Fatty Acid Oxidation A 16 C fatty acid








































- Slides: 46

Fatty Acid Oxidation

A 16 -C fatty acid with numbering conventions is shown. Most naturally occurring fatty acids have an even number of carbon atoms & unsaturated fatty acids are in the cis configuration The pathway for catabolism of fatty acids is referred to as the b-oxidation pathway, because oxidation occurs at the b-carbon (C-3).

Triacylglycerols (triglycerides) are the most abundant dietary lipids. Each triacylglycerol has a glycerol backbone to which are esterified 3 fatty acids Most triacylglycerols are “mixed. ” The 3 fatty acids differ in chain length & number of double bonds.

Lipases hydrolyze triacylglycerols, yielding glycerol and three fatty acids

Free fatty acids are transported in the blood bound to albumin, a plasma protein produced by the liver. Several proteins have been identified that facilitate transport of long chain fatty acids into cells

Fatty acid activation: Acyl-Co. A Synthases (Thiokinases) of ER & outer mitochondrial membranes catalyze activation of long chain fatty acids, esterifying them to coenzyme A. This process is ATP-dependent, & occurs in 2 steps. There are different Acyl-Co. A Synthases for fatty acids of different chain lengths.

Acyl-Co. A Synthases Exergonic PPi (P~P) hydrolysis, is catalyzed by Pyrophosphatase 2 ~P bonds of ATP are cleaved. The acyl-Co. A product includes one "~" thioester linkage.

Summary of fatty aid activation: fatty acid + ATP + HS-Co. A acyl-Co. A + AMP + 2 Pi

Fatty acid b-oxidation is considered to occur in the mitochondrial matrix. Fatty acids must enter the matrix to be oxidized. Fatty acyl-Co. A formed outside can pass through the outer mitochondrial membrane, but cannot penetrate the inner membrane.

Transfer of the fatty acid across the inner mitochondrial membrane involves carnitine. Carnitine Palmitoyl Transferases catalyze transfer of a fatty acid between the thiol of Coenzyme A and the hydroxyl on carnitine.

Carnitine-mediated transfer of the fatty acyl into the mitochondrial matrix is a 3 -step process: 1. Carnitine Palmitoyl Transferase I, an enzyme on the cytosolic surface of the outer mitochondrial membrane, transfers a fatty acid from Co. A to the OH on carnitine. 2. An antiporter in the inner mitochondrial membrane mediates exchange of carnitine for acylcarnitine.

3. Carnitine Palmitoyl Transferase II, an enzyme within the matrix, transfers the fatty acid from carnitine to Co. A. (Carnitine exits the matrix in step 2. ) The fatty acid is now esterified to Co. A in the matrix.

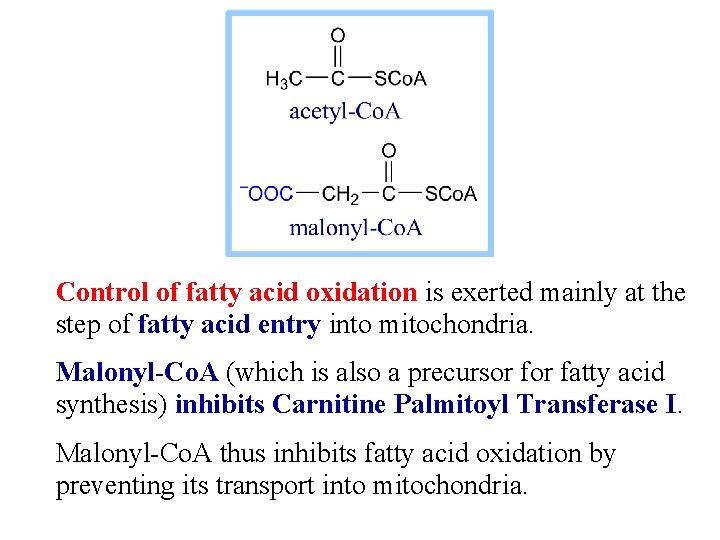
Control of fatty acid oxidation is exerted mainly at the step of fatty acid entry into mitochondria. Malonyl-Co. A (which is also a precursor fatty acid synthesis) inhibits Carnitine Palmitoyl Transferase I. Malonyl-Co. A thus inhibits fatty acid oxidation by preventing its transport into mitochondria.

b-Oxidation Pathway: Step 1. Acyl-Co. A Dehydrogenase catalyzes oxidation of the fatty acid of acyl-Co. A to produce a double bond between carbon atoms 2 & 3. There are different Acyl-Co. A Dehydrogenases for short (4 -6 C), medium (6 -10 C), long and very long (12 -18 C) chain fatty acids.

