Clinical Practice Guidelines Nonalcoholic fatty liver disease About

Clinical Practice Guidelines Non-alcoholic fatty liver disease

About these slides • These slides give a comprehensive overview of the EASL clinical practice guidelines on non-alcoholic fatty liver disease • The guidelines were published in full in the June 2016 issue of the Journal of Hepatology – The full publication can be downloaded from the Clinical Practice Guidelines section of the EASL website – Please cite the published article as: EASL–EASD–EASO 2016 Clinical Practice Guidelines on the management of non-alcoholic fatty liver disease. J Hepatol 2016; 64: 1388– 402 • Please feel free to use, adapt, and share these slides for your own personal use; however, please acknowledge EASL as the source

About these slides • Definitions of all abbreviations shown in these slides are provided within the slide notes • When you see a home symbol like this one: , you can click on this to return to the outline or topics pages, depending on which section you are in These slides are intended for use as an educational resource and should not be used in isolation to make patient management decisions. All information included should be verified before treating patients or using any therapies described in these materials • Please send any feedback to: slidedeck_feedback@easloffice. eu

Guideline panel • Chairs – EASL: Giulio Marchesini – EASD: Michael Roden – EASO: Roberto Vettor • Panel members – EASL: Christopher P Day, Jean-François Dufour, Ali Canbay, Valerio Nobili, Vlad Ratziu, Herbert Tilg – EASD: Amalia Gastaldelli, Hannele Yki-Järvinen, Fritz Schick – EASO: Gema Frühbeck, Lisbeth Mathus-Vliegen • Reviewers – Elisabetta Bugianesi, Helena Cortez-Pinto, Stephen Harrison EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Outline Methods • Grading evidence and recommendations Background • Definitions of NAFLD, NAFL and NASH Guidelines • Key topics and recommendations EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Methods Grading evidence and recommendations

