The Second Law of Thermodynamics Chapter 7 w




















- Slides: 37

The Second Law of Thermodynamics Chapter 7

w The first law of thermodynamics states that during any cycle that a system undergoes, the cyclic integral of the heat is equal to the cyclic integral of the work. w However, we know from our experience that because a proposed cycle does not violate the first law does not ensure that the cycle will actually occur. w It is this kind of experimental evidence that led to the formulation of the second law of thermodynamics.

w In its broader significance the second law acknowledges that processes proceed in a certain direction but not in the opposite direction. w A hot cup of coffee cools by virtue of heat transfer to the surroundings, but heat will not flow from the cooler surroundings to the hotter cup of coffee. w Gasoline is used as a car drives up a hill, but the fuel level in the gasoline tank cannot be restored to its original level when the car coasts down the hill.

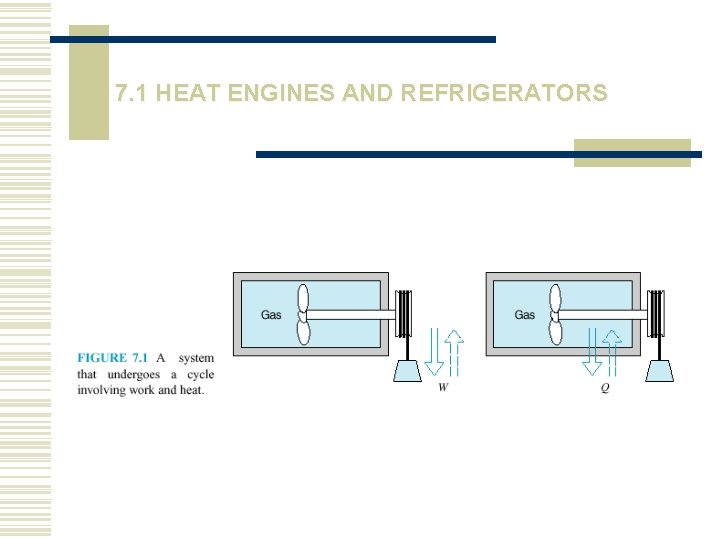
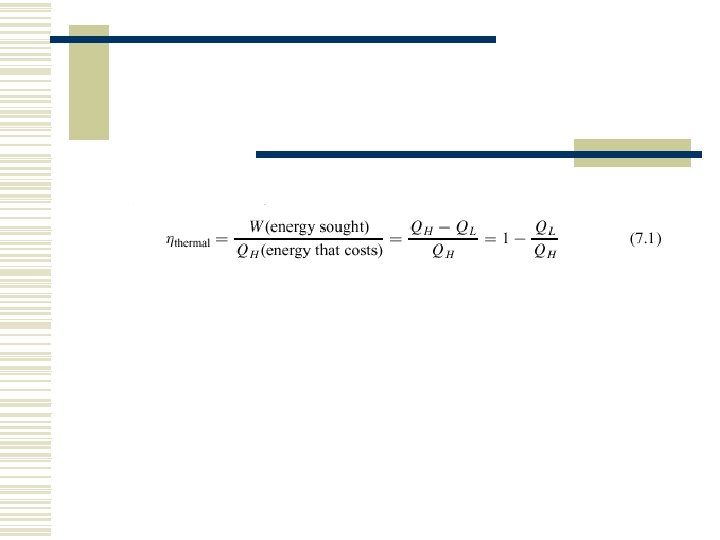
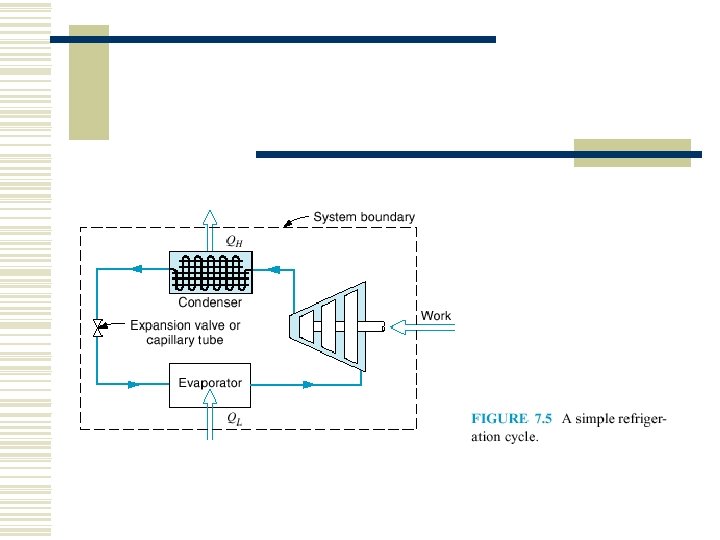
7. 1 HEAT ENGINES AND REFRIGERATORS