FAD is the prosthetic group that functions as e- acceptor for Acyl-Co. A Dehydrogenase. The reaction is stereospecific, yielding a trans double bond in enoyl-Co. A.

Step 2. Enoyl-Co. A Hydratase catalyzes stereospecific hydration of the trans double bond produced in the 1 st step, yielding L-hydroxyacyl. Coenzyme A.

Step 3. Hydroxyacyl-Co. A Dehydrogenase catalyzes oxidation of the hydroxyl in the b position (C 3) to a ketone. NAD+ is the electron acceptor.

Step 4. b-Ketothiolase catalyzes thiolytic cleavage. Thiol sulfur of Co. A attacks the bketo carbon Acetyl-Co. A is released, leaving the fatty acyl in thioester linkage to the Co. A -fatty acyl-Co. A (2 C less).

Summary of one round of the b-oxidation pathway: fatty acyl-Co. A + FAD + NAD+ + HS-Co. A fatty acyl-Co. A (2 C less) + FADH 2 + NADH + H+ + acetyl-Co. A The b-oxidation pathway is cyclic. The product, 2 carbons shorter, is the input to another round of the pathway. If, as is usually the case, the fatty acid contains an even number of C atoms, in the final reaction cycle butyryl-Co. A is converted to 2 molecules of acetyl-Co. A.

w FADH 2 & NADH produced during fatty acid oxidation are reoxidized by transfer of electrons to respiratory chain. Transfer of electrons in the respiratory chain leads to production of ATP w Acetyl-Co. A can enter Krebs cycle, yielding additional NADH, FADH 2, and ATP. Fatty acid oxidation is a major source of cell ATP.

The reactions presented accomplish catabolism of a fatty acid with an even number of C atoms & no double bonds. Additional enzymes deal with catabolism of fatty acids with an odd number of C atoms or with double bonds. w The final round of b-oxidation of a fatty acid with an odd number of C atoms yields acetyl-Co. A & propionyl-Co. A. Propionyl-Co. A is converted to the Krebs cycle intermediate succinyl-Co. A, by a pathway involving vitamin B 12.

w Most double bonds of naturally occurring fatty acids have the cis configuration. They are not correct substrates for Enoyl-Co. A hydratase, which acts only on trans compounds. Additional enzymes, isomerase and reductase , are required for oxidation of unsaturated fatty acids.

b-Oxidation of very long-chain fatty acids also occurs within peroxisomes. Within the peroxisome, FADH 2 generated by fatty acid oxidation is reoxidized producing hydrogen peroxide: FADH 2 + O 2 FAD + H 2 O 2 The peroxisomal enzyme Catalase degrades H 2 O 2: 2 H 2 O 2 2 H 2 O + O 2 These reactions produce no ATP. Once fatty acids are reduced in length within the peroxisomes they may shift to the mitochondria to be catabolized to acetyl. Co. A.

During fasting or carbohydrate starvation, oxaloacetate is depleted in liver due to gluconeogenesis. This impedes entry of acetyl-Co. A into Krebs cycle. Acetyl-Co. A in liver mitochondria is converted then to ketone bodies, acetoacetate & b-hydroxybutyrate.