Grading evidence and recommendations • Grading is adapted from the GRADE system 1 Grade of evidence A: High quality Further research is very unlikely to change our confidence in the estimate effect B: Moderate quality Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate effect C: Low/very low quality Further research is very likely to have an important impact on our confidence in the estimate of effect and may change the estimate effect. Any estimate of effect is uncertain Grade of recommendation 1: Strong Factors influencing the strength of the recommendation included the quality of the evidence, presumed patient-important outcomes, and cost 2: Weaker Variability in preferences and values, or more uncertainty: more likely a weak recommendation is warranted. Recommendation is made with less certainty; higher cost or resource consumption 1. Chalasani N, et al. Hepatology 2012; 55: 2005 23; EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Background Definitions of NAFLD, NAFL and NASH
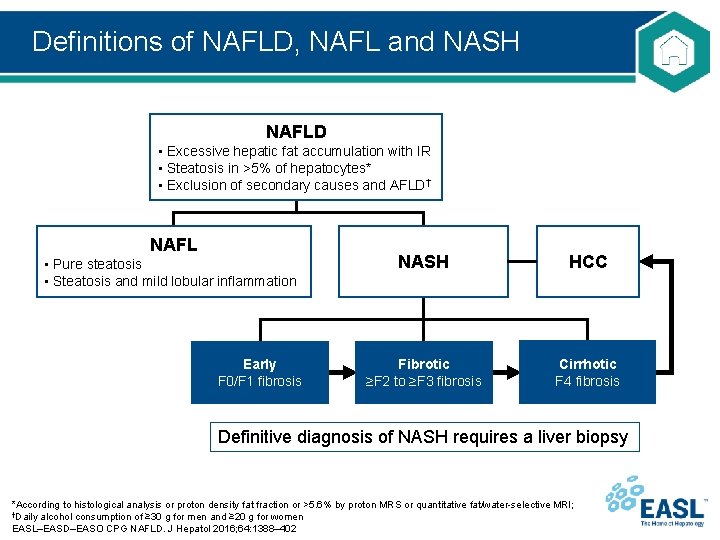
Definitions of NAFLD, NAFL and NASH NAFLD • Excessive hepatic fat accumulation with IR • Steatosis in >5% of hepatocytes* • Exclusion of secondary causes and AFLD† NAFL • Pure steatosis • Steatosis and mild lobular inflammation Early F 0/F 1 fibrosis NASH HCC Fibrotic ≥F 2 to ≥F 3 fibrosis Cirrhotic F 4 fibrosis Definitive diagnosis of NASH requires a liver biopsy *According to histological analysis or proton density fat fraction or >5. 6% by proton MRS or quantitative fat/water-selective MRI; †Daily alcohol consumption of ≥ 30 g for men and ≥ 20 g for women EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Spectrum of NAFLD and concurrent disease Sub-classification of NAFLD* Most common concurrent diseases NAFL • Pure steatosis • Steatosis and mild lobular inflammation AFLD† Drug-induced fatty liver disease† HCV-associated fatty liver disease (GT 3)† Others† • Haemochromatosis • Autoimmune hepatitis • Coeliac disease • Wilson disease • A/hypo-betalipoproteinaemia lipoatrophy • Hypopituitarism, hypothyroidism • Starvation, parenteral nutrition • Inborn errors of metabolism – Wolman disease (lysosomal acid lipase deficiency) NASH • Early NASH (no or mild fibrosis) • Fibrotic NASH (significant/advanced fibrosis) • NASH cirrhosis HCC‡ *Also called primary NAFLD and associated with metabolic risk factors/components of Met. S: 1. Waist circumference ≥ 94/≥ 80 cm for Europid men/women; 2. Arterial pressure ≥ 130/85 mm. Hg or treated for hypertension; 3. Fasting glucose ≥ 100 mg/dl (5. 6 mmol/L) or treated for T 2 DM; 4. Serum triacylglycerols >150 mg/dl (>1. 7 mmol/L); 5. HDL cholesterol <40/50 mg/dl for men/women (<1. 0/<1. 3 mmol/L); †Also called secondary NAFLD. Note that primary and secondary NAFLD may coexist in individual patients. Also NAFLD and AFLD may coexist in subjects with metabolic risk factors and drinking habits above safe limits; ‡Can occur in the absence of cirrhosis and histological evidence of NASH, but with metabolic risk factors suggestive of ‘‘burned-out” NASH EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
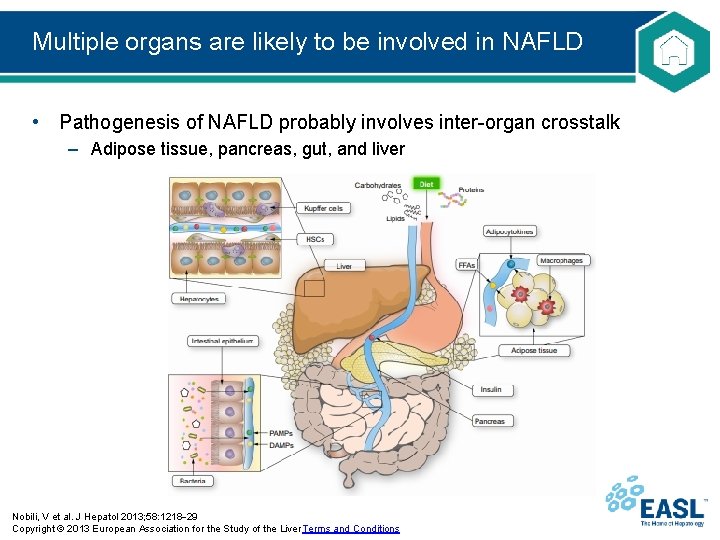
Multiple organs are likely to be involved in NAFLD • Pathogenesis of NAFLD probably involves inter-organ crosstalk – Adipose tissue, pancreas, gut, and liver Nobili, V et al. J Hepatol 2013; 58: 1218 29 Copyright © 2013 European Association for the Study of the Liver Terms and Conditions
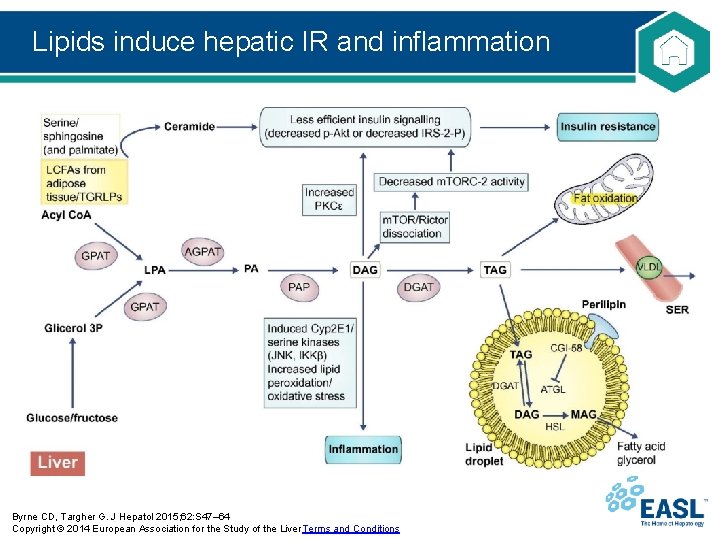
Lipids induce hepatic IR and inflammation Byrne CD, Targher G. J Hepatol 2015; 62: S 47– 64 Copyright © 2014 European Association for the Study of the Liver Terms and Conditions

Guidelines Key topics and recommendations

Topics 1. 2. 3. 4. 5. 6. 7. 8. Screening, prevalence and incidence Pathogenesis: lifestyle and genes Liver biopsy Non-invasive assessments Common related metabolic disorders Diagnosis Natural history and complications Treatment – – Diet and lifestyle changes Drug treatment Paediatric NAFLD Surgery EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Click on a topic to skip to that section

