Fatty acid metabolism Part I Fatty acid catabolism

Fatty acid metabolism Part I: Fatty acid catabolism 1

Fatty acids have four major physiological roles 1. Fatty Acids (FAs) are building blocks of phospholipids and glycolipids. 2. Many proteins are modified by the covalent attachment of FAs, which targets them to membrane locations. 3. FAs are fuel molecules that are stored as triacylglycerols (TAG). 4. FAs derivatives serve as hormones and intracellular messengers. 2
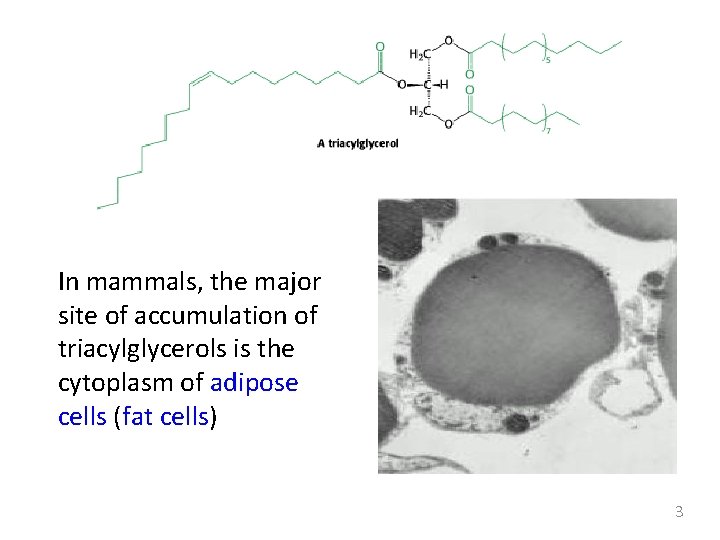
In mammals, the major site of accumulation of triacylglycerols is the cytoplasm of adipose cells (fat cells) 3
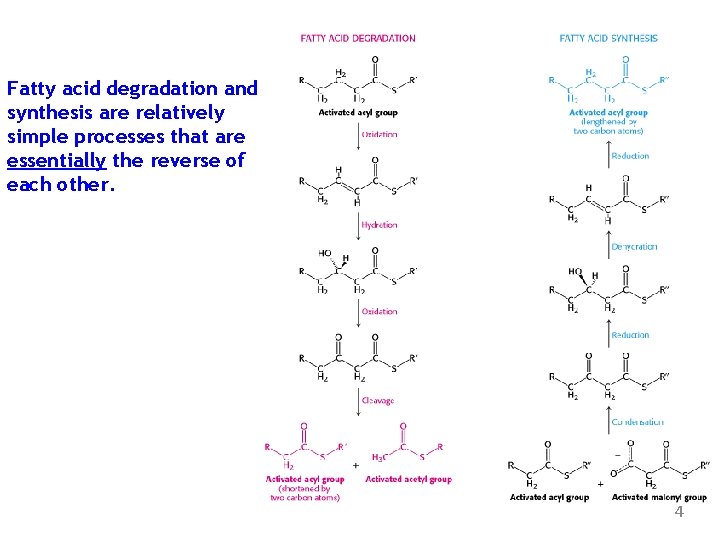
Fatty acid degradation and synthesis are relatively simple processes that are essentially the reverse of each other. 4