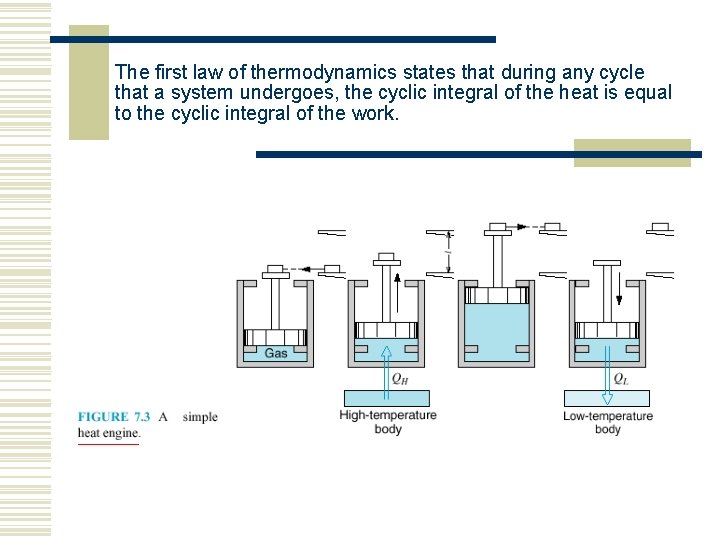
The first law of thermodynamics states that during any cycle that a system undergoes, the cyclic integral of the heat is equal to the cyclic integral of the work.






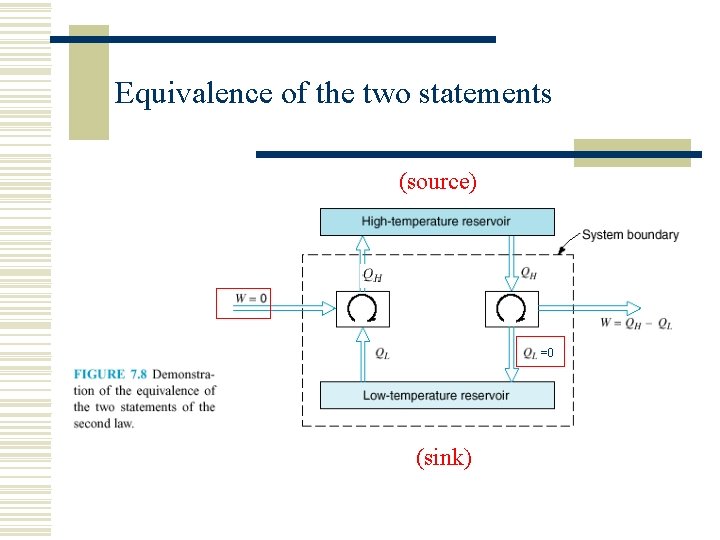
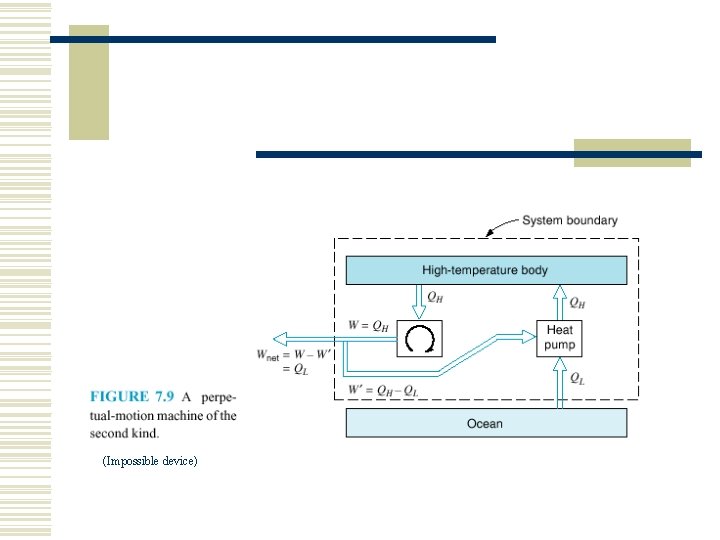
7. 2 THE SECOND LAW OF THERMODYNAMICS w Two classical statements of the second law n n Kelvin–Planck statement Clausius statement.

