Homework 17 posted Due Wed 16 Nov in












- Slides: 14

Homework 17 posted. Due Wed 16 Nov in class • Check on using Planck’s equation ? • De. Broglie’s big idea • Bohr uses De. Broglie idea to fix Rutherford atom • Bohr’s atom gets hammered too • The revenge of the experimentalists-singing spdf

Overview of how Rutherford’s atom gets fixed: 1) Explain photoelectric effect: (Einstein) (light is not a wave but acts sort of particle like) E/photon = hf=hc/ light 2) If light is particle-like then matter can act wave-like (De. Broglie) mv=h/ matter 3) Combine above + `old’ physics and make matter fit around a circle and all is fixed (Bohr) EH(J)= -2. 178*10 -18 n 2

…so what is light if not a wave ? ? ? “I GOT THIS…” Einstein’s image…. LIGHT IS A “PHOTON” =A MASSLESS BULLET OF ENERGY OLD NEW wave See also Figure 7. 5 page 302 Stream of photons

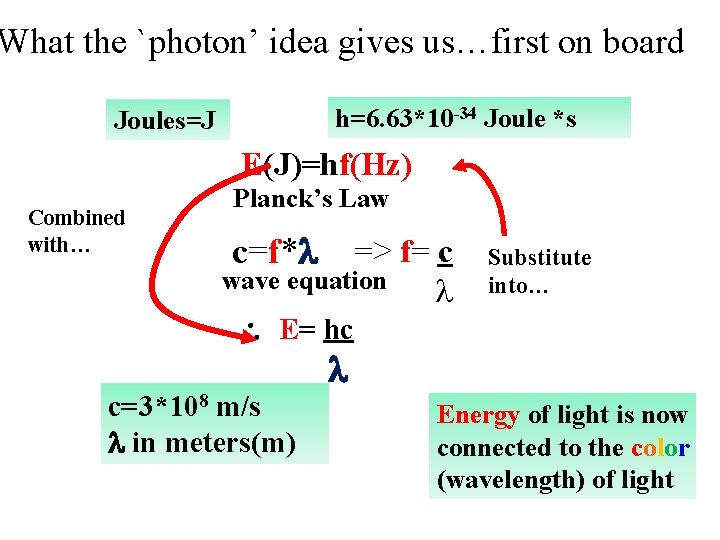
What the `photon’ idea gives us…first on board h=6. 63*10 -34 Joule *s Joules=J E(J)=hf(Hz) Combined with… Planck’s Law c=f* => f= c wave equation Substitute into… E= hc c=3*108 m/s in meters(m) Energy of light is now connected to the color (wavelength) of light

In-class practice on board: Planck’s law exercises …more practice ? ? E(J)= hf =6. 63*10 -34 *f(1/s) Or, since f= c/ E(J) = hc/ = 1. 989*10 -25 / (m)

De Broglie’s* hypothesis (pp. 63 -64) If light acts `particle-like’ …. then matter can act `wave-like. ’ *Louis-Victor-Pierre-Raymond, 7 th duc de Broglie

De. Broglie’s basic thinking connecting matter to waves: Rest energy of matter* 1) Steal from Einstein: 2) Steal from Planck: E=mc 2 energy of light `wave’ E = hf=hc/ 3) Combine them and simplify: E=mc 2=hc/ mc =h/ * Energy you can convert mass m to when it is not moving (at rest), e. g. , v=0…source of atomic bombs.

De Broglie’s Hypothesis (continued) His big idea…. assume a similar relationship exists when the mass is moving at a nonzero velocity v. => Replace c with v: mc =h/ mv =h/ Rearrange…to define the wavelength of a mass m moving at velocity v =>De. Broglie’s equation: = h/mv

E= hf Planck’s Law explains Photoelectric effect = h/mv De. Broglie’s equation connects matter to a hypothetical How do these fix the problems with Rutherford’s atom ? ? ?

Ask him ! (see also pp. 68 -72) Niels “the kid” Bohr at 27 soon after he makes his big theoretical breakthrough.

What the `kid’ does: Takes a dollop of Old School physics Mixes in New School Planck and De. Broglie equations: E= hf = h/mv Forces the wavelength to fit a circular orbit around the nucleus: see Figure 2. 11 pg. 73

From pure theory Bohr derives the Energy, EH, of the electron around Hydrogen and proposes the Bohr model of the atom: Eq. 2. 1 of text EH(J)= -2. 178*10 -18 n 2 n=1, 2, 3…are integers defining circular orbits around positive nucleus n=2 n=1 n=3

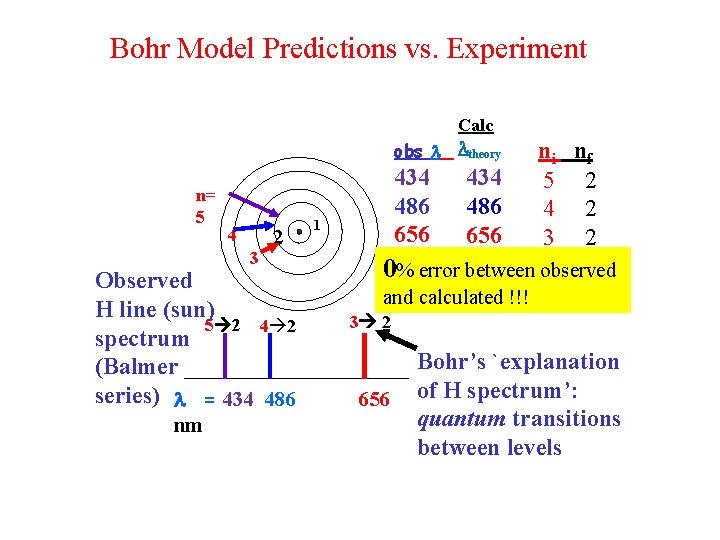
Bohr Model Predictions vs. Experiment Calc obs theory n= 5 4 3 2 Observed H line (sun) 5 2 4 2 spectrum (Balmer series) = 434 486 nm 434 486 656 1 434 486 656 ni nf 5 2 4 2 3 2 0% error between observed and calculated !!! 3 2 656 Bohr’s `explanation of H spectrum’: quantum transitions between levels

Let’s confirm that n=5 n=2 electron jump predicts =434 nm for H using Bohr model: h=6. 63*10 -34 J s c=3*108 m/s h*c= 1. 989*10 -25 J*m