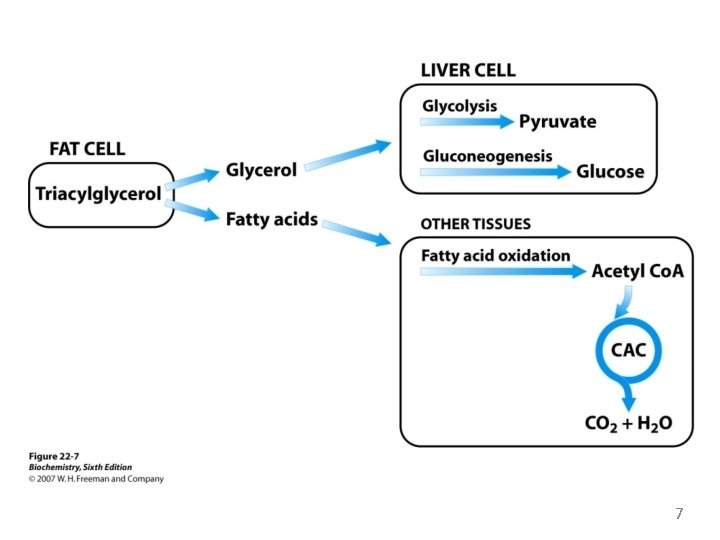
The Utilization of Fatty Acids as Fuel Requires Three Stages of Processing 1. The lipids must be mobilized: TAG are degraded to FAs and glycerol They are released from the adipose tissue and transported to the energy-requiring tissues. 2. At these tissues, the fatty acids must be activated and transported into mitochondria for degradation. 3. The fatty acids are broken down step-by-step into acetyl Co. A, which is then processed in the citric acid cycle. 5
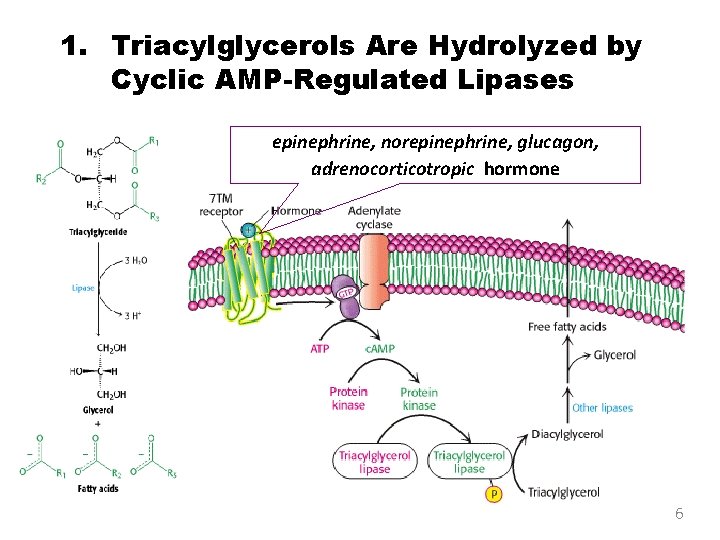
1. Triacylglycerols Are Hydrolyzed by Cyclic AMP-Regulated Lipases epinephrine, norepinephrine, glucagon, adrenocorticotropic hormone 6
7
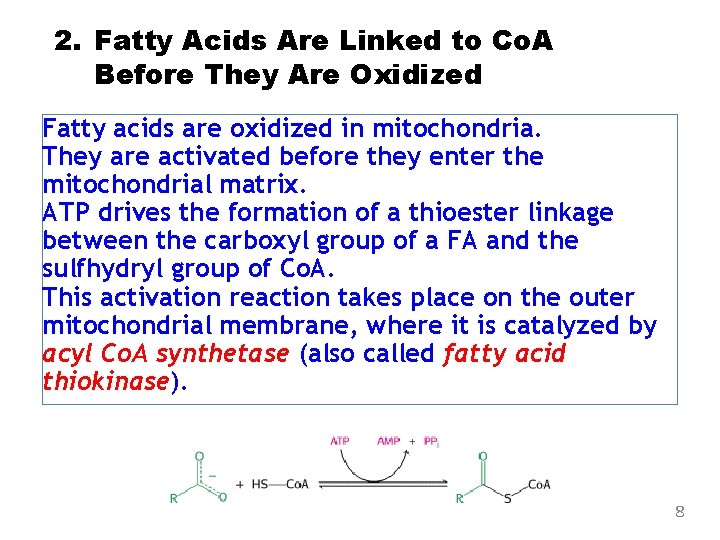
2. Fatty Acids Are Linked to Co. A Before They Are Oxidized Fatty acids are oxidized in mitochondria. They are activated before they enter the mitochondrial matrix. ATP drives the formation of a thioester linkage between the carboxyl group of a FA and the sulfhydryl group of Co. A. This activation reaction takes place on the outer mitochondrial membrane, where it is catalyzed by acyl Co. A synthetase (also called fatty acid thiokinase). 8
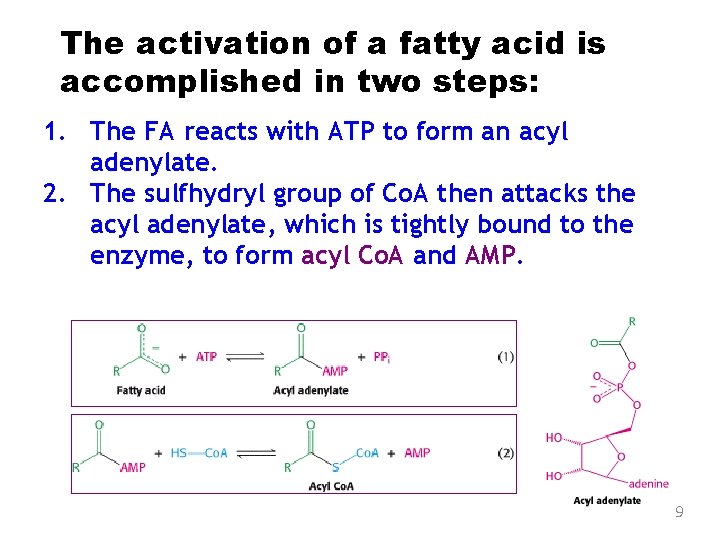
The activation of a fatty acid is accomplished in two steps: 1. The FA reacts with ATP to form an acyl adenylate. 2. The sulfhydryl group of Co. A then attacks the acyl adenylate, which is tightly bound to the enzyme, to form acyl Co. A and AMP. 9
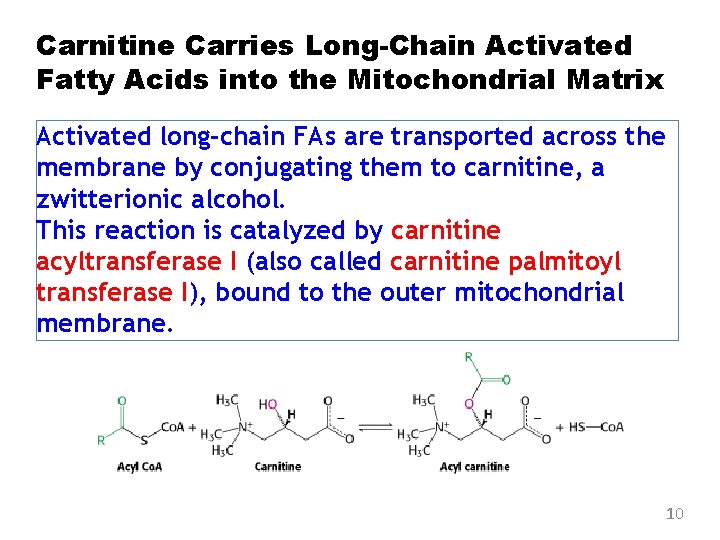
Carnitine Carries Long-Chain Activated Fatty Acids into the Mitochondrial Matrix Activated long-chain FAs are transported across the membrane by conjugating them to carnitine, a zwitterionic alcohol. This reaction is catalyzed by carnitine acyltransferase I (also called carnitine palmitoyl transferase I), bound to the outer mitochondrial membrane. 10
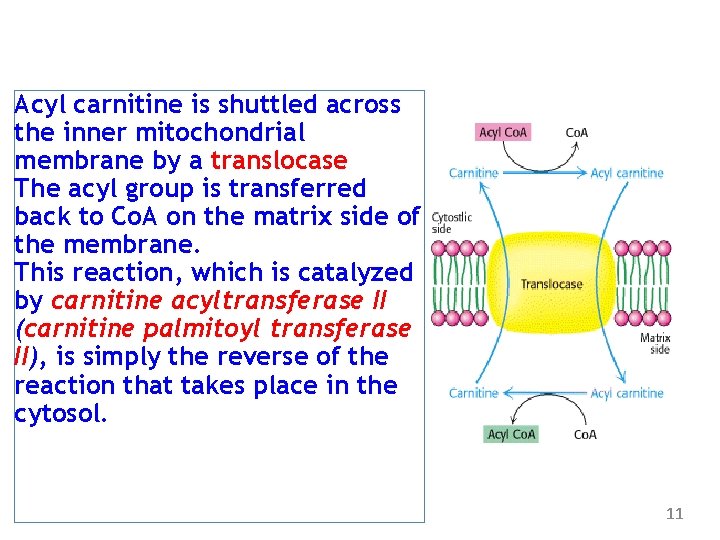
Acyl carnitine is shuttled across the inner mitochondrial membrane by a translocase The acyl group is transferred back to Co. A on the matrix side of the membrane. This reaction, which is catalyzed by carnitine acyltransferase II (carnitine palmitoyl transferase II), is simply the reverse of the reaction that takes place in the cytosol. 11

