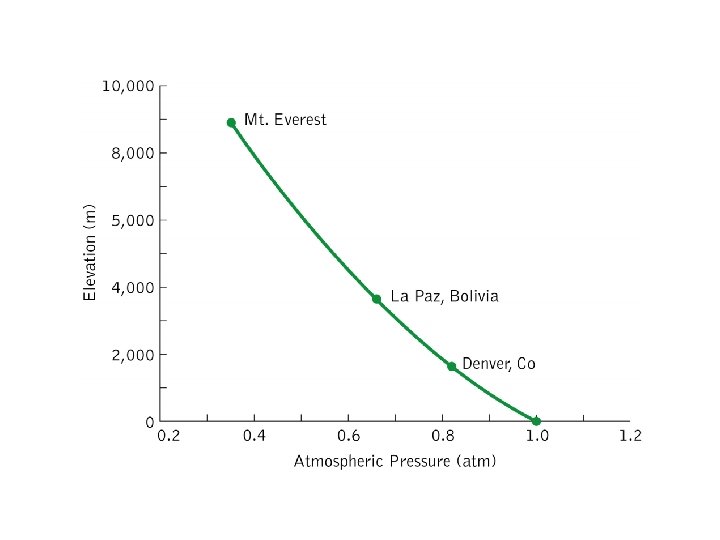
Pressure Conversions 1 atm 1 01325 x 105














- Slides: 17

Pressure Conversions • • • 1 atm = 1. 01325 x 105 Pa 1 bar = 1 x 105 Pa 1 millibar (mb) = 100 Pa 1 atm = 1. 01325 bar 1 atm = 760 torr 1 torr = 1 mm Hg



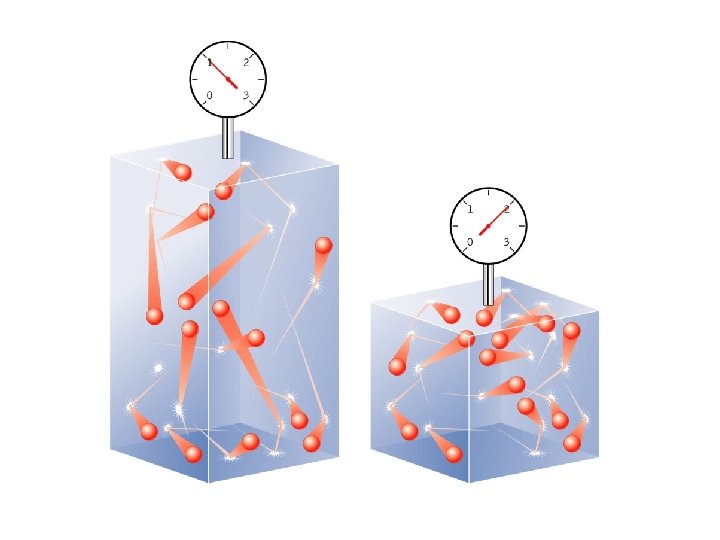
Change volume (V) and the pressure (P) will change (assuming that temperature and the number of molecules are constant) Boyle’s Law PV = constant Pressure and Volume are inversely proportional P 1 V 1 = P 2 V 2

A gas occupying a volume of 725 m. L at a pressure of 0. 970 atm is allowed to expand at constant temperature until its pressure reaches 0. 541 atm. What is the final volume? (725 ml)(0. 970 atm) = (V 2)(0. 541 atm) V 2 = 1300 ml or 1. 30 L

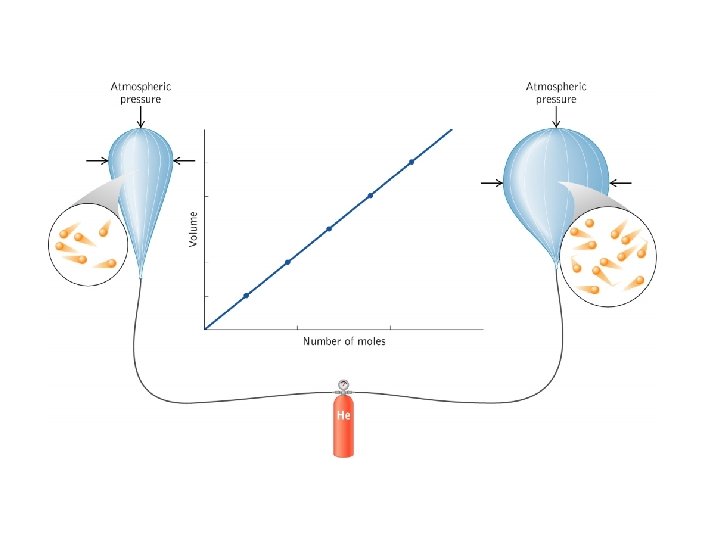
Change the amount of gas (n) and the volume (V) will change (assuming that temperature and pressure constant) Avogadro’s Law n V Number of Moles and Volume are proportional V 1 = V 2 n 1 n 2


Change the temperature (T) and the pressure (P) will change (assuming that the volume and number of moles are constant) Gay-Lussac’s Law T P Temperature and Pressure are proportional P 1 = P 2 T 1 T 2

An aerosol can is under a pressure of 3. 00 atm at 25 C. Directions on the can caution the user to keep the can in a place where the temperature does not exceed 52 C. What would the pressure of the gas in the aerosol can be at 52 C? 3. 00 atm 298 K = X atm 325 K X = 3. 27 atm

Change the temperature (T) and the volume (V) will change (assuming that the pressure and number of moles are constant) Charles’ Law T V Temperature and Pressure are proportional V 1 = V 2 T 1 T 2

A sample of neon gas has a volume of 752 m. L at 25 C. What volume will the gas occupy at 50 C if the pressure remains constant? 752 ml = 298 K X ml 323 K X = 815 ml

What we find is that everything is interrelated… The combined Gas Law n PV = constant T Such that P 1 V 1 P 2 V 2 = T 1 T 2

A He filled balloon has a volume of 50. 0 L at 15 C and 820 mm. Hg. What volume will it occupy at 650 mm. Hg and 10 C? (50. 0 L)(820 mm. Hg) = (X L)(650 mm. Hg) (288 K) (283 K) X = 62. 0 L

! l a e Id s a G w a L n PV = constant T By defining the constant we can convert the proportionality into “workable” equation PV = n. RT R is a constant which changes according to units, See Table 8. 1 on page 401 R = 0. 08206 L. atm/mol. K R = 8. 314 J/mol. K

Ideal Gas Conditions • • Negligible Interactions Negligible Particle Size High Temperature Low Pressure

Standard Temperature and Pressure (STP) 0 o. C 1 atm Under standard conditions, what is the volume of 1. 00 mol of gas? PV = n. RT (1 atm)( V ) = (1 mol)(0. 08206 L. atm/mol. K)(273 K) V = 22. 4 L

How many moles of gas are in my 600 ml Pepsi bottle? (assume that the room temperature is 22 o. C) PV = n. RT (1 atm)(0. 6 L) = n (0. 08206 L. atm/mol. K)(295 K) n = 0. 025 mols