Screening, prevalence and incidence • NAFLD is the most common liver disorder in Western countries, affecting 17– 46% of adults 1 – Parallels the prevalence of metabolic syndrome (Met. S) and its components, which also increase the risk of more advanced disease – NAFLD is also present in 7% of normal-weight (lean) individuals 2 Recommendations Grade of evidence Grade of recommendation Patients with IR and/or metabolic risk factors (i. e. obesity or Met. S) should undergo procedures for the diagnosis of NAFLD A 1 Screen individuals with steatosis for secondary causes of NAFLD, including a careful assessment of alcohol intake. Always consider the interaction between moderate amounts of alcohol and metabolic factors in fatty liver A 1 Identify other chronic liver diseases that may coexist with NAFLD as these might result in more severe liver injury B 1 1. Vernon G, et al. Aliment Pharmacol Ther 2011; 34: 274 85; 2. Younossi ZM, et al. Medicine 2012; 91: 319 27; EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Screening, prevalence and incidence • Value of screening for NAFLD in the community is limited – – High direct and indirect costs Low predictive value of non-invasive tests Risks associated with liver biopsy Lack of effective treatments • Diagnosis of NASH provides important diagnostic information – Points to increased risk of fibrosis progression, cirrhosis and possibly HCC Recommendations Grade of evidence Grade of recommendation All individuals with steatosis should be screened for features of Met. S, independent of liver enzymes. All individuals with persistently abnormal liver enzymes should be screened for NAFLD A 1 In subjects with obesity or Met. S, screening for NAFLD should be part of routine work-up. In high-risk individuals* case finding of advanced disease is advisable A 2 *Aged >50 years, T 2 DM, Met. S EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Pathogenesis: lifestyle and genes • A Western diet/lifestyle has been associated with weight gain and obesity, and NAFLD 1 High calorie intake Excess (saturated) fat High fructose intake Obesity NAFLD Sedentary behaviour Recommendation Grade of evidence Unhealthy lifestyles play a role in the development and progression of NAFLD. The assessment of dietary and physical activity habits is part of comprehensive NAFLD screening 1. Barrera F, George J. Clin Liver Dis 2014; 18: 91– 112; EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Grade of recommendation A 1