3. Fatty Acid Oxidation Through a sequence of 4 reactions: 1. 2. 3. 4. Oxidation by FAD Hydration by H 2 O Oxidation by NAD+ Thiolysis by Co. A 12
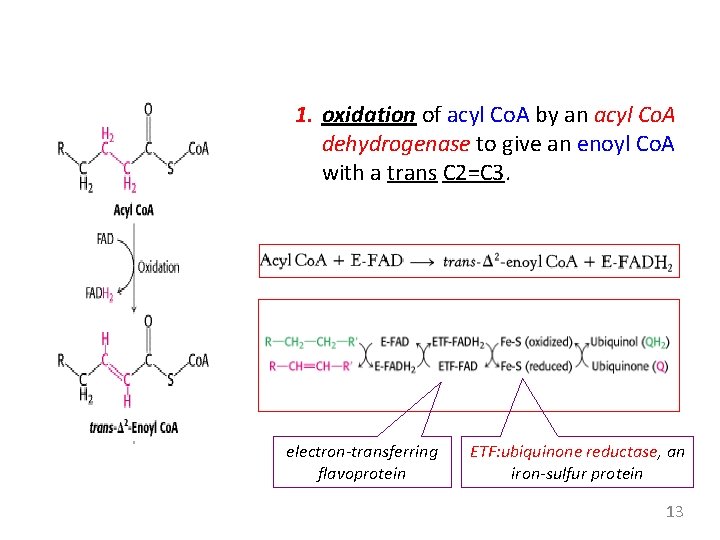
1. oxidation of acyl Co. A by an acyl Co. A dehydrogenase to give an enoyl Co. A with a trans C 2=C 3. electron-transferring flavoprotein ETF: ubiquinone reductase, an iron-sulfur protein 13

2. Stereospecific hydration of the C 2=C 3 by enoyl Co. A hydratase. 14
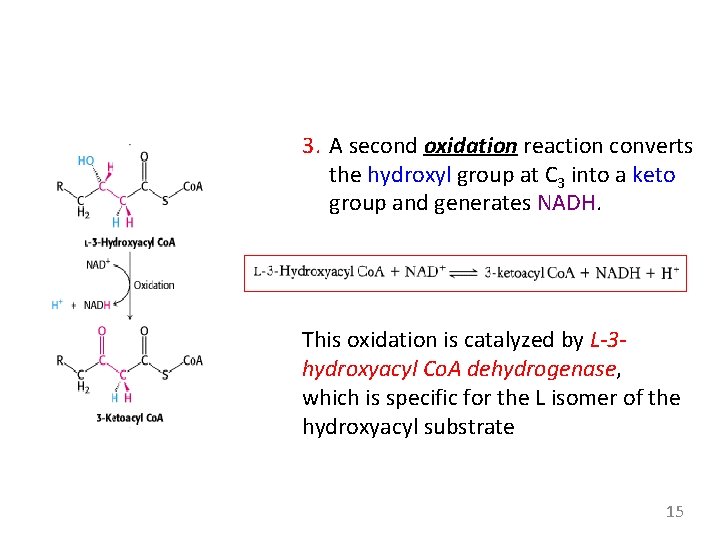
3. A second oxidation reaction converts the hydroxyl group at C 3 into a keto group and generates NADH. This oxidation is catalyzed by L-3 hydroxyacyl Co. A dehydrogenase, which is specific for the L isomer of the hydroxyacyl substrate 15

4. Thiolysis of 3 -ketoacyl Co. A by the thiol group of a second molecule of Co. A yields acetyl Co. A and an acyl Co. A shortened by two carbon atoms. This thiolytic cleavage is catalyzed by bketothiolase. 16

The shortened acyl Co. A then undergoes another cycle of oxidation, starting with the reaction catalyzed by acyl Co. A dehydrogenase 17
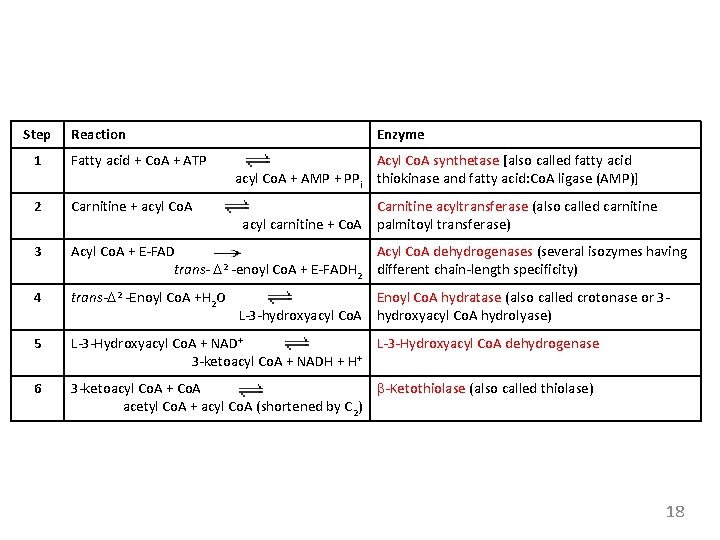
Step Reaction Enzyme 1 Fatty acid + Co. A + ATP 2 Carnitine + acyl Co. A 3 Acyl Co. A + E-FAD Acyl Co. A dehydrogenases (several isozymes having 2 trans- D -enoyl Co. A + E-FADH 2 different chain-length specificity) 4 trans-D 2 -Enoyl Co. A +H 2 O 5 L-3 -Hydroxyacyl Co. A + NAD+ L-3 -Hydroxyacyl Co. A dehydrogenase 3 -ketoacyl Co. A + NADH + H+ 6 3 -ketoacyl Co. A + Co. A b-Ketothiolase (also called thiolase) acetyl Co. A + acyl Co. A (shortened by C 2) Acyl Co. A synthetase [also called fatty acid acyl Co. A + AMP + PPi thiokinase and fatty acid: Co. A ligase (AMP)] Carnitine acyltransferase (also called carnitine acyl carnitine + Co. A palmitoyl transferase) Enoyl Co. A hydratase (also called crotonase or 3 L-3 -hydroxyacyl Co. A hydrolyase) 18

The Complete Oxidation of Palmitate Yields 106 Molecules of ATP The degradation of palmitoyl Co. A (C 16 -acyl Co. A) requires seven reaction cycles. In seventh cycle, C 4 -ketoacyl Co. A is thiolyzed to two molecules of acetyl Co. A. Hence, the stoichiometry of oxidation of palmitoyl Co. A is: 19

Approximately 2. 5 of ATP are generated per each of the 7 molecules of NADH. 1. 5 of ATP are formed for each of the 7 molecules of FADH 2. Recall that the oxidation of acetyl Co. A by the citric acid cycle yields 10 molecules of ATP. Hence, the number of ATP molecules formed in the oxidation of palmitoyl Co. A is: 10. 5 from the 7 molecules of FADH 2 17. 5 from the 7 molecules of NADH 80 from the 8 molecules of acetyl Co. A ~2 ATP are consumed in the activation of palmitate (the total is 106 ATP). 20

Certain Fatty Acids Require Additional Steps for Degradation 1. The oxidation of unsaturated fatty acids requires additional steps. 2. Fatty acids containing an odd number of carbon atoms yield a propionyl Co. A at the final thiolysis step. 3. Must be converted into an easily usable form by additional enzyme reactions. 21

