 (2005)

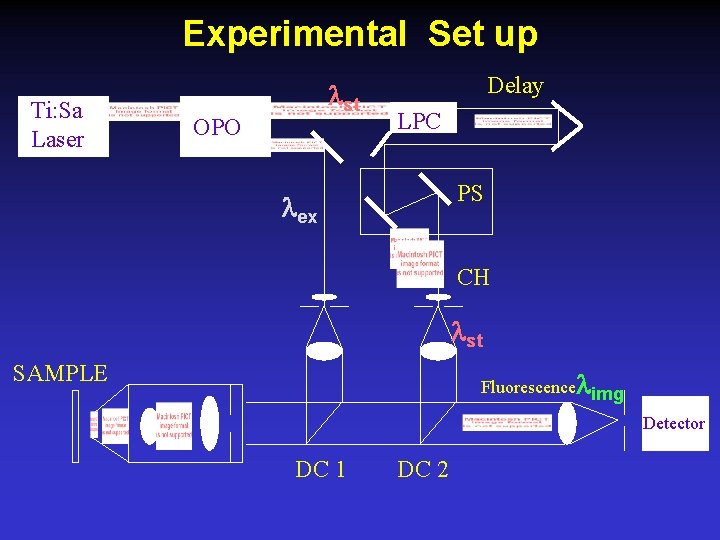
Experimental Set up Ti: Sa Laser st OPO Delay LPC PS ex CH st SAMPLE Fluorescence img Detector DC 1 DC 2

Collaboration: Prof. Costas Grigoropoulos (ME Dept, UCB)

On-Going Work • Quantum dot as substitute to fluorescent tags - To test compatibility of Q Dots with STED microscopy to overcome photobleaching of fluorescent labels Nucleus with actin fibres Hines et al, Advanced Materials, 15, 1845, 2003 Alivisatos et al, Nature Biotech. , 22, 47 – 52, 2004

Advantages of STED Microscopy • • • High resolution can be achieved routinely ( < 50 nm) No need of probe/tip Signal can be collected at far-field Better than confocal microscope in terms of resolution Multiple areas can be probed by forming multiple spots on the sample Resolution depends on the laser intensity ( FWHM = f( Intensity)) Incorporates most widely used fluorescence technique by biologists Can image live biological sample Optical system is simple to understand Can scan the sample in z direction for 3 D image 3 D Image Yeast Mammalian Mitochondria Golgi Hell et al, J. Opt. Soc. Am. A , 9(12), 2159 – 2166, 1992