Radiocarbon Dating of Groundwater Elisabetta Boaretto Radiocarbon Dating

Radiocarbon Dating of Groundwater Elisabetta Boaretto Radiocarbon Dating and Cosmogenic Isotopes Laboratory Kimmel Center for Archaeological Sciences, Weizmann Institute of Science and Dept of Land of Israel Studies and Archaeology Bar Ilan University
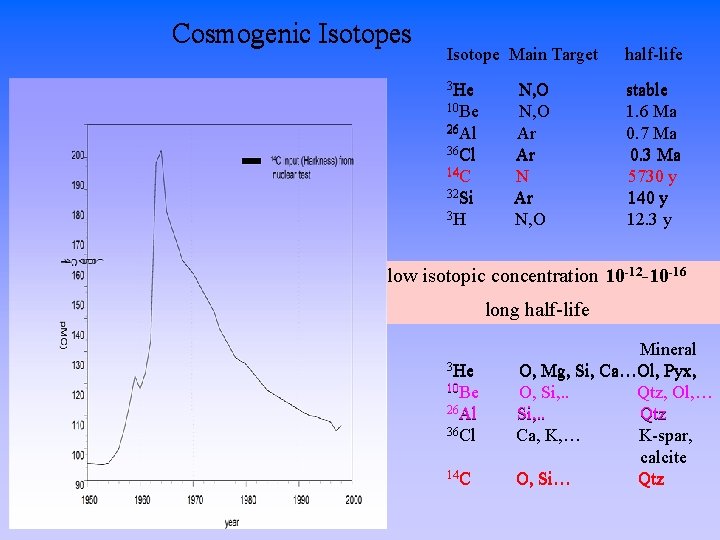
Cosmogenic Isotopes Isotope Main Target half-life 3 He stable 1. 6 Ma 0. 7 Ma 0. 3 Ma 5730 y 140 y 12. 3 y 10 Be 26 Al 36 Cl 14 C 32 Si 3 H N, O Ar Ar N, O low isotopic concentration 10 -12 -10 -16 long half-life 3 He 10 Be 26 Al 36 Cl 14 C Mineral O, Mg, Si, Ca…Ol, Pyx, O, Si, . . Qtz, Ol, … Si, . . Qtz Ca, K, … K-spar, calcite O, Si… Qtz
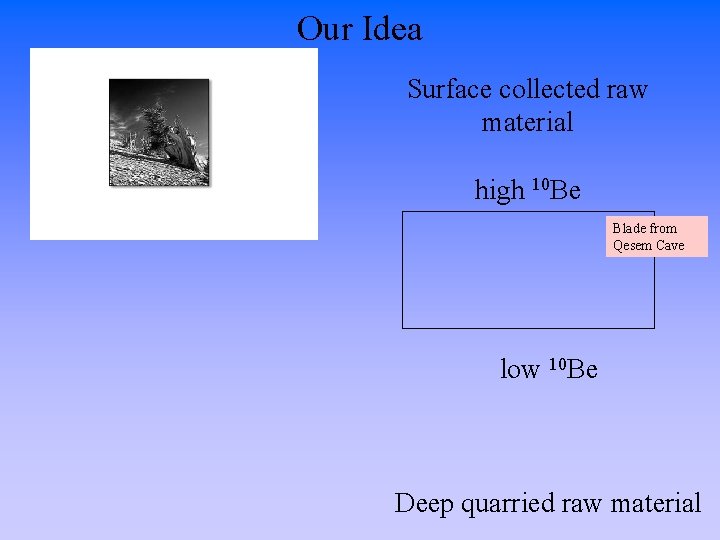
Our Idea Surface collected raw material high 10 Be Blade from Qesem Cave low 10 Be Deep quarried raw material
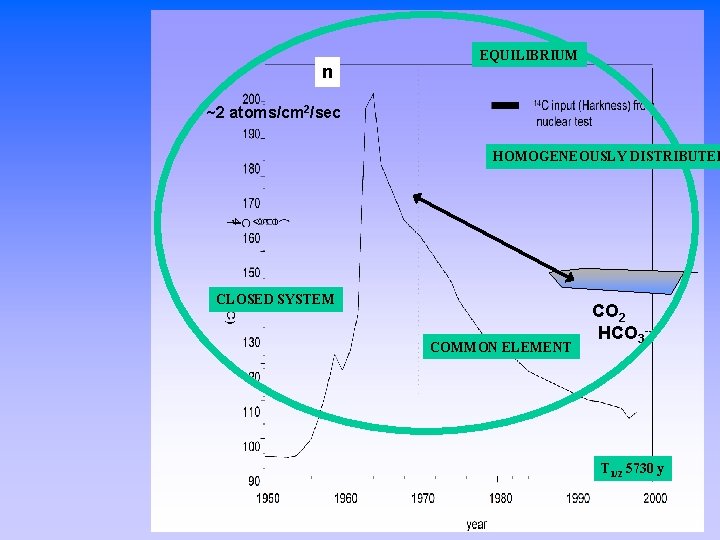