Pathogenesis: lifestyle and genes • Several genetic modifiers of NAFLD have been identified 1 – A minority have been robustly validated • PNPLA 3 I 148 M and TM 6 SF 2 E 167 K carriers have a higher liver fat content* – Increased risk of NASH – NAFLD not systematically associated with features of IR Recommendation Grade of evidence Genotyping may be considered in selected patients and clinical studies but is not recommended routinely *Grade of evidence B, grade of recommendation 2 1. Anstee QM, et al. Nat Rev Gastroenterol Hepatol 2013; 10: 330– 44; EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Grade of recommendation B 2
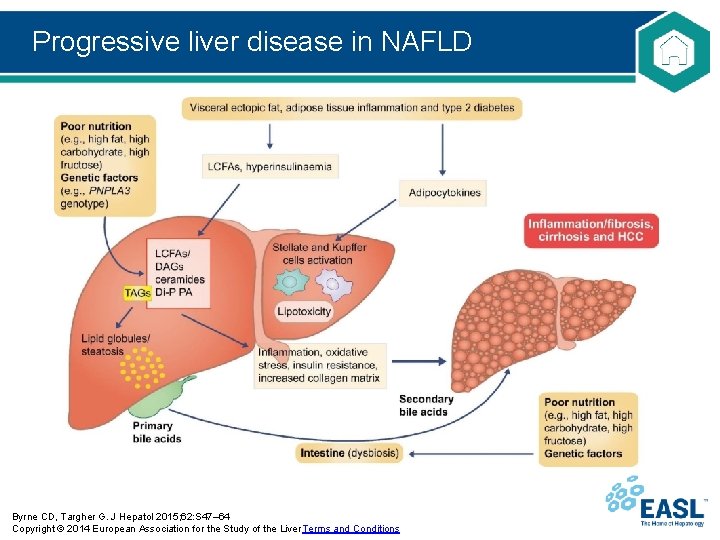
Progressive liver disease in NAFLD Byrne CD, Targher G. J Hepatol 2015; 62: S 47– 64 Copyright © 2014 European Association for the Study of the Liver Terms and Conditions
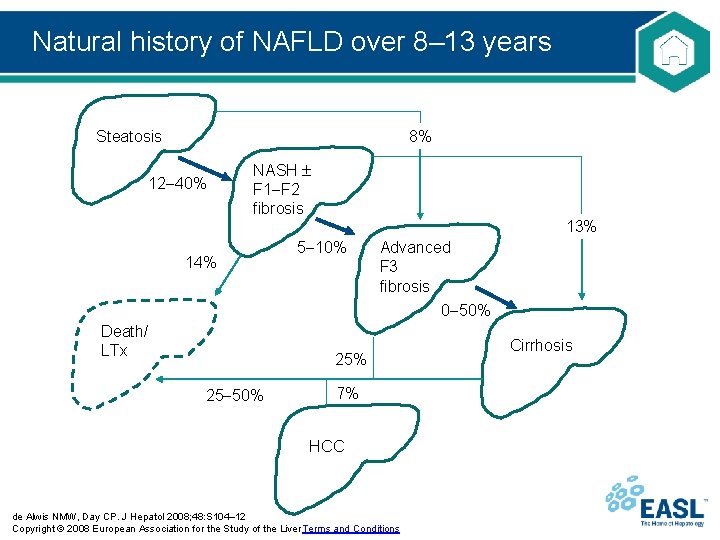
Natural history of NAFLD over 8– 13 years Steatosis 8% 12 40% NASH F 1 F 2 fibrosis 13% 14% 5 10% Advanced F 3 fibrosis 0 50% Death/ LTx 25% 25 50% 7% HCC de Alwis NMW, Day CP. J Hepatol 2008; 48: S 104– 12 Copyright © 2008 European Association for the Study of the Liver Terms and Conditions Cirrhosis
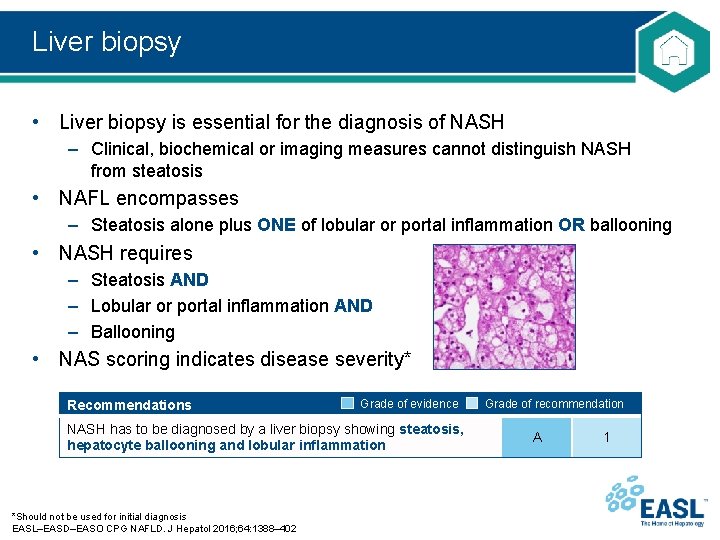
Liver biopsy • Liver biopsy is essential for the diagnosis of NASH – Clinical, biochemical or imaging measures cannot distinguish NASH from steatosis • NAFL encompasses – Steatosis alone plus ONE of lobular or portal inflammation OR ballooning • NASH requires – Steatosis AND – Lobular or portal inflammation AND – Ballooning • NAS scoring indicates disease severity* Recommendations Grade of evidence NASH has to be diagnosed by a liver biopsy showing steatosis, hepatocyte ballooning and lobular inflammation *Should not be used for initial diagnosis EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Grade of recommendation A 1