Ketone body synthesis: b-Ketothiolase. The final step of the boxidation pathway runs backward. HMG-Co. A Synthase catalyzes condensation with a 3 rd acetate (from acetyl-Co. A). HMG-Co. A Lyase cleaves HMG-Co. A to yield acetoacetate & acetyl-Co. A.

b-Hydroxybutyrate Dehydrogenase catalyzes reversible interconversion of the ketone bodies acetoacetate & b-hydroxybutyrate. Ketone bodies are transported in the blood to other cells, where they are converted back to acetyl-Co. A for catabolism in Krebs cycle, to generate ATP. Ketone bodies thus function as an alternative fuel. Ketoacidosis is caused by excess of ketone bodies.

Fatty Acid Synthesis

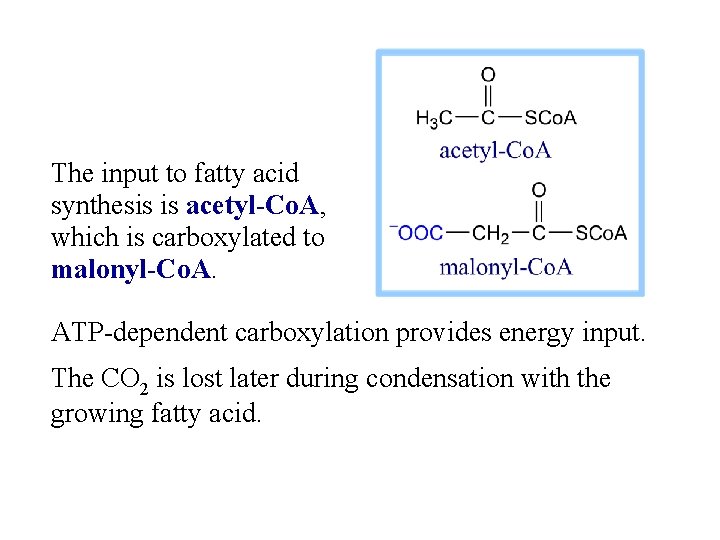
The input to fatty acid synthesis is acetyl-Co. A, which is carboxylated to malonyl-Co. A. ATP-dependent carboxylation provides energy input. The CO 2 is lost later during condensation with the growing fatty acid.

Acetyl-Co. A Carboxylase catalyzes the 2 -step reaction by which acetyl-Co. A is carboxylated to form malonyl. Co. A. As with other carboxylation reactions, the enzyme prosthetic group is biotin. ATP-dependent carboxylation of the biotin, carried out at one active site 1 , is followed by transfer of the carboxyl group to acetyl-Co. A at a second active site 2.

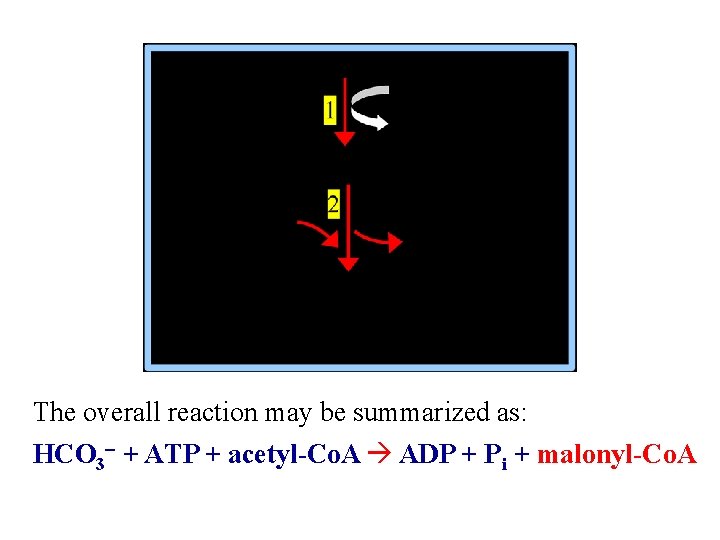
The overall reaction may be summarized as: HCO 3 - + ATP + acetyl-Co. A ADP + Pi + malonyl-Co. A

Biotin is linked to the enzyme by an amide bond between the terminal carboxyl of the biotin side chain and the e-amino group of a lysine residue.