n EQUILIBRIUM ~2 atoms/cm 2/sec HOMOGENEOUSLY DISTRIBUTED CLOSED SYSTEM COMMON ELEMENT CO 2 HCO 3 -- T 1/2 5730 y
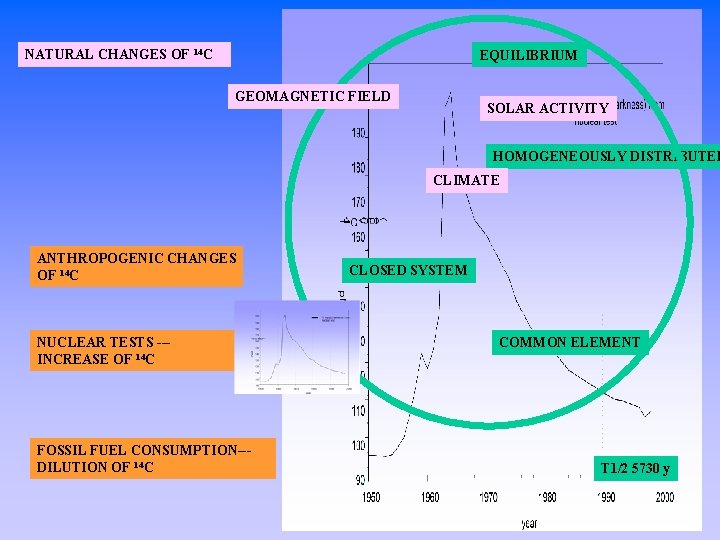
NATURAL CHANGES OF 14 C EQUILIBRIUM GEOMAGNETIC FIELD SOLAR ACTIVITY HOMOGENEOUSLY DISTRIBUTED CLIMATE ANTHROPOGENIC CHANGES OF 14 C NUCLEAR TESTS --INCREASE OF 14 C FOSSIL FUEL CONSUMPTION--DILUTION OF 14 C CLOSED SYSTEM COMMON ELEMENT T 1/2 5730 y
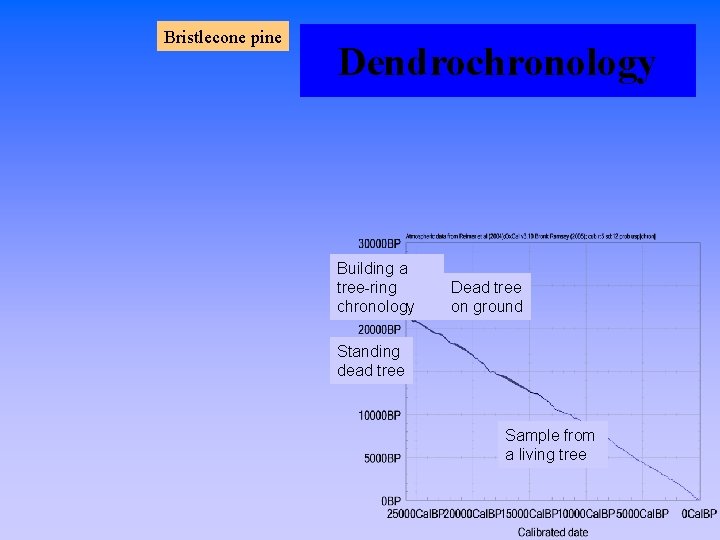
Bristlecone pine Dendrochronology Building a tree-ring chronology Dead tree on ground Standing dead tree Sample from a living tree
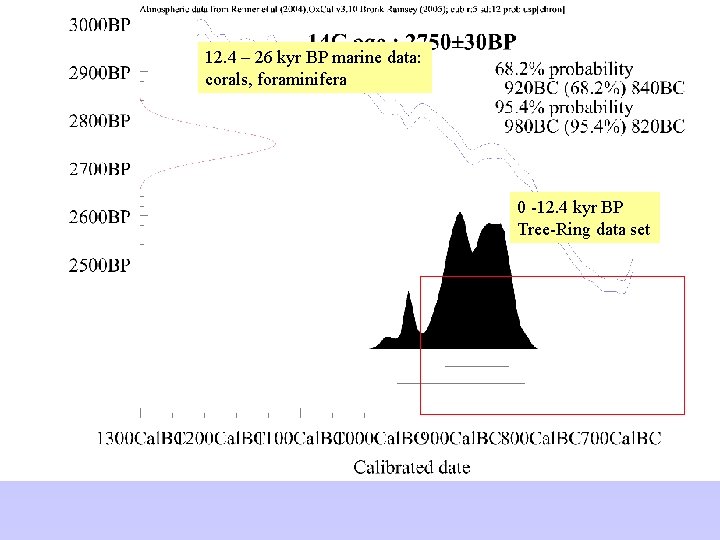
12. 4 – 26 kyr BP marine data: corals, foraminifera 0 -12. 4 kyr BP Tree-Ring data set


Radiocarbon • • • Activity is expressed in Pmc: percent modern carbon Age is calculated with the Decay Law Precision of 0. 5 -0. 3% = 40 -25 years Range 50, 000 year (LSC 60, 000 year) Isotopic fractionation between 12 C, 13 C and 14 C: (13 C/12 C)sample-(13 C/12 C)standard δ 13 C = -------------------- x 1000 (13 C/12 C)standard δ 13 C