1. Unsaturated fatty acids Palmitoleate, C 16 unsaturated fatty acid has one double bond between C-9 and C-10, (D 9). It is activated and transported across the inner mitochondrial membrane in the same way as saturated fatty acids. Palmitoleoyl Co. A then undergoes 3 cycles of degradation; carried out by the same enzymes as in saturated ones. 22
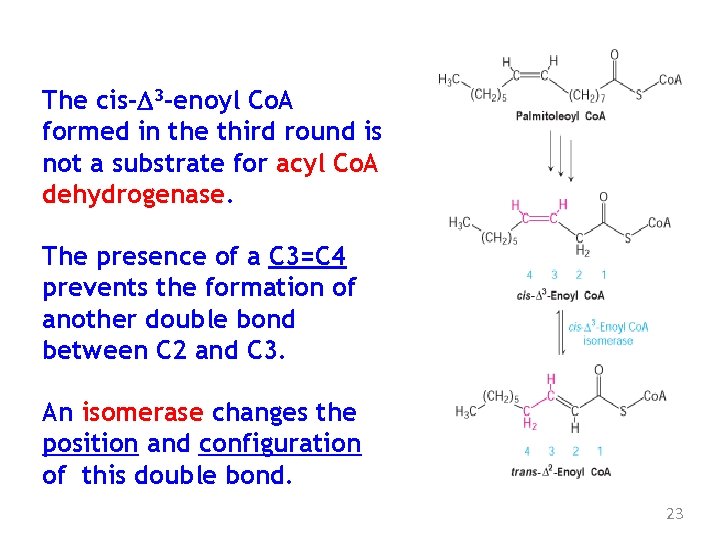
The cis-D 3 -enoyl Co. A formed in the third round is not a substrate for acyl Co. A dehydrogenase. The presence of a C 3=C 4 prevents the formation of another double bond between C 2 and C 3. An isomerase changes the position and configuration of this double bond. 23
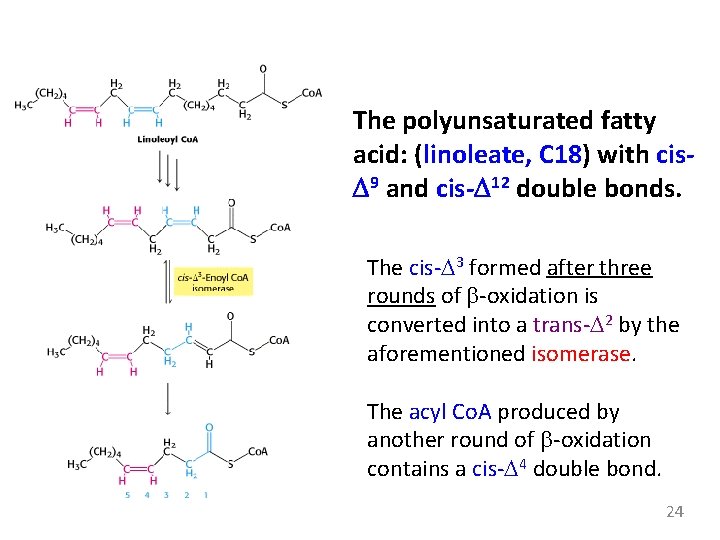
The polyunsaturated fatty acid: (linoleate, C 18) with cis. D 9 and cis-D 12 double bonds. The cis-D 3 formed after three rounds of b-oxidation is converted into a trans-D 2 by the aforementioned isomerase. The acyl Co. A produced by another round of b-oxidation contains a cis-D 4 double bond. 24
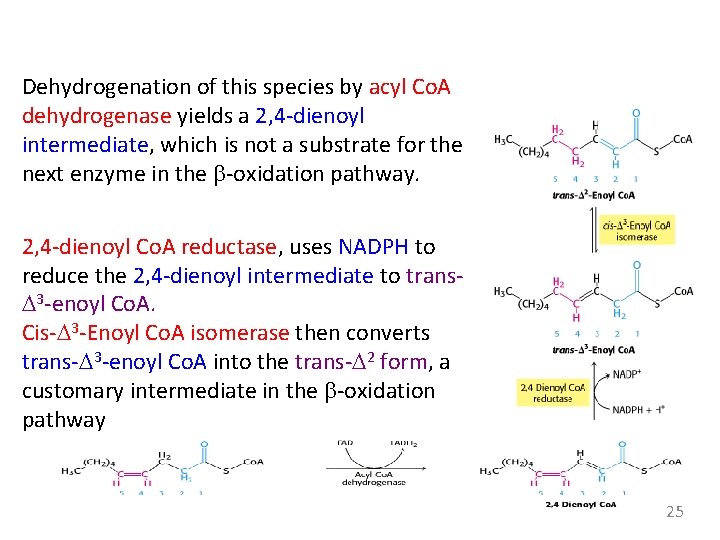
Dehydrogenation of this species by acyl Co. A dehydrogenase yields a 2, 4 -dienoyl intermediate, which is not a substrate for the next enzyme in the b-oxidation pathway. 2, 4 -dienoyl Co. A reductase, uses NADPH to reduce the 2, 4 -dienoyl intermediate to trans. D 3 -enoyl Co. A. Cis-D 3 -Enoyl Co. A isomerase then converts trans-D 3 -enoyl Co. A into the trans-D 2 form, a customary intermediate in the b-oxidation pathway 25

Only two extra enzymes needed for oxidation of any polyunsaturated fatty acid: Odd-numbered double bonds are handled by the isomerase. Even-numbered ones by both reductase and isomerase. 26

Odd-Chain Fatty Acids Yield Propionyl Coenzyme A in the Final Thiolysis Step Fatty acids having an odd number of carbon atoms are minor species. They’re oxidized in same way as those having an even number of C. Propionyl Co. A and acetyl Co. A, rather than two molecules of acetyl Co. A, are produced in the final round of degradation. 27
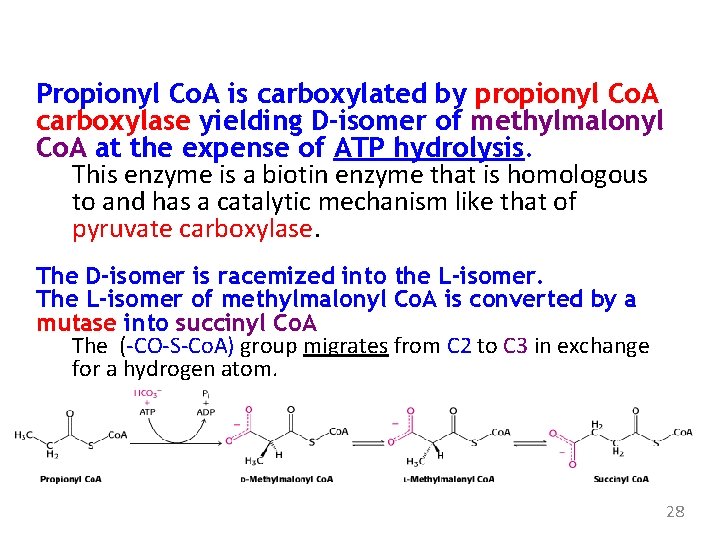
Propionyl Co. A is carboxylated by propionyl Co. A carboxylase yielding D-isomer of methylmalonyl Co. A at the expense of ATP hydrolysis. This enzyme is a biotin enzyme that is homologous to and has a catalytic mechanism like that of pyruvate carboxylase. The D-isomer is racemized into the L-isomer. The L-isomer of methylmalonyl Co. A is converted by a mutase into succinyl Co. A The (-CO-S-Co. A) group migrates from C 2 to C 3 in exchange for a hydrogen atom. 28