Fatty acid synthesis from acetyl-Co. A & malonyl-Co. A occurs by a series of reactions that are: w catalyzed by individual domains of a very large polypeptide that includes an ACP domain. This multienzyme complex is called Fatty Acid Synthase NADPH serves as electron donor in the two reactions involving substrate reduction. The NADPH is produced mainly by the Pentose Phosphate Pathway.

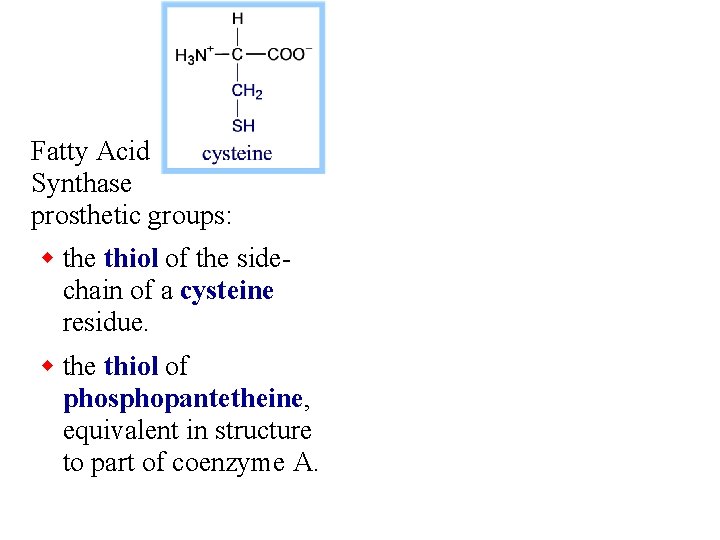
Fatty Acid Synthase prosthetic groups: w the thiol of the sidechain of a cysteine residue. w the thiol of phosphopantetheine, equivalent in structure to part of coenzyme A.

Phosphopantetheine (Pant) is covalently linked via a phosphate ester to a serine OH of the acyl carrier protein (ACP) of Fatty Acid Synthase.

Acetyl-Co. A-ACP Transacylase Malonyl-Co. A-ACP Transacylase The condensation reaction (step 3) involves decarboxylation of the malonyl, followed by attack of the acetyl (or acyl).

4. The b-ketone is reduced to an alcohol by e- transfer from NADPH. 5. Dehydration yields a trans double bond. 6. Reduction by NADPH yields a saturated chain.

Malonyl-Co. A-ACP Transacylase (repeat) Following transfer of the growing fatty acid from phosphopantetheine to cysteine sulfhydryl, the cycle begins again, with another malonyl-Co. A.

Product release: When the fatty acid is 16 carbon atoms long, a Thioesterase catalyzes hydrolysis of the thioester linking the fatty acid to phosphopantetheine. The 16 -C saturated fatty acid palmitate is the final product of the Fatty Acid Synthase complex.

Palmitate, a 16 -C saturated fatty acid, is the final product of the Fatty Acid Synthase reactions. Summary (ignoring H+ & water): acetyl-Co. A + 7 malonyl-Co. A + 14 NADPH palmitate + 7 CO 2 + 14 NADP+ + 8 Co. A Accounting for ATP-dependent synthesis of malonate: 8 acetyl-Co. A + 14 NADPH + 7 ATP palmitate + 14 NADP+ + 8 Co. A + 7 ADP + 7 Pi Fatty acid synthesis occurs in the cytosol. Acetyl-Co. A generated in mitochondria is transported to the cytosol via a shuttle mechanism involving citrate.

b-Oxidation & Fatty Acid Synthesis Compared

Elongation beyond the 16 -C length of the palmitate product of Fatty Acid Synthase occurs in mitochondria and endoplasmic reticulum (ER). w Fatty acid elongation within mitochondria involves the b-oxidation pathway running in reverse, but NADPH serves as electron donor for the final reduction step. w Fatty acids esterified to Co. A are substrates for the ER elongation machinery, which uses malonyl-Co. A as donor of 2 -carbon units. The reaction sequence is similar to Fatty Acid Synthase but individual steps are catalyzed by separate proteins. A family of enzymes designated Fatty Acid Elongases catalyze the initial condensation step for elongation of saturated or polyunsaturated fatty acids.