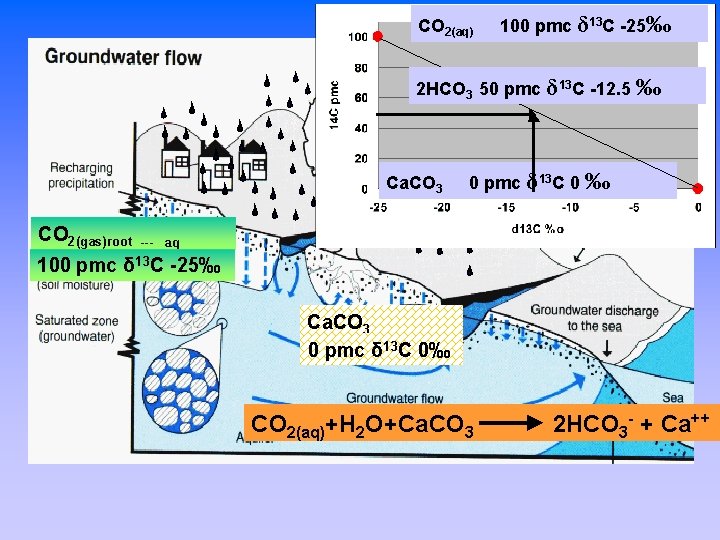
CO 2(aq) 100 pmc δ 13 C -25‰ 2 HCO 3 50 pmc δ 13 C -12. 5 ‰ How does carbon and radiocarbon get in the groundwater? Ca. CO 3 0 pmc δ 13 C 0 ‰ CO 2(gas)root 100 pmc --- aq δ 13 C -25‰ Ca. CO 3 0 pmc δ 13 C 0‰ CO 2(aq)+H 2 O+Ca. CO 3 2 HCO 3 - + Ca++
Different water environments Kinneret with Ohalo II Mediterranean Coast Dead sea and Lisan lake 14 C stalg Lisan Med Coast and U-Th
Lake Kinneret Using modern (alive) and fossil material, water
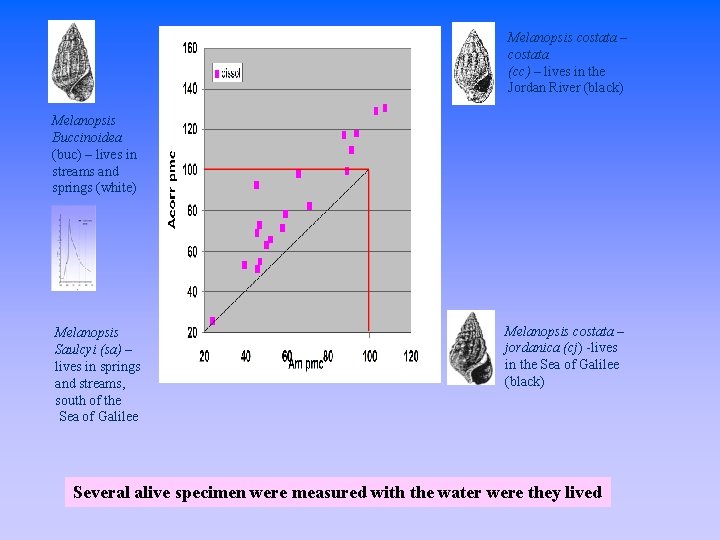
Melanopsis costata – costata (cc) – lives in the Jordan River (black) Melanopsis Buccinoidea (buc) – lives in streams and springs (white) Melanopsis Saulcyi (sa) – lives in springs and streams, south of the Sea of Galilee Melanopsis costata – jordanica (cj) -lives in the Sea of Galilee (black) Several alive specimen were measured with the water were they lived