Fatty Acids Are Also Oxidized in Peroxisomes Some oxidation takes place in cellular organelles called peroxisomes that have high concentrations of catalase. Fatty acid oxidation in these organelles, stops at octanyl Co. A. It may serve to shorten long chains to make them better substrates of boxidation in mitochondria. 29

Peroxisomal oxidation differs from b-oxidation in the initial dehydrogenation reaction. In peroxisomes, a flavoprotein dehydrogenase transfers electrons to O 2 to yield H 2 O 2 Catalase is needed to convert the H 2 O 2 produced in the initial reaction into H 2 O and O 2. Subsequent steps are identical with their mitochondrial counterparts, although they are carried out by different isoforms of the enzymes. 30

Fats burn in the flame of carbohydrates The entry of acetyl Co. A into the citric acid cycle depends on the availability of oxaloacetate for the formation of citrate. Oxaloacetate is normally formed from pyruvate, the product of glycolysis, by pyruvate carboxylase The concentration of oxaloacetate is lowered if carbohydrate is unavailable or improperly utilized. 31

In fasting or diabetes Oxaloacetate is consumed to form glucose by the gluconeogenesis. Under these conditions, acetyl Co. A is diverted to the formation of acetoacetate and D-3 -hydroxybutyrate. Acetoacetate, D-3 -hydroxybutyrate, and acetone are often referred to as ketone bodies. 32
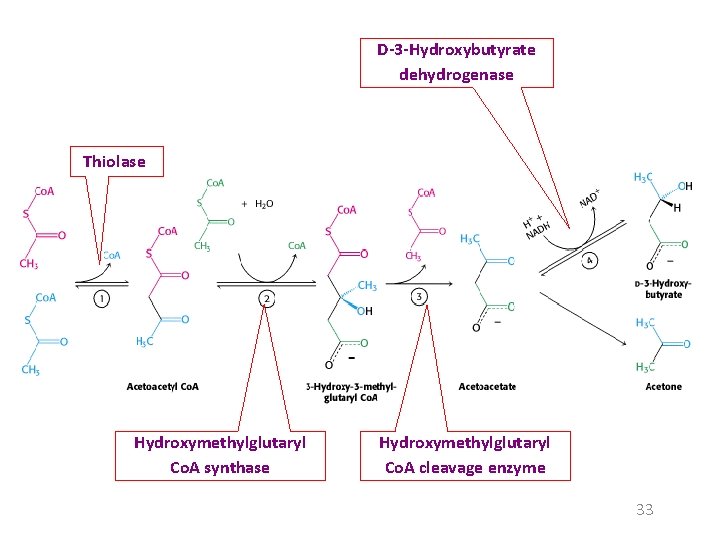
D-3 -Hydroxybutyrate dehydrogenase Thiolase Hydroxymethylglutaryl Co. A synthase Hydroxymethylglutaryl Co. A cleavage enzyme 33

Ketone Bodies Are a Major Fuel in Some Tissues Acetoacetate and 3 -hydroxybutyrate are produced mainly in the liver mitochondria. Acetoacetate and 3 -hydroxybutyrate are normal fuels of respiration and are quantitatively important as sources of energy. Heart muscle and the renal cortex use acetoacetate in preference to glucose. In contrast, glucose is the major fuel for the brain and red blood cells in normal conditions. However, the brain adapts to the utilization of acetoacetate during starvation and diabetes 34
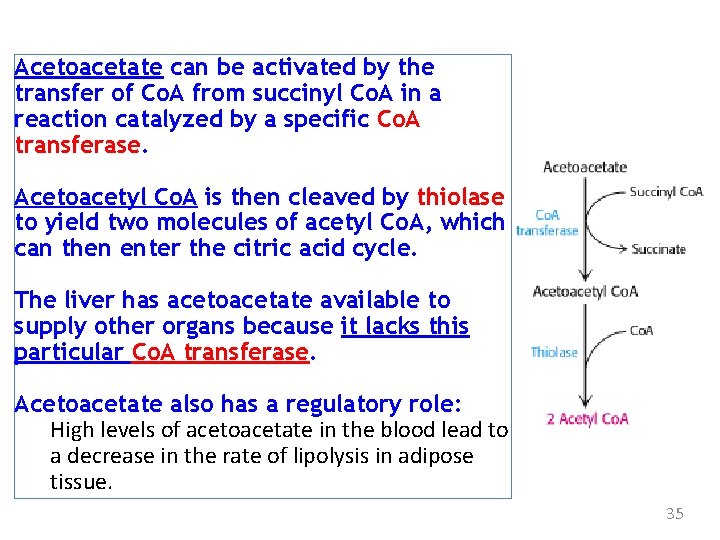
Acetoacetate can be activated by the transfer of Co. A from succinyl Co. A in a reaction catalyzed by a specific Co. A transferase. Acetoacetyl Co. A is then cleaved by thiolase to yield two molecules of acetyl Co. A, which can then enter the citric acid cycle. The liver has acetoacetate available to supply other organs because it lacks this particular Co. A transferase. Acetoacetate also has a regulatory role: High levels of acetoacetate in the blood lead to a decrease in the rate of lipolysis in adipose tissue. 35

Excessive production of ketone bodies in diabetes Rate of formation of ketone bodies is greater than the rate of their use, their levels begin to rise In the blood (ketonemia) and eventually in the urine (ketonuria) An elevation of the ketone body concentration in the blood results in acidemia. Ketoacidosis may also be seen in cases of fasting 36

37
Overview • Fatty acids are de-novo synthesized whenever an excess of calories is ingested. • The major source of carbon for the synthesis of fatty acids is dietary carbohydrate. • An excess of dietary protein also can result in an increase in fatty acid synthesis. In this case, the carbon source is amino acids that can be converted to acetyl-Co. A or tricarboxylic acid (TCA) cycle intermediates. • Fatty acid synthesis occurs primarily in the liver in humans. 38

Fatty Acids Are Synthesized and Degraded by Different Pathways Some important differences between the pathways are: 1. Synthesis takes place in the cytosol, in contrast with degradation, which takes place primarily in the mitochondrial matrix. 2. Intermediates in fatty acid synthesis are covalently linked to the sulfhydryl groups of an acyl carrier protein (ACP), whereas intermediates in fatty acid breakdown are covalently attached to the sulfhydryl group of coenzyme A. 3. The enzymes of fatty acid synthesis in higher organisms are joined in a single polypeptide chain called fatty acid synthase. In contrast, the degradative enzymes do not seem to be associated. 39