The Kelvin–Planck statement: w It is impossible to construct a device that will operate in a cycle and produce no effect other than the raising of a weight and the exchange of heat with a single reservoir. See Fig. 7. 6.

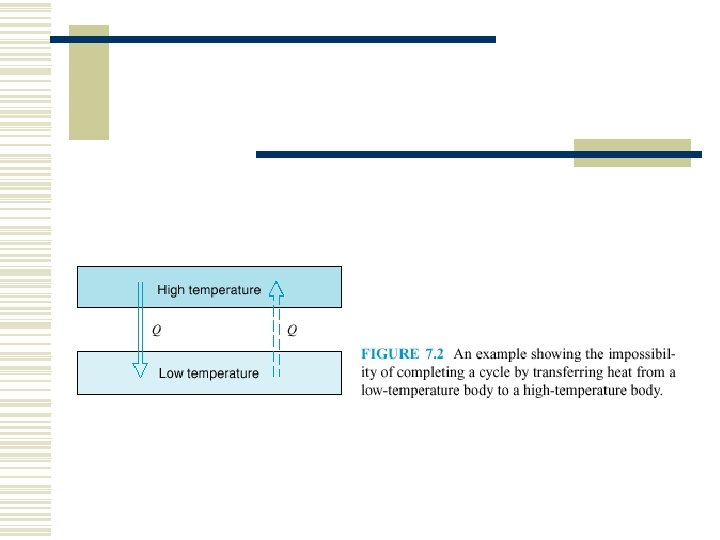
The Clausius statement: w It is impossible to construct a device that operates in a cycle and produces no effect other than the transfer of heat from a cooler body to a hotter body. See Fig. 7. 7.

Equivalence of the two statements (source) =0 (sink)

(Impossible device)

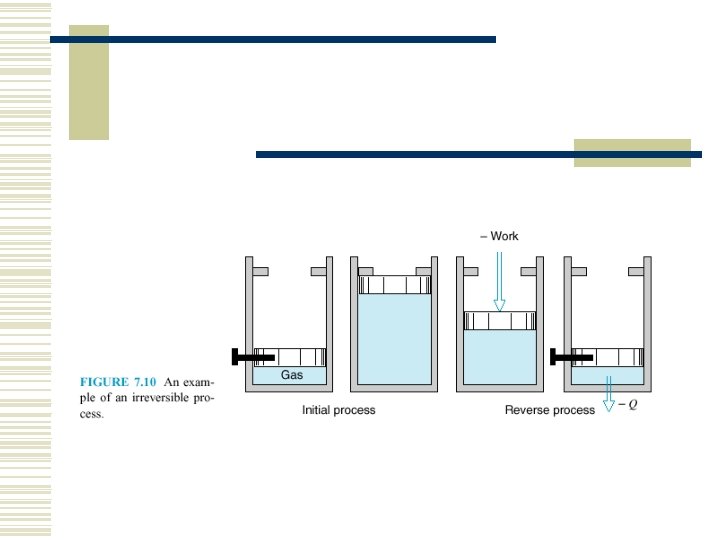
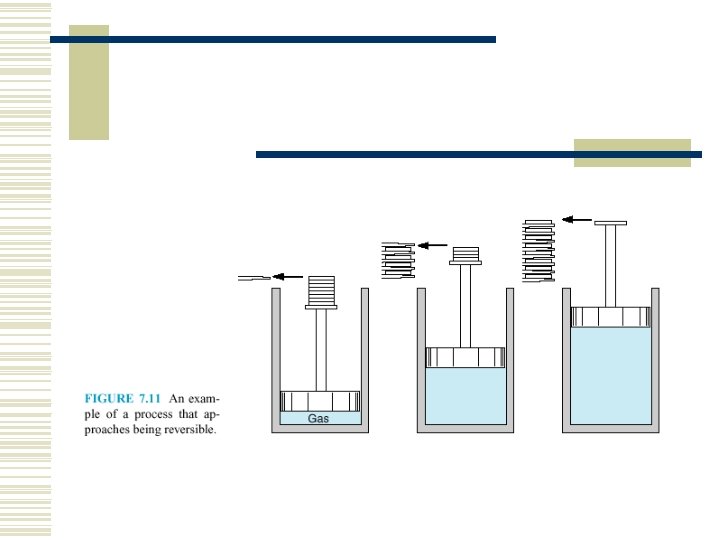
7. 3 THE REVERSIBLE PROCESS A reversible process for a system -- is defined as a process that once having taken place can be reversed and in so doing leave no change in either system or surroundings.