“Water’ plant -32. 5
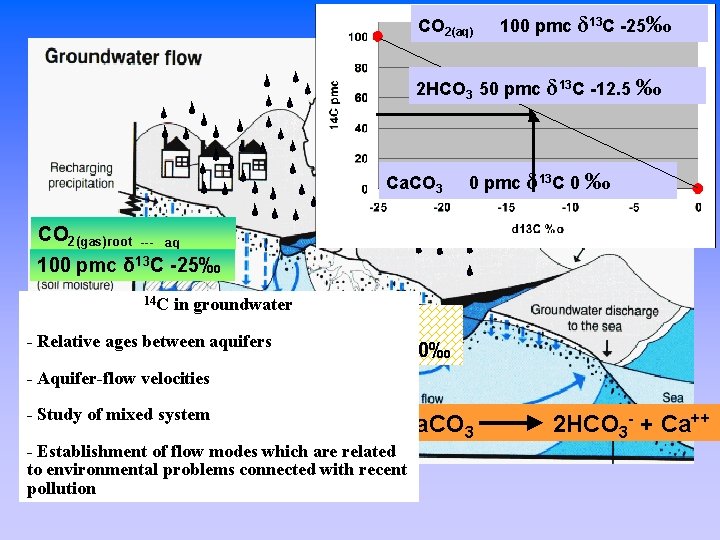
CO 2(aq) 100 pmc δ 13 C -25‰ 2 HCO 3 50 pmc δ 13 C -12. 5 ‰ How does carbon and radiocarbon get in the groundwater? Ca. CO 3 0 pmc δ 13 C 0 ‰ CO 2(gas)root 100 pmc --- aq δ 13 C -25‰ 14 C in groundwater - Relative ages between aquifers Ca. CO 3 0 pmc δ 13 C 0‰ - Aquifer-flow velocities - Study of mixed system CO 2(aq)+H 2 O+Ca. CO 3 - Establishment of flow modes which are related to environmental problems connected with recent pollution 2 HCO 3 - + Ca++

Hvinningdal Aquifer - Raised plain 80 m asl - 800 mm/y precipitation - >200 mm/y ca. recharge - Matrix: Tertiary quartz sand covered with quaternary meltwater sand/gravel - Low carbonate, some tertiary lignite and reworked black org material - p. H ca. 6 - T 8 -10 ºC - O 2 12 mg/l saturation value - Some wells have nitrate -Tritium (15 -62 TU) is present in almost all the wells indicating post-bomb groundwater origin.