4. Ä 5. 6. Ä The growing fatty acid chain is elongated by the sequential addition of two-carbon units derived from acetyl Co. A. The activated donor of two carbon units in the elongation step is malonyl ACP. The elongation reaction is driven by the release of CO 2. The reductant in fatty acid synthesis is NADPH, whereas the oxidants in fatty acid degradation are NAD+ and FAD. Elongation by the fatty acid synthase complex stops on formation of palmitate (C 16). Further elongation and the insertion of double bonds are carried out by other enzyme systems. 40
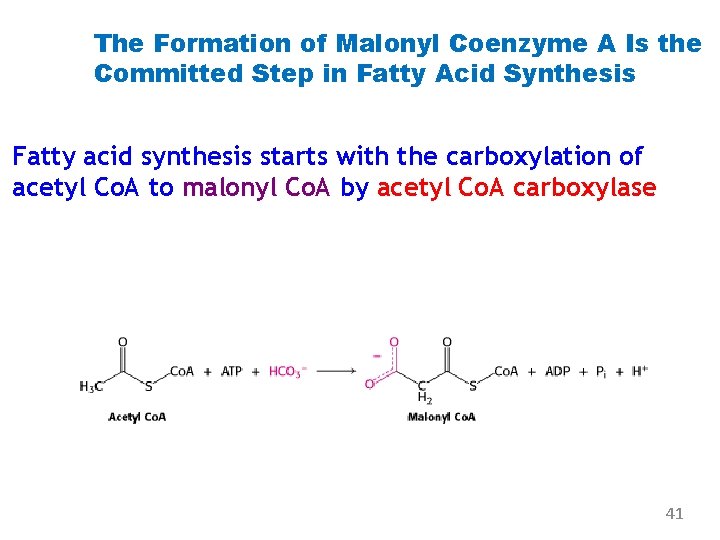
The Formation of Malonyl Coenzyme A Is the Committed Step in Fatty Acid Synthesis Fatty acid synthesis starts with the carboxylation of acetyl Co. A to malonyl Co. A by acetyl Co. A carboxylase 41
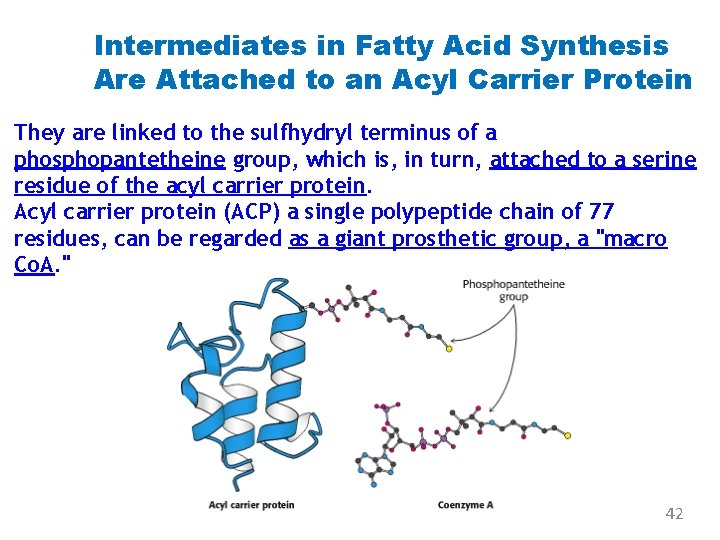
Intermediates in Fatty Acid Synthesis Are Attached to an Acyl Carrier Protein They are linked to the sulfhydryl terminus of a phosphopantetheine group, which is, in turn, attached to a serine residue of the acyl carrier protein. Acyl carrier protein (ACP) a single polypeptide chain of 77 residues, can be regarded as a giant prosthetic group, a "macro Co. A. " 42

The Elongation Cycle in Fatty Acid Synthesis The enzyme system that catalyzes the synthesis of saturated long-chain fatty acids from acetyl Co. A, malonyl Co. A, and NADPH is called the fatty acid synthase. The elongation phase of fatty acid synthesis starts with the formation of acetyl ACP and malonyl ACP. Acetyl transacylase and malonyl transacylase catalyze these reactions. 43

The decarboxylation of malonyl ACP contributes a substantial decrease in free 1 energy. In effect, ATP drives the condensation reaction: ATP is used to carboxylate acetyl Co. A to malonyl Co. A. The free energy stored in malonyl Co. A is released in the decarboxylation accompanying the formation of acetoacetyl ACP. 44
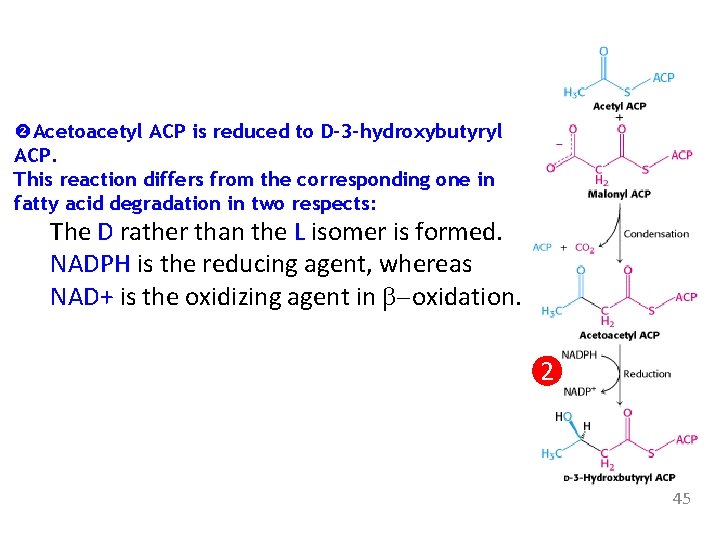
Acetoacetyl ACP is reduced to D-3 -hydroxybutyryl ACP. This reaction differs from the corresponding one in fatty acid degradation in two respects: The D rather than the L isomer is formed. NADPH is the reducing agent, whereas NAD+ is the oxidizing agent in b-oxidation. 2 45
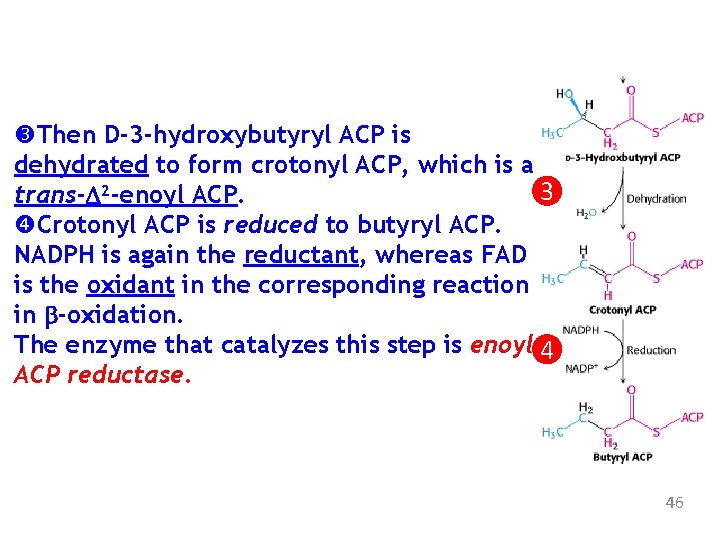
Then D-3 -hydroxybutyryl ACP is dehydrated to form crotonyl ACP, which is a 3 trans-D 2 -enoyl ACP. Crotonyl ACP is reduced to butyryl ACP. NADPH is again the reductant, whereas FAD is the oxidant in the corresponding reaction in b-oxidation. The enzyme that catalyzes this step is enoyl 4 ACP reductase. 46