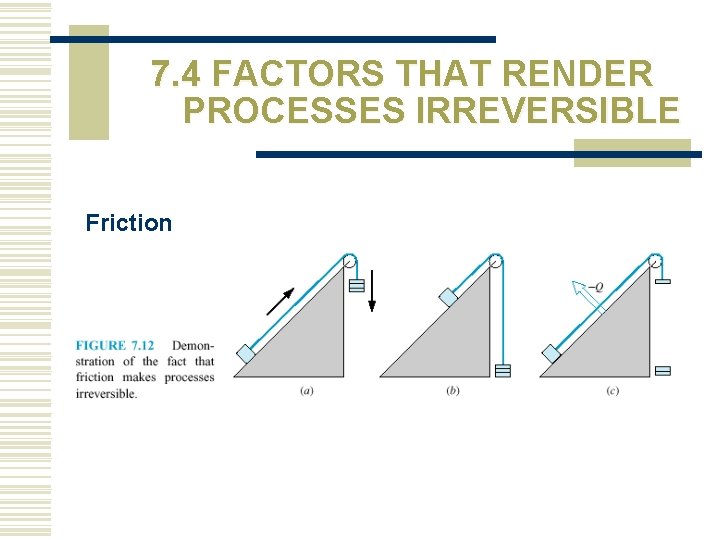
7. 4 FACTORS THAT RENDER PROCESSES IRREVERSIBLE Friction

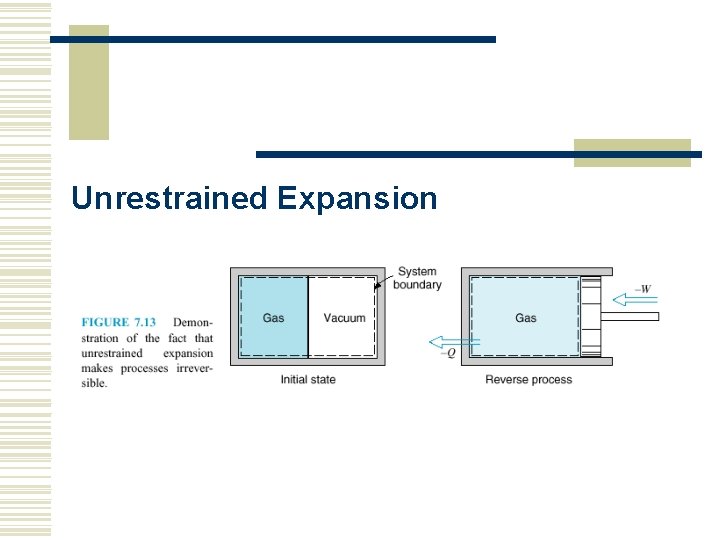
Unrestrained Expansion

w Heat Transfer through a Finite Temperature Difference w Mixing of Two Different Substances

w Other Factors n n n hysteresis effects i 2 R loss encountered in electrical circuits ordinary combustion

