Psychrometrics Introduction to Food Engineering Psychrometrics n Thermodynamic


















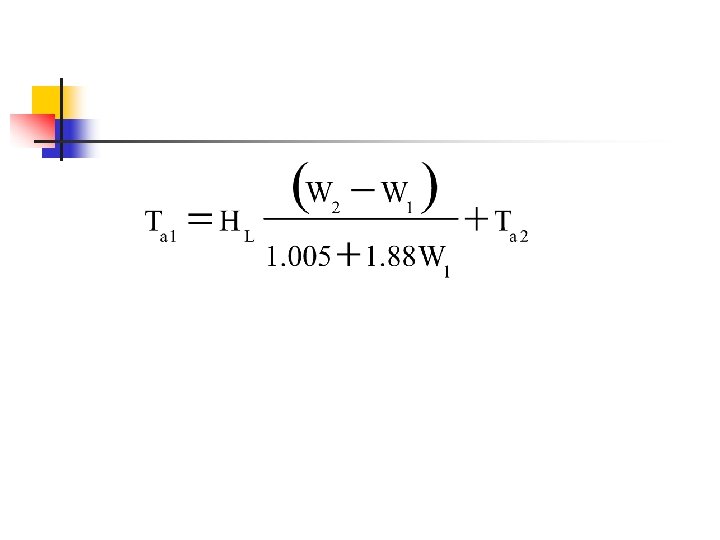








- Slides: 30

Psychrometrics Introduction to Food Engineering

Psychrometrics n Thermodynamic properties of gas-vapor mixtures (air-water(

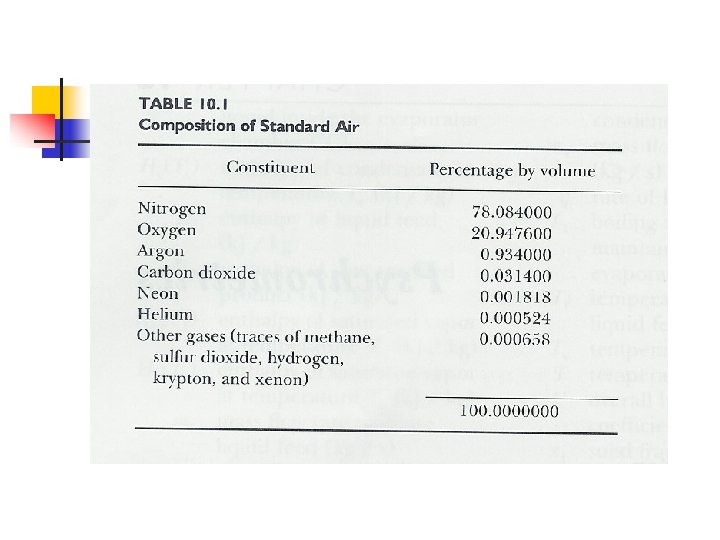
Properties of Dry Air n Composition n N 2 78 %, O 2 20. 9 % Standard dry air MW = 28. 9645 Gas constant for dry air Ra = 287. 055 m 3. Pa/kg. K


Properties of Dry Air n Specific Volume Ra = gas constant TA = absolute temperature (K) Pa = partial pressure of dry air

Properties of Dry Air n Specific Heat (Cpa) n n n At 1 atm (101. 325 k. Pa), T = - 40 – 60 C Average value 1. 005 k. J/kg. K Enthalpy (heat content) n Reference 1 atm, 0 C

Properties of Dry Air n Dry Bulb Temperature n Indicated by sensor

Properties of Water Vapor n n n Moist air = dry air + water vapor Vapor in the air is superheated steam at low pressure & temperature Moist air is clear or foggy MW of water = 18. 01534 Gas constant for water vapor Rw = 461. 52 m 3 Pa/kg. K

Properties of Water Vapor n Specific Volume of Water Vapor n Below 66 C vapor follows ideal gas law

Properties of Water Vapor n Specific Heat of Water Vapor n n Within – 71 to 184 C Cpw = 1. 88 k. J/kg. K Enthalpy of water vapor Ta = dry bulb temp

Properties of Air-Vapor Mixtures n Gibbs-Dalton Law n Up to 3 atm air-water mixtures follow perfect gas laws PB = barometric (total pressure) of moist air (k. Pa)

Dew-Point Temperature Water vapors in air = steam at low pressure n n Saturation temperature = dew point Obtained from steam table at partial pressure exerted by water vapor Below this temp, condensation of moisture

Humidity Ratio n Humidity Ratio (Moisture Content) or specific humidity, W (Kg water / ky dry air)

X = mole fraction

Relative Humidity n Relative to maximum amount of moisture at dry bulb temperature

Relative Humidity n Density of water vapor/density of saturated vapor at dry bulb temp of air

Humid Heat n n Of air-water vapor mixture Heat required to raise temp of 1 kg dry air + water vapor by 1 K (k. J/kg dry air. K) W = humidity ratio