In the second round of FA synthesis, butyryl ACP condenses with malonyl ACP to form a C 6 -b-ketoacyl ACP. The elongation cycles continue until C 16 -acyl ACP is formed. This intermediate is a good substrate for a thioesterase that hydrolyzes C 16 -acyl ACP to yield palmitate and ACP. 47

FAs with an odd number of carbon atoms are synthesized starting with propionyl ACP, which is formed from propionyl Co. A by acetyl transacylase. 48

Fatty Acids Are Synthesized by a Multifunctional Enzyme Complex in Eukaryotes Many eukaryotic multienzyme complexes are covalently linked enzymes in multifunctional proteins: 1. 2. 3. An advantage of this arrangement is that the synthetic activity of different enzymes is coordinated. A multienzyme complex consisting of covalently joined enzymes is more stable than one formed by noncovalent attractions. Intermediates can be efficiently handed from one active site to another without leaving the assembly. 49
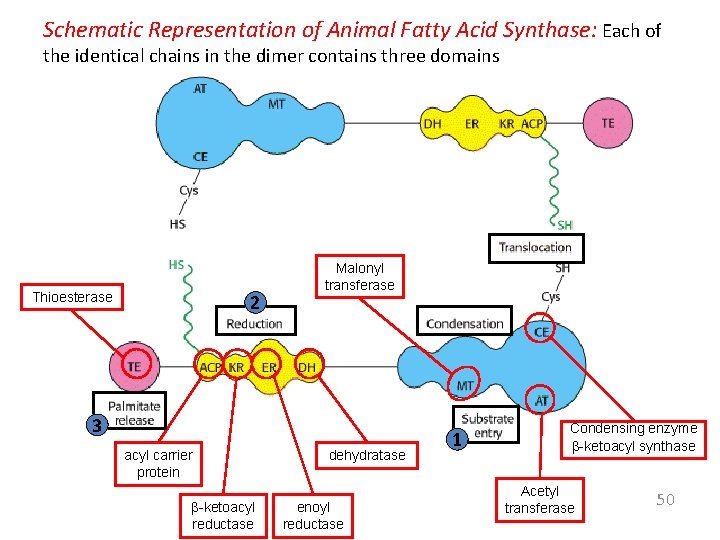
Schematic Representation of Animal Fatty Acid Synthase: Each of the identical chains in the dimer contains three domains Thioesterase 2 Malonyl transferase 3 acyl carrier protein b-ketoacyl reductase dehydratase enoyl reductase 1 Condensing enzyme b-ketoacyl synthase Acetyl transferase 50
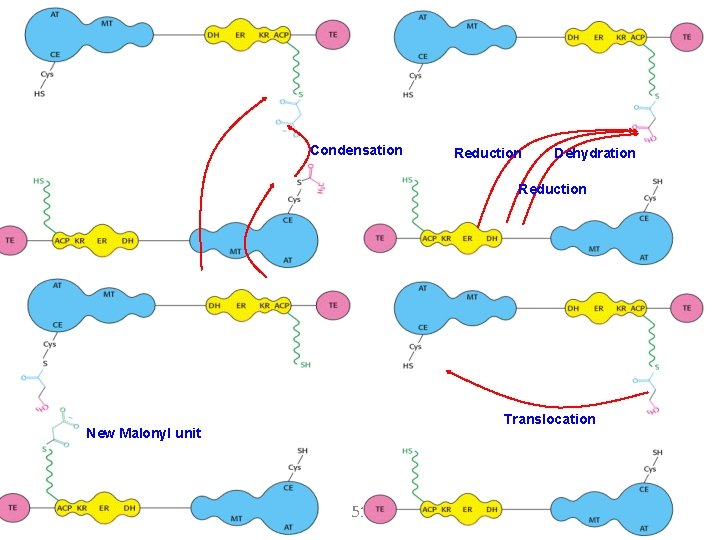
Condensation Reduction Dehydration Reduction Translocation New Malonyl unit 51

Five more rounds of condensation and reduction produce a palmitoyl (C 16) chain on the condensing enzyme. Palmitoyl is hydrolyzed to palmitate by the thioesterase on domain 3 of the opposite chain. The flexibility and 20 -Å maximal length of the phosphopantetheinyl moiety are critical for the function of this multienzyme complex. The enzyme subunits need not undergo large structural rearrangements to interact with the substrate. Instead, the substrate is on a long, flexible arm that can reach of the numerous active sites. 52

Elongation of Fatty Acids The major product of fatty acid synthase is palmitate. In eukaryotes, longer fatty acids are formed by reactions catalyzed by enzymes on the cytosolic face of the endoplasmic reticulum membrane. These reactions add two-carbon units sequentially to the carboxyl ends of both saturated and unsaturated fatty acyl Co. A substrates. Malonyl Co. A is the two-carbon donor in the elongation of fatty acyl Co. As. Condensation is driven by the decarboxylation of malonyl Co. A. 53

Membrane-Bound Enzymes Generate Unsaturated Fatty Acids Double bonds are introduced into long-chain acyl Co. As in endoplasmic reticulum systems. For example: in the conversion of stearoyl Co. A into oleoyl Co. A, a cis-D 9 double bond is inserted by an oxidase. 54

