Biogeochemistry Dating relativeabsolute RelativeAbsolute dating Dating by assemblages

Biogeochemistry Dating (relative/absolute)

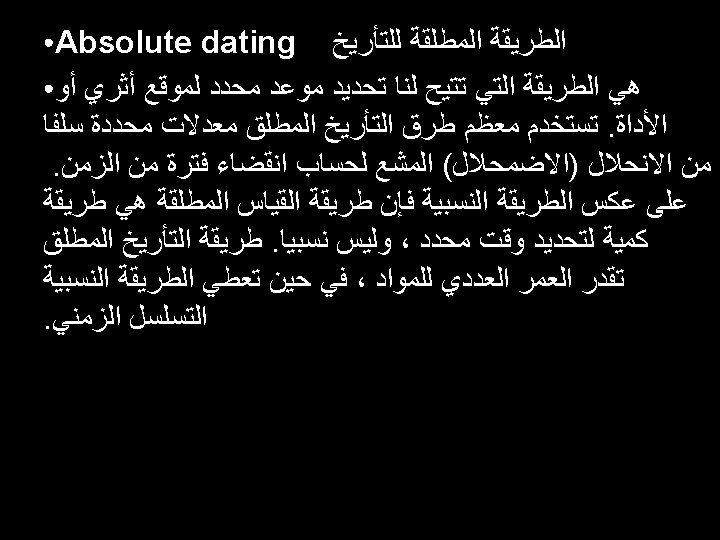
Relative/Absolute dating • Dating by assemblages of organisms is referred to as R D. • Dating with numerical numbers using radio active method referred to as A D.



Problem ﺍﻟﻤﺸﺎﻛﻞ ﺑﻌﺾ ﺍﻟﻤﺸﻌﺔ ﺍﻟﻤﻮﺍﺩ ﻣﻦ ﻣﺨﺘﻠﻔﺔ ﺗﻌﻴﻦ ﻃﺮﻕ ﺑﺎﺳﺘﺨﺪﺍﻡ ﺍﻟﻨﺎﺋﺞ • ﺗﻀﺎﺭﺏ ﺍﻟﻌﻴﻨﺔ ﻟﻨﻔﺲ Sometimes rocks that are thought to be much • older than other rocks (because they are buried several layers under them) date much younger than the rocks found above them! Often rocks that are known to be very young • (because they come from recent volcanoes whose eruption date is known) give dates using radioactive dating methods that are very old and therefore known to be erroneous

NOW • Research is not easy interpretation should be logic • It is not a matter of putting your samples in an instrument and having the result • As it has been said earlier result could be contradicting

C 14 • Carbon atom weighs 12 atomic mass units. • One in every trillion carbon atoms (in earth atmosphere TODAY) weighs 14 atomic unit. • C 14 is a radioactive atom • Half of the C 14 will decay in about 5730 years from the time it was formed to form Nitrogen (and half of the reminder will decay into N in another 5730 years and so on)

• N. B: Carbon dating used on organic material (not on rocks or even fossils), C 14 not used for dating on anything considered to be older than 50000 years. • When N in the earth’s upper atmosphere is struck by cosmic radiation it turns into C 14 • N(14) C(14) • Most C 14 quickly will combines with oxygen to form radioactive CO 2 • plant absorb CO 2 (small amount will be C(radioactive)O 2; C(14)O 2)

• Feeding on plants animals will take than some of the C 14 in their bodies • When animal and plant die they stop absorbing C 14 and this C 14 will slowly decay into nitrogen • By measuring the amount left in the dead animals and with some assumption scientist could determine the date of the animal death.

Assumptions Made in Radiocarbon Dating • 1 -A constant rate of decay - i. e. the half-life of carbon-14 has always been 5, 730 years • 2 -No loss or gain of parent or daughter - i. e. the specimen being dated has not taken in or lost any carbon-14 since the time it died (e. g through contamination).

• 3 - Known amounts of daughter at the start. This requires that the following things be true: • – The ratio of carbon-14 to carbon-12 that exists in the earth’s atmosphere today has always been the same. Has it always been the same? • – The ratio of carbon-14 to carbon-12 that exists in all living things always matches the ratio that exists in the earth’s atmosphere. Does it?






- Slides: 18