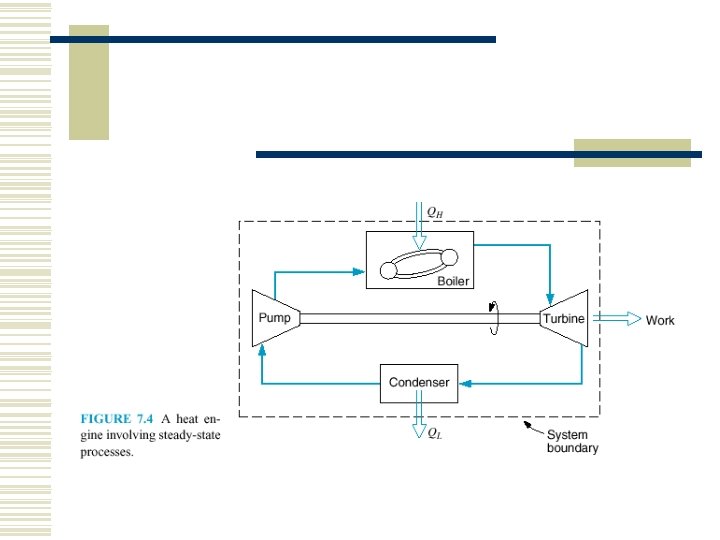
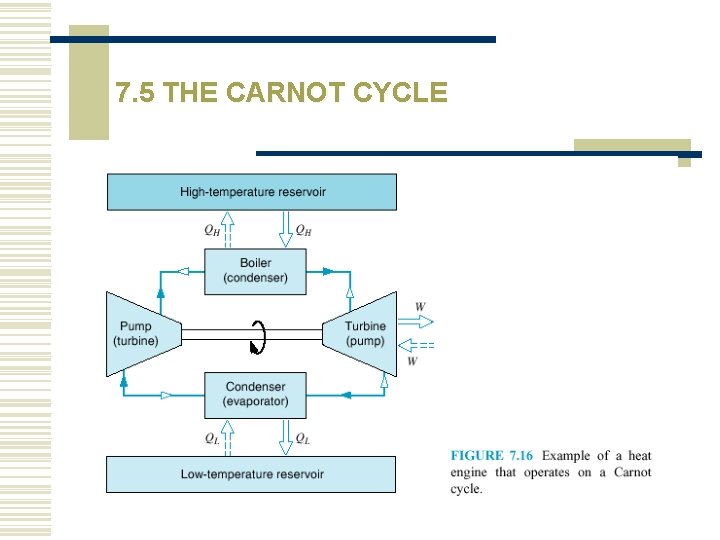
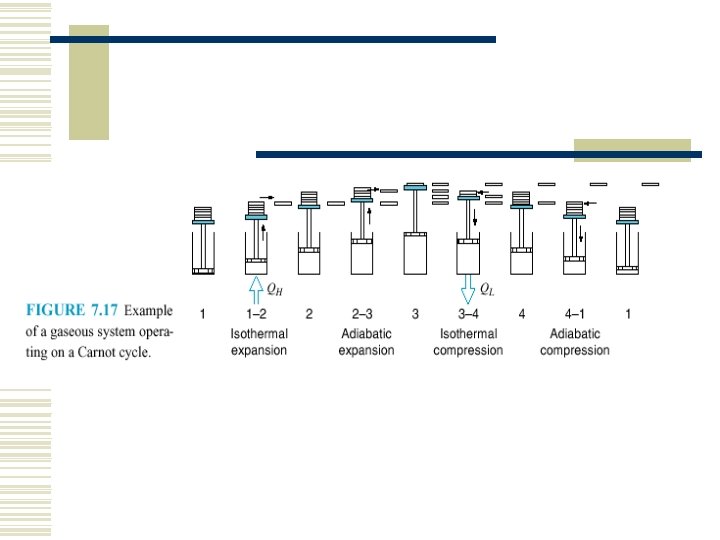
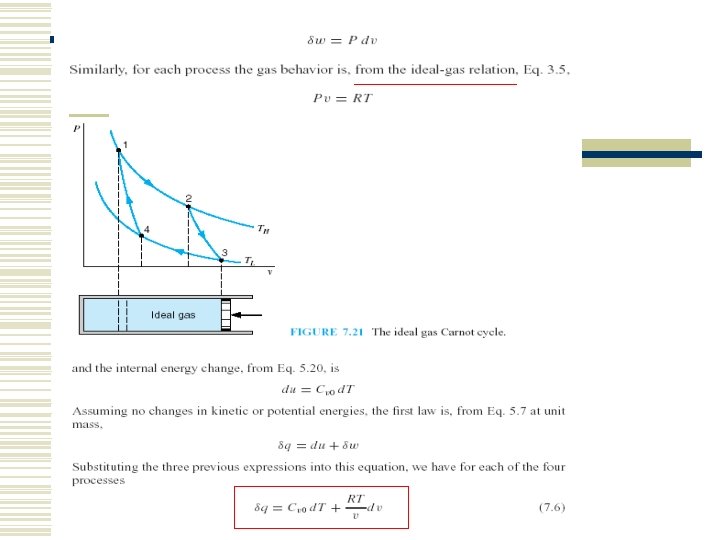
7. 5 THE CARNOT CYCLE