Role of non-invasive assessments • Non-invasive markers should aim to: – Identify the risk of NAFLD among individuals with increased metabolic risk in primary care – Identify those with a worse prognosis in secondary and tertiary care • E. g. severe NASH – Monitor disease progression – Predict response to therapeutic interventions Achieving these aims could reduce the need for liver biopsy EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Non-invasive assessment of steatosis • Steatosis should be documented whenever NAFLD is suspected – Predicts future T 2 DM, cardiovascular events and arterial hypertension – Quantification of fat content is of limited clinical relevance • Except as a surrogate of treatment effectiveness Recommendations Grade of evidence Grade of recommendation US is the preferred first-line diagnostic procedure for imaging of NAFLD, as it provides additional diagnostic information A 1 Whenever imaging tools are not available or feasible serum biomarkers and scores are an acceptable alternative for the diagnosis of steatosis B 2 A quantitative estimation of liver fat can only be obtained by 1 H-MRS. This technique is of value in clinical trials and experimental studies, but is expensive and not recommended in the clinical setting A 1 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Non-invasive assessment of fibrosis • Fibrosis is the most important prognostic factor in NAFLD – Correlates with liver-related outcomes and mortality – Advanced fibrosis indicates thorough investigation Recommendations Grade of evidence Grade of recommendation Biomarkers, fibrosis scores, and transient elastography, are acceptable non-invasive procedures to identify those at low risk of advanced fibrosis/cirrhosis A 2 Biomarkers/scores PLUS transient elastography might confer additional diagnostic accuracy and reduce need for liver biopsy B 2 Monitoring of fibrosis progression may rely on biomarkers/scores and transient elastography, although this strategy requires validation C 2 The identification of advanced fibrosis or cirrhosis by serum biomarkers/scores and/or elastography is less accurate and needs to be confirmed by liver biopsy, according to the clinical context B 2 In selected patients at high risk of liver disease progression, monitoring should include a repeat biopsy after 5 -year follow-up C 2 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
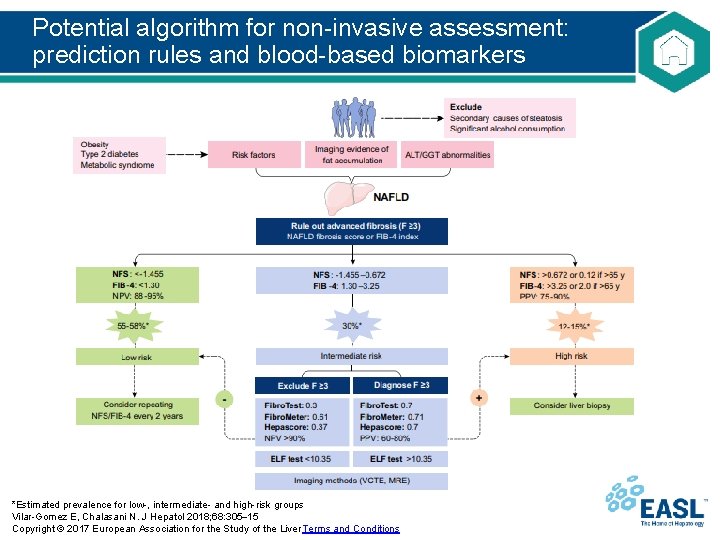
Potential algorithm for non-invasive assessment: prediction rules and blood-based biomarkers *Estimated prevalence for low-, intermediate- and high-risk groups Vilar-Gomez E, Chalasani N. J Hepatol 2018; 68: 305 15 Copyright © 2017 European Association for the Study of the Liver Terms and Conditions

Non-invasive assessment of paediatric NAFLD • NAFLD should always be suspected in obese children – Exclude other causes – Evaluate elevated aminotransferase levels and liver hyperechogenicity • Due to the poor sensitivity in overweight/obese children, non -invasive markers and imaging techniques are the first diagnostic step Recommendations Grade of evidence Grade of recommendation In children, predictors of fibrosis, including elastometry, ARFI imaging and serum biomarkers might help reduce the number of biopsies Roberts EA. J Hepatol 2007; 46: 1133 42 Copyright © 2007 European Association for the Study of the Liver Terms and Conditions EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 B 2