Specific Volume n Volume of 1 kg dry air + water vapor

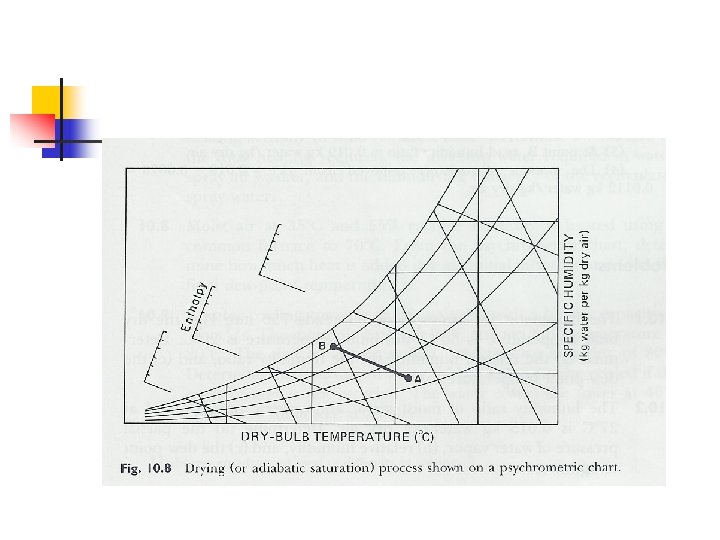
Adiabatic Saturation of Air n Evaporation of water by sensible heat of entering air
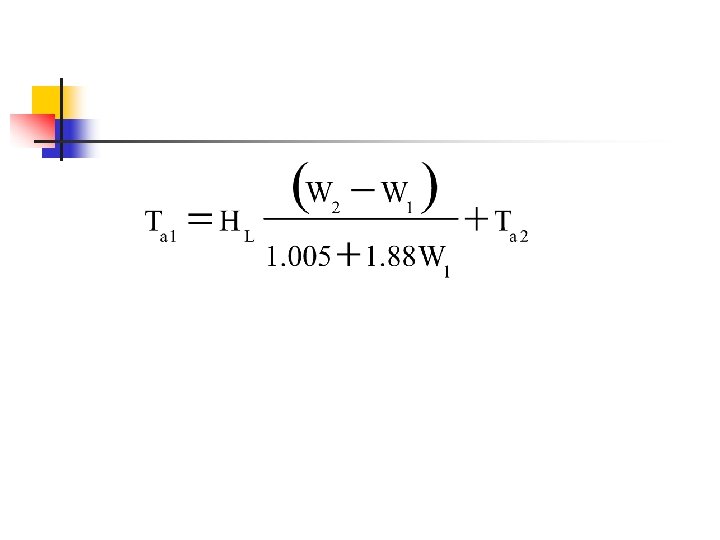

Wet-Bulb Temperature n Psychrometric wet bulb temperature n n Movement of air Thermodynamic wet bulb temperature

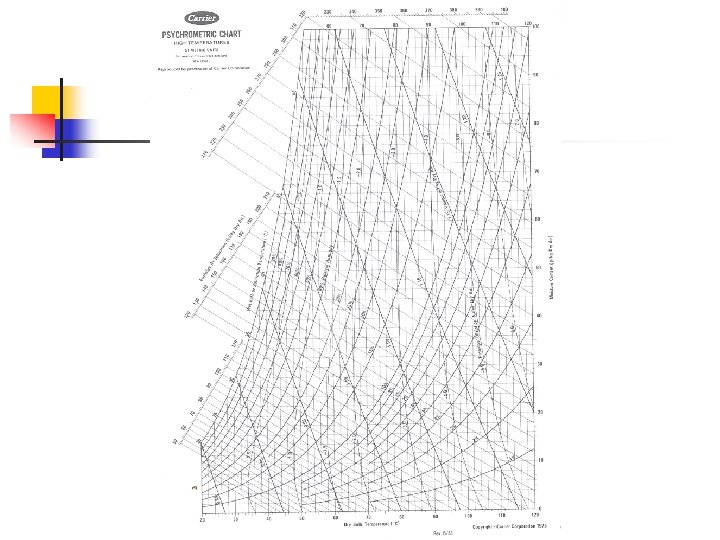
The Psychrometric Chart

Example n An air-vapor mixture is at 60 C dry bulb temp and 35 C wet bulb. Determine relative humidity, humidity ratio, specific volume, enthalpy and dew-point temp. n n RH = 20 %, W = 0. 026 kg/kg Enthalpy = 129 k. J/kg dry air Specific volume = 0. 98 m 3/kg dry air Dew-point temp = 29 C

Use of psychrometric chart to evaluate complex air-conditioning processes n Heating or Cooling of Air n Humidity ratio constant

Example n Calculate the rate of thermal energy required to heat 10 m 3/s of outside air at 30 C dry bulb temp and 80 % RH to a dry bulb temp of 80 C Mass flow rate = vol flow rate/ specific vol

Mixing of Air Inverse proportion

Drying n Adiabatic saturation process n n n Heat of evaporation is supplied only by drying air Dry bulb temp decreases, enthalpy constant ie. constant wet bulb Humidity ratio increases (gain moisture)


Example n Heated air at 50 C, 10 % RH is used to dry rice. Air exits under saturated condition. Determine amount of water removed per kg of dry air. n n W 1 = 0. 0078 kg/kg Follow constant enthalpy line W 2 = 0. 019 kg/kg Moisture removed = 0. 0112 kg/kg