7. 6 TWO PROPOSITIONS REGARDING THE EFFICIENCY OF A CARNOT CYCLE w First Proposition It is impossible to construct an engine that operates between two given reservoirs and is more efficient than a reversible engine operating between the same two reservoirs.

w Second Proposition All engines that operate on the Carnot cycle between two given constant temperature reservoirs have the same efficiency.

The thermodynamic scale


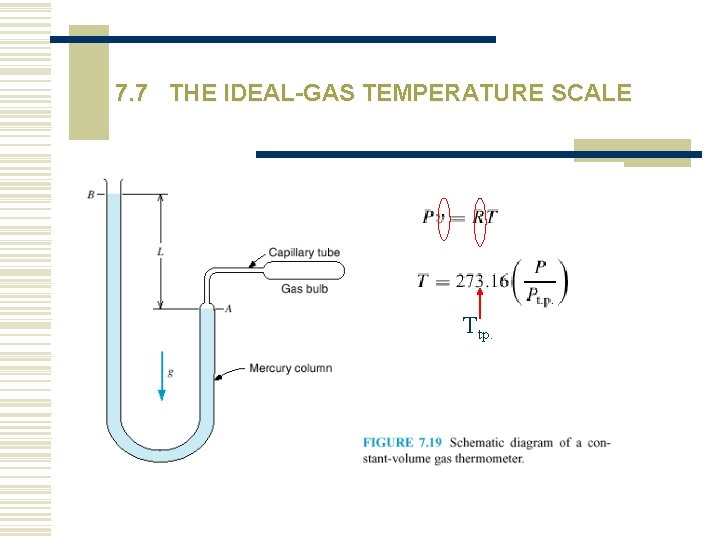
7. 7 THE IDEAL-GAS TEMPERATURE SCALE Ttp.

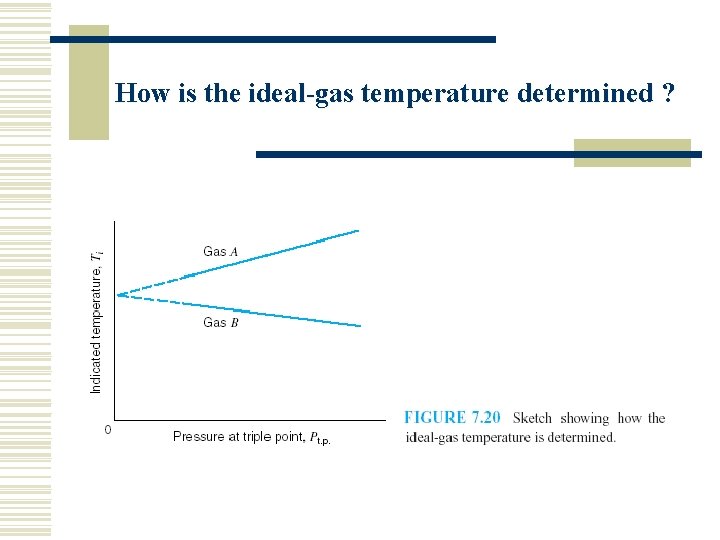
How is the ideal-gas temperature determined ?



7. 8 Ideal versus Real Machines