Common related metabolic disorders • NAFLD is closely associated with: – IR in the liver as well as adipose and muscle tissue – Met. S • Three of: impaired fasting glucose or T 2 DM, hypertriglyceridaemia, low HDL-C, * increased waist circumference, † high blood pressure All components of Met. S correlate with liver fat content: Evaluate risk of NAFLD in patients with Met. S Evaluate Met. S in patients with NAFLD *Gender-adjusted; †ethnicity-adjusted EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Common related metabolic disorders • In individuals without diabetes, HOMA-IR can be considered as a surrogate for IR HOMA-IR: Fasting glucose (mmol/L) + insulin (m. U/ml) 22. 5 Recommendations Grade of evidence Grade of recommendation HOMA-IR can be recommended if proper reference values have been established A 1 HOMA-IR is of limited use for NAFLD diagnosis in patients with metabolic risk factors. It could confirm altered insulin sensitivity, thereby favouring a diagnosis of IR-associated NAFLD in cases of diagnostic uncertainty* B 2 During follow-up, HOMA-IR might help identify patients at risk of NASH or fibrosis progression in selected cases. Improvement of HOMA-IR during weight loss may indicate metabolic improvement C 2 *E. g. US-defined steatosis with normal body weight EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Common related metabolic disorders: obesity • BMI and waist circumference are positively related to NAFLD – Predictors of advanced disease, particularly in the elderly Recommendations Grade of evidence Grade of recommendation Follow up is mandatory in obesity, which is the major phenotype and risk condition for NAFLD, driven by IR, and also increases the risk of advanced disease A 1 Most lean individuals with NAFLD display IR and altered body fat distribution even though they have less severe metabolic disturbance than overweight NAFLD. Follow-up is nonetheless required because of possible disease progression B 2 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Common related metabolic disorders: T 2 DM • Irrespective of liver enzymes, diabetes risk and T 2 DM are closely associated with: – – Severity of NAFLD Progression to NASH Presence of advanced fibrosis Development of HCC Recommendations Grade of evidence Grade of recommendation In individuals with NAFLD, screening for diabetes is mandatory, by fasting or random blood glucose or Hb. A 1 c… A 1 …and if available, by the standardized 75 g OGTT in high-risk groups B 1 Look for NAFLD in patients with T 2 DM, irrespective of liver enzyme levels, due to high risk of disease progression A 2 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Diagnosis: protocol for evaluation of NAFLD • Incidental discovery of steatosis indicates comprehensive evaluation – Family and personal history of NAFLD-associated diseases – Exclusion of secondary causes of steatosis Level Variable Initial evaluation 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Alcohol intake: <20 g/day (women), <30 g/day (men) Personal and family history of diabetes, hypertension and CVD BMI, waist circumference, change in body weight Hepatitis B/hepatitis C virus infection History of steatosis-associated drugs Liver enzymes (ALT, AST, GGT) Fasting blood glucose, Hb. A 1 c, OGTT, (fasting insulin [HOMA-IR]) Complete blood count Serum total and HDL cholesterol, triacylglycerol, uric acid Ultrasonography (if suspected for raised liver enzymes) Extended* evaluation 1. 2. 3. Ferritin and transferrin saturation Tests for coeliac and thyroid diseases, polycystic ovary syndrome Tests for rare liver diseases (Wilson, autoimmune disease, AATD) *According to a priori probability or clinical evaluation EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
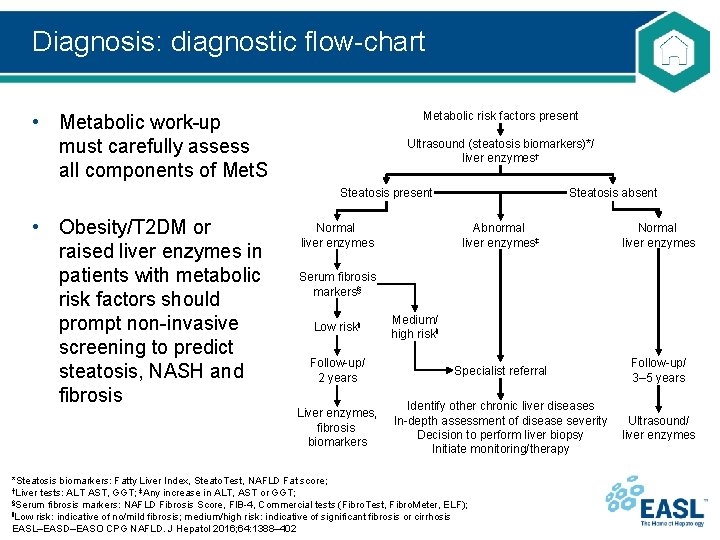
Diagnosis: diagnostic flow-chart Metabolic risk factors present • Metabolic work-up must carefully assess all components of Met. S Ultrasound (steatosis biomarkers)*/ liver enzymes† Steatosis present • Obesity/T 2 DM or raised liver enzymes in patients with metabolic risk factors should prompt non-invasive screening to predict steatosis, NASH and fibrosis Normal liver enzymes Steatosis absent Abnormal liver enzymes‡ Normal liver enzymes Follow-up/ 2 years Specialist referral Follow-up/ 3– 5 years Liver enzymes, fibrosis biomarkers Identify other chronic liver diseases In-depth assessment of disease severity Decision to perform liver biopsy Initiate monitoring/therapy Ultrasound/ liver enzymes Serum fibrosis markers§ Low risk‖ Medium/ high risk‖ *Steatosis biomarkers: Fatty Liver Index, Steato. Test, NAFLD Fat score; †Liver tests: ALT AST, GGT; ‡Any increase in ALT, AST or GGT; §Serum fibrosis markers: NAFLD Fibrosis Score, FIB-4, Commercial tests (Fibro. Test, Fibro. Meter, ELF); ‖Low risk: indicative of no/mild fibrosis; medium/high risk: indicative of significant fibrosis or cirrhosis EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
Natural history and complications: progression • In general, NAFLD is a slowly progressive disease, both in adults and in children – Rate of progression corresponds to 1 fibrosis stage every 14 years in NAFL and every 7 years in NASH – Rate of progression is doubled by arterial hypertension 1 – Progression of fibrosis is more rapid in about 20% of cases 1 Recommendations Grade of evidence NASH patients with fibrosis associated with hypertension should receive closer monitoring because of a higher risk of disease progression Grade of recommendation B 1 • Paediatric NAFLD is of concern – Potential for severe liver-related complications later in life – NASH-related cirrhosis has been reported as early as 8 years of age 1. Sing S et al. Clin Gastroenterol Hepatol 2015; 13: 643– 54; EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Natural history and complications: CVD • Prevalence and incidence of CVD is higher in NAFLD than in matched controls – Driven by the association between NAFLD and Met. S components • CVD should be identified in NAFLD, regardless of traditional risk factors • CVD and metabolic risk factors are also reported in adolescents and children with NAFLD Recommendations Grade of evidence Screening of the cardiovascular system is mandatory in all individuals with NAFLD because CV complications frequently dictate the outcome EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Grade of recommendation A 1
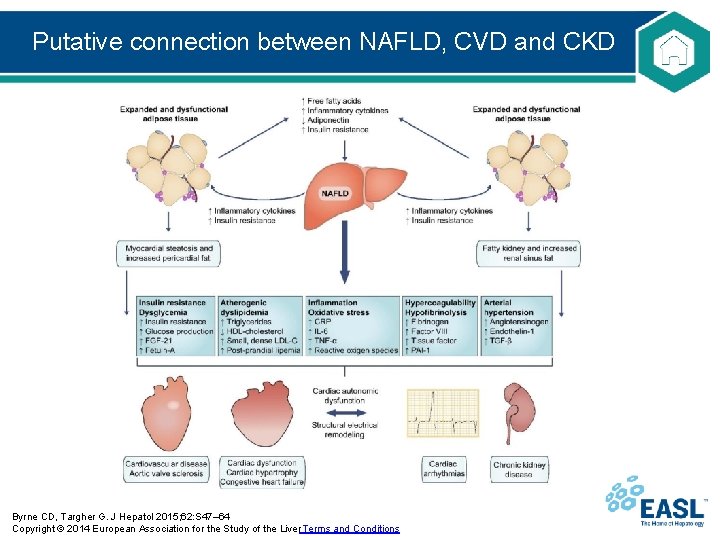
Putative connection between NAFLD, CVD and CKD Byrne CD, Targher G. J Hepatol 2015; 62: S 47– 64 Copyright © 2014 European Association for the Study of the Liver Terms and Conditions