18 wells analyzed. Tritium 15 -62 TU post-bomb
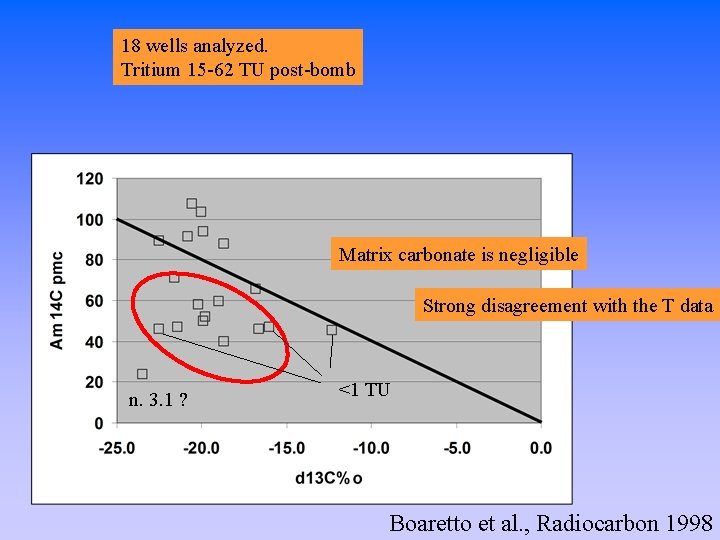
18 wells analyzed. Tritium 15 -62 TU post-bomb Matrix carbonate is negligible Strong disagreement with the T data n. 3. 1 ? <1 TU Boaretto et al. , Radiocarbon 1998

Oxygen as possible candidate for organic material oxidation T ºC 7 8 9 O 2 mg/l 11. 8 11. 5 11. 2 O 2 + (CH 2 O)n CO 2 + H 2 O
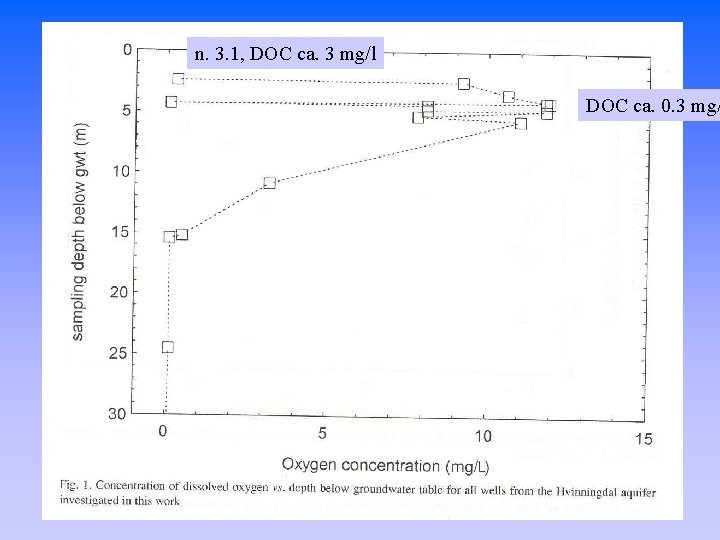
n. 3. 1, DOC ca. 3 mg/l DOC ca. 0. 3 mg/
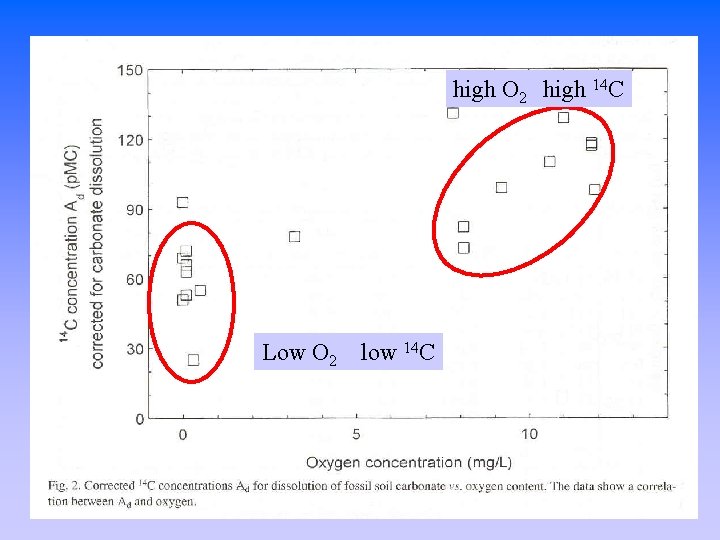
high O 2 high 14 C Low O 2 low 14 C

Acorr diss = Am(-25)/δ 13 Cm
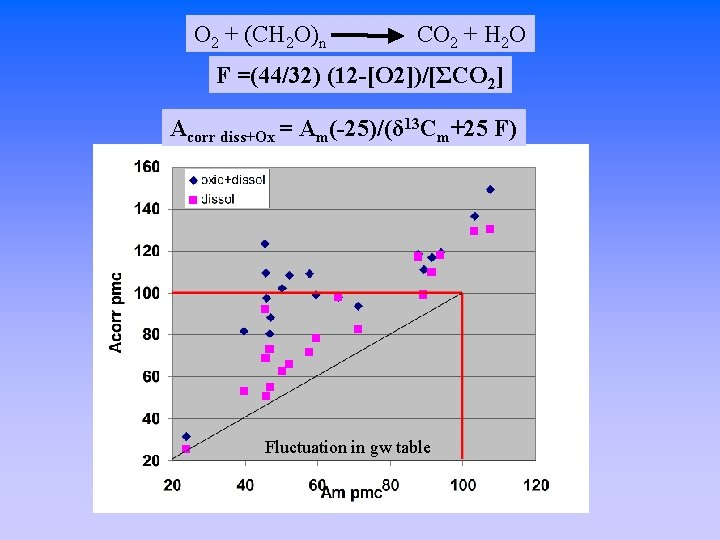