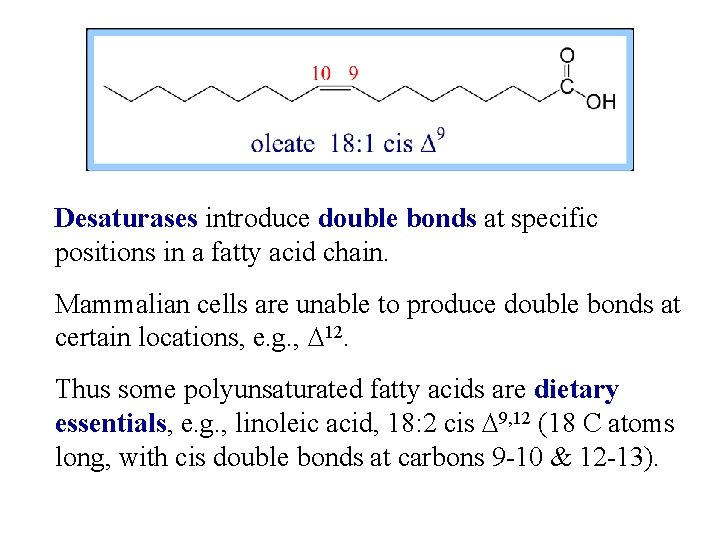
Desaturases introduce double bonds at specific positions in a fatty acid chain. Mammalian cells are unable to produce double bonds at certain locations, e. g. , D 12. Thus some polyunsaturated fatty acids are dietary essentials, e. g. , linoleic acid, 18: 2 cis D 9, 12 (18 C atoms long, with cis double bonds at carbons 9 -10 & 12 -13).

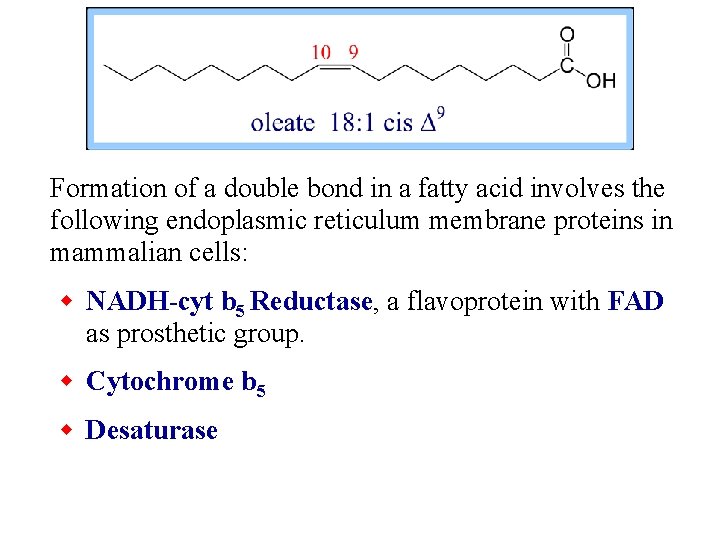
Formation of a double bond in a fatty acid involves the following endoplasmic reticulum membrane proteins in mammalian cells: w NADH-cyt b 5 Reductase, a flavoprotein with FAD as prosthetic group. w Cytochrome b 5 w Desaturase

The overall reaction for desaturation of stearate (18: 0) to form oleate (18: 1 cis D 9) is: stearate + NADH + H+ + O 2 oleate + NAD+ + 2 H 2 O There is a 4 -electron reduction of O 2 2 H 2 O as a fatty acid is oxidized to form a double bond. w 2 e- pass from NADH to the desaturase via the FAD-containing reductase & cytochrome b 5, the order of electron transfer being: NADH FAD cyt b 5 desaturase w 2 e- are extracted from the fatty acid as the double bond is formed.

Control of fatty acid synthesis is exerted mainly at acetyl-Co. A carboxylase step • Citrate & Insulin activate the enzyme • Acyl-Co. A & Glucagon & Epinephrine inhibite the enzyme

Thank you for your attention