Natural history and complications: HCC • Cumulative incidence of NAFLD-associated HCC varies according to study population • Large number of NAFLD cases at risk of HCC makes systematic surveillance largely impracticable – PNPLA 3 rs 738409 C>G gene polymorphism is associated with increased HCC risk – However, HCC surveillance in NAFLD is not yet considered cost effective Recommendations Grade of evidence Grade of recommendation Although NAFLD is a risk factor for HCC, which may also develop in the pre-cirrhotic stage, and the risk is further increased by the presence of the PNPLA 3 rs 738409 C>G polymorphism, no recommendation can be currently made on the timing of surveillance and its cost effectiveness EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 B 1

Treatment: diet and lifestyle changes • Epidemiology suggests a close relationship between an unhealthy lifestyle and NAFLD • Diet and lifestyle changes are mandatory in all patients – Modest weight loss reduces liver fat, improves hepatic IR, and can result in NASH regression – Weight loss of 7% is associated with histological improvement Recommendations Grade of evidence Grade of recommendation Structured programmes aimed at lifestyle changes towards healthy diet and habitual physical activity are advisable in NAFLD C 2 Patients without NASH or fibrosis should receive counselling for healthy diet and physical activity but no pharmacotherapy B 2 In overweight/obese NAFLD, a 7– 10% weight loss is the target of most lifestyle interventions, and results in improvement of liver enzymes and histology B 1 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Treatment: diet and lifestyle changes • A pragmatic, individually tailored approach is required – Dietary restriction PLUS – Progressive increase in aerobic exercise/resistance training Recommendations Grade of evidence Grade of recommendation Dietary recommendations should consider energy restriction and exclusion of NAFLD-promoting components (processed food, and food and beverages high in added fructose). The macronutrient composition should be adjusted according to the Mediterranean diet B 1 Both aerobic exercise and resistance training effectively reduce liver fat. The choice of training should be tailored based on patients’ preferences to be maintained in the long-term B 2 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Components of a lifestyle approach to NAFLD Fructose intake Energy restriction • • • Calorie restriction (500 1, 000/day) 7 10% weight loss target Long-term maintenance approach • Avoid fructose-containing food and drink Daily alcohol intake Coffee consumption • No liver-related limitations • Comprehensive lifestyle approach Strictly below 30 g men and 20 g women Macronutrient composition Physical activity • • Low-to-moderate fat Moderate-to-high carbohydrate Low-carbohydrate ketogenic diets or high protein EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 • 150 200 min/week moderate intensity in 3 5 sessions Resistance training to promote musculoskeletal fitness and improve metabolic factors

Treatment: pharmacotherapy • Treatment should be indicated in: – Progressive NASH – Early-stage NASH with risk of fibrosis progression* – Active NASH with high necroinflammatory activity • Treatment should reduce NASH-related mortality and progression to cirrhosis or HCC – Resolution of NASH-defining lesions accepted as surrogate endpoint • Safety and tolerability are prerequisites – Extensive comorbidities associated with significant polypharmacy and increased likelihood of DDIs Recommendations Grade of evidence Pharmacotherapy should be reserved for patients with NASH, particularly for those with significant fibrosis (stage F 2 and higher). Patients with less severe disease, but at high risk of disease progression could also be candidates for treatment *Age > 50 years, diabetes, Met. S, increased ALT EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Grade of recommendation B 1