O 2 + (CH 2 O)n CO 2 + H 2 O F =(44/32) (12 -[O 2])/[ΣCO 2] Acorr diss+Ox = Am(-25)/(δ 13 Cm+25 F) Fluctuation in gw table
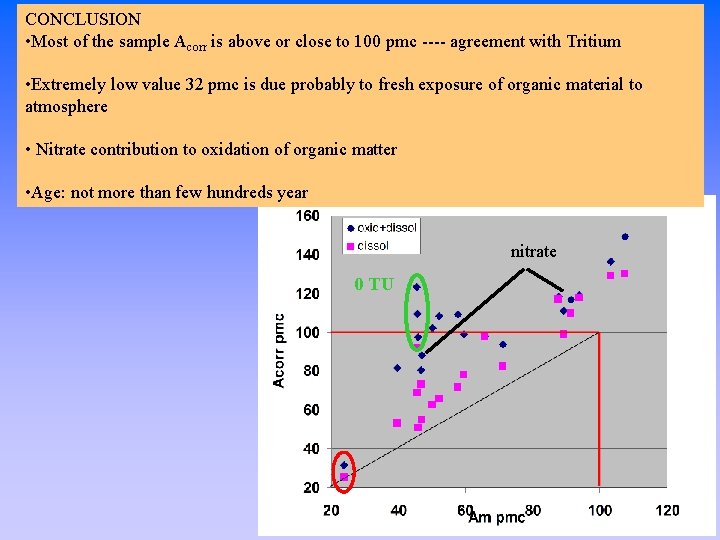
CONCLUSION • Most of the sample Acorr is above or close to 100 pmc ---- agreement with Tritium • Extremely low value 32 pmc is due probably to fresh exposure of organic material to atmosphere • Nitrate contribution to oxidation of organic matter • Age: not more than few hundreds year nitrate 0 TU
Radiocarbon Dating in Water Environments Very important for: -control and use of hydrological resources -chronologies Variables are several modeling is necessary
- Slides: 25