Mammals lack the enzymes to introduce double bonds at carbon atoms beyond C-9 in the fatty acid chain. Hence, mammals cannot synthesize linoleate (18: 2 cis-D 9, D 12) and linolenate (18: 3 cis-D 9, D 12, D 15). The two essential fatty acids linoleate and linolenate furnished by the diet are the starting points for the synthesis of a variety of other unsaturated fatty acids. 55
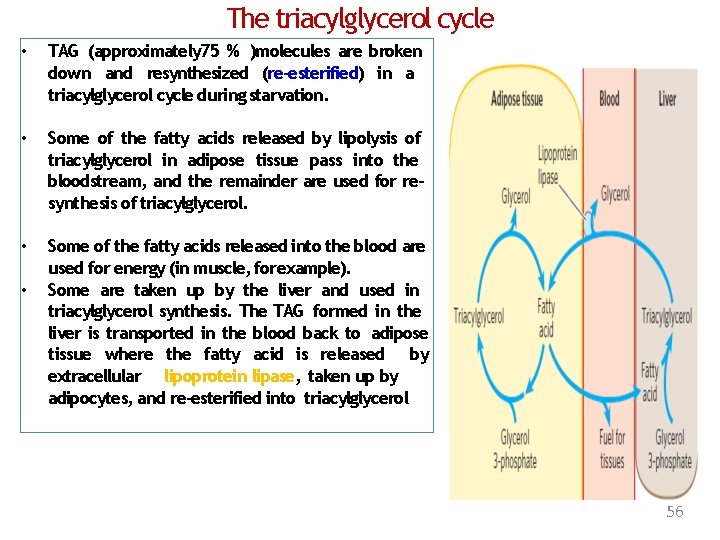
The triacylglycerol cycle • TAG (approximately 75 % )molecules are broken down and resynthesized (re-esterified) in a triacylglycerol cycle during starvation. • Some of the fatty acids released by lipolysis of triacylglycerol in adipose tissue pass into the bloodstream, and the remainder are used for resynthesis of triacylglycerol. • Some of the fatty acids released into the blood are used for energy (in muscle, forexample). Some are taken up by the liver and used in triacylglycerol synthesis. The TAG formed in the liver is transported in the blood back to adipose tissue where the fatty acid is released by extracellular lipoprotein lipase, taken up by adipocytes, and re-esterified into triacylglycerol • 56

• What is the source of glycerol 3 phosphate required theglycerol reesterification during starvation? for • Glycolysis is suppressed under starvation conditions so little DHAP is available • Glycerol released during lipolysis cannot be converted directly to glycerol 3 -phosphate in adipose tissue, because these cells lack glycerol kinase. 57

Adipose Tissue Generates Glycerol 3 -Phosphate by Glyceroneogenesis • Glyceroneogenesis is a shortened version Of gluconeogenesis, from pyruvate to DHAP, followed by conversion of the DHAP to glycerol 3 -phosphate by cytosolic NAD-linked glycerol 3 -phosphate dehydrogenase. 58

Diseases related to fatty acid metabolism 59

Carnitine deficiencies • Sources of carnitine: In meat products Enzymatic biosynthesis from lysine and methionine by in the liver and kidney but not in skeletal or heart muscle. Skeletal and heart muscle are totally dependent on carnitine provided by hepatocytes or the diet, and distributed by the blood. Skeletal muscle contains about 97 % of all carnitine in the body. • Consequences of a carnitine deficiency Decreased ability of tissues to use LCFA as a metabolic fuel. Accumulation of toxic amounts of free fatty acids and branched chain acyl groups in cells. 60

• Such deficiencies result in a decreased ability of tissues to use LCFAs as a fuel. Primary carnitine deficiency • caused by defects in a membrane transporter that prevent uptake of carnitine by cardiac and skeletal muscle and kidney. • Treatment includes carnitine supplementation. Secondary carnitine deficiency • occurs primarily as a result of defects in fatty acid oxidation leading to the accumulation of acylcarnitines that are excreted in the urine, decreasing carnitine availability. Acquired secondary carnitine deficiency Patients with liver diseases and or those taking the antiseizure drug valproic acid (decreased renal reabsorption). 61

Treatment: Avoidance of prolonged fasts Adaptation of a diet high in carbohydrates, low in LCFA, but supplemented with MCFA Oral supplementation with carnitine 62

Inhireted Carnitine deficiencies Carnitine Palmitoyltransferase I Deficiency • Carnitine palmitoyltransferase 1 A (CPT-1 A) deficiency is an inborn error of metabolism that affects mitochondrial oxidation of long chain fatty acids (LCFA) in the liver and kidneys, and is characterized by recurrent attacks of fastinginduced hypoketotic hypoglycemia and risk of liver failure. • CPT-1 A deficiency is due to mutations in the CPT 1 A gene that codes for the liver isoform of the CPT 1 enzyme located within the external mitochondrial membrane. 63

Diagnostics During metabolic crisis, blood tests reveal • hypoglycemia, • elevated levels of plasma carnitine and liver transaminase • mild hyperammonemia. 64

Management and treatment • Treatment consists primarily of avoidance of fasting. • Additional measures may be considered, including nighttime feeds with uncooked cornstarch during childhood and/or a low-fat diet supplemented with medium chain triglycerides that can be metabolized by mitochondria independently from the carnitine cycle. • Regular surveillance of liver enzymes and function is necessary. 65

Deficiencies in Acyl-Co. A Dehydrogenase There are four fatty acyl COA dehydrogenases in the mitochonderia: Very long-chain acyl-Co. A dehydrogenase (VLCAD) 22 or more carbons Long-chain acyl-Co. A dehydrogenase (LCAD) 13 -22 carbons Medium-chain acyl-Co. A dehydrogenase (MCAD) 6– 12 carbons Short-chain acyl-Co. A dehydrogenase (SCAD) Medium-chain fatty Co. A dehydrogenase (MCAD) deficiency MCAD is most common form of acyl-Co. A dehydrogenase deficiency Infants are the main affected group due to their reliance on milk, which contains MCAD It results in decreased ability to oxidize M CFA and thus in severe hypoglycemia (because the tissues must increase their reliance on glucose. Symptoms include vomiting, and excessive urinary excretion of medium-chain dicarboxylic acids (diagnostic marker) Treatment: avoidance of prolonged fasting + carbonhydrate rich diet. MCAD deficiency has been identified as the cause of some cases of sudden infant death syndrome (SIDS). 66

Peroxisomal deficiencies related to β-oxidation Refsum disease • Results from a genetic defect in phytanoyl-Co. A hydroxylase, • leads to very high blood levels of phytanic acid and severeneurological problems, including blindness and deafness. • Because phytanic acid is obtained solely from the diet, placing patients on a low phytanic acid diet (less beef and less milk products) has resulted in marked improvement. 67

Zellweger Syndrome • Zellweger syndrome is a genetic disease caused by a mutation in any one of several genes involved in peroxisome biogenesis. • The disease is characterized by a deficiency of peroxisomes that causes an accumulation of very long chain fatty acids (C 24 -C 26) and several unusual fatty acids, such as hydroxylated and branched fatty acids. • Mechanism: a defect in fatty acid efflux from peroxisomes. The most common features are enlarged liver, vision problems. • In affected infants, there is failure to grow, mental retardation, abnormal muscle tone, and multiple develop mental abnormalities. • Infants usually die within their first year. 68

Vitamin B 12 deficiency Propionate and methylmalonate are secreted in the urine of the patients (diagnostic biomarker). Disease results in metabolic acidosis with developmental retardation in some patients. Pernicious anemia results failure to absorb from efficiently from the intestine, where. B 12 it vitamin is synthesized by intestinal bacteria or obtained from digestion of meat. Administration of large doses of vitamin B 12 alleviates these symptoms in at least some cases. 69
- Slides: 69