Treatment: pharmacotherapy • Treatment should be indicated in: – Progressive NASH – Early-stage NASH with risk of fibrosis progression* – Active NASH with high necroinflammatory activity • Treatment should reduce NASH-related mortality and progression to cirrhosis or No HCCdrugs are approved for NASH – Resolution of NASH-defining accepted as surrogate endpoint No specific therapylesions can be recommended • Safety and tolerability are treatment prerequisitesis off label Any drug – Extensive comorbidities associated with significant polypharmacy and increased likelihood of DDIs Recommendations Grade of evidence Pharmacotherapy should be reserved for patients with NASH, particularly for those with significant fibrosis (stage F 2 and higher). Patients with less severe disease, but at high risk of disease progression could also be candidates for treatment *Age > 50 years, diabetes, Met. S, increased ALT EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 Grade of recommendation B 1

Treatment: pharmacotherapy • Insulin sensitizers – Little evidence of histological efficacy with metformin – PPAR agonist pioglitazone better than placebo • Improved all histological features except fibrosis • Achieved resolution of NASH more often • Antioxidants – Vitamin E may improve steatosis, inflammation and ballooning and resolve NASH in some patients • Concerns about long-term safety exist Recommendations Grade of evidence Grade of recommendation While no firm recommendations can be made, pioglitazone* or vitamin E† or their combination could be used for NASH B 2 The optimal duration of therapy is unknown; in patients with increased ALT at baseline, treatment should be stopped if there is no reduction in aminotransferases after 6 months of therapy‡ C 2 *Most efficacy data, but off-label outside T 2 DM; †Better safety and tolerability than pioglitazone in the short-term; ‡No recommendations can be made in patients with normal baseline ALT EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
Treatment: pharmacotherapy • Lipid-lowering agents – Statins have not been adequately tested in NASH Recommendations Grade of evidence Grade of recommendation Statins may be confidently used to reduce LDL cholesterol and prevent cardiovascular risk, with no benefits or harm to liver disease. Similarly, n-3 polyunsaturated fatty acids reduce both plasma and liver lipids, but there are no data to support their use specifically for NASH EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 B 1
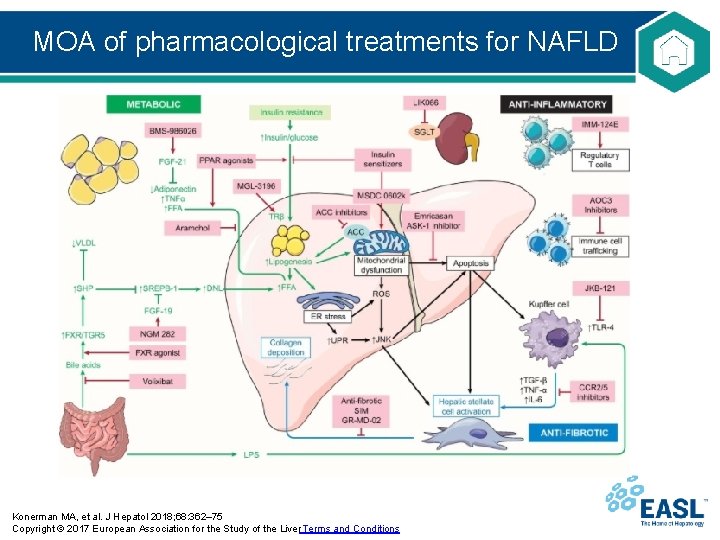
MOA of pharmacological treatments for NAFLD Konerman MA, et al. J Hepatol 2018; 68: 362– 75 Copyright © 2017 European Association for the Study of the Liver Terms and Conditions

Treatment: paediatric NAFLD • Diet and exercise training reduce steatosis, but do not affect ballooning, inflammation, and fibrosis • The long-term outcome of paediatric NASH remains poor – Drugs have shown beneficial effects but fibrotic lesions are refractory to treatment Recommendations Grade of evidence Grade of recommendation Diet and physical activity improve steatosis and hepatic inflammation in paediatric NAFLD, but no beneficial effects on fibrosis have ever been demonstrated. No safe drug treatment has proven effective on fibrosis in paediatric NAFLD EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402 B 1

Treatment: surgery • Bariatric surgery is an option in patients unresponsive to lifestyle changes and pharmacotherapy – Reduces weight and metabolic complications – Stable results in the long term • NAFLD-associated cirrhosis is one of the top three indications for LTx Recommendations for bariatric surgery Grade of evidence Grade of recommendation Bariatric surgery reduces liver fat and is likely to reduce NASH progression; prospective data have shown an improvement in all histological lesions of NASH, including fibrosis B 1 A 1 Recommendations for liver transplant LTx is an accepted procedure in patients with NASH and end-stage liver disease. Overall survival is comparable to other indications, despite a higher cardiovascular mortality. Patients with NASH and liver failure and/or HCC are candidates for liver transplantation EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
- Slides: 46