Electrons Configurations Cartoon courtesy of Nearing Zero net

























- Slides: 42

Electrons Configurations Cartoon courtesy of Nearing. Zero. net

Wave-Particle Duality JJ Thomson won the Nobel prize for describing the electron as a particle. His son, George Thomson won the Nobel prize for describing the wave-like nature of the electron. The electron is a particle! The electron is an energy wave!

The Wave-like Electron The electron propagates through space as an energy wave. To understand the atom, one must understand the behavior of electromagnetic waves. Louis de. Broglie

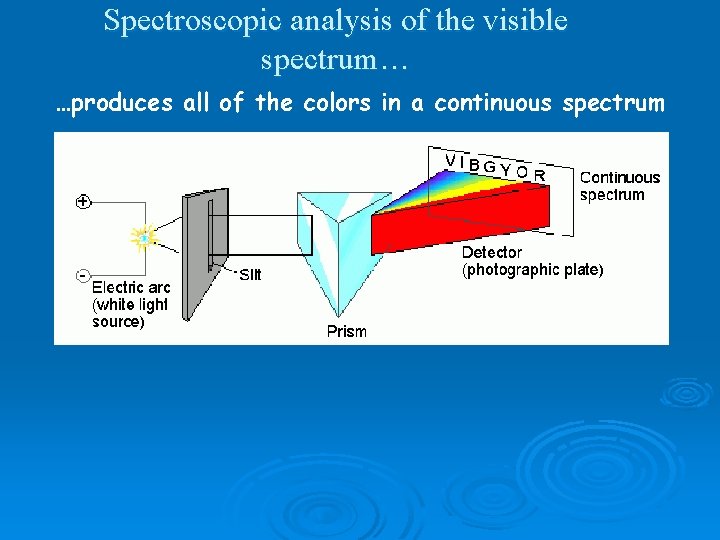
Spectroscopic analysis of the visible spectrum… …produces all of the colors in a continuous spectrum

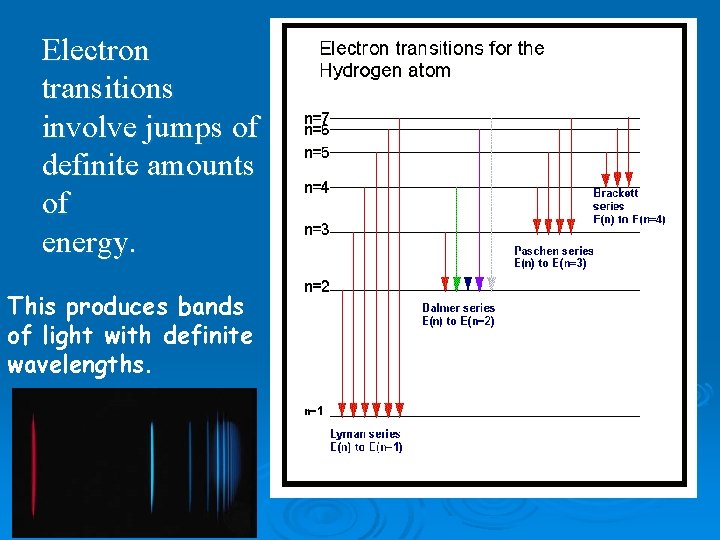
Electron transitions involve jumps of definite amounts of energy. This produces bands of light with definite wavelengths.

Quantum Numbers Each electron in an atom has a unique set of 4 quantum numbers which describe it. v v Principal quantum number Angular momentum quantum number Magnetic quantum number Spin quantum number

Pauli Exclusion Principle No two electrons in an atom can have the same four quantum numbers. Wolfgang Pauli

Principal Quantum Number Generally symbolized by n, it denotes the shell (energy level) in which the electron is located. Number of electrons that can fit in a shell: 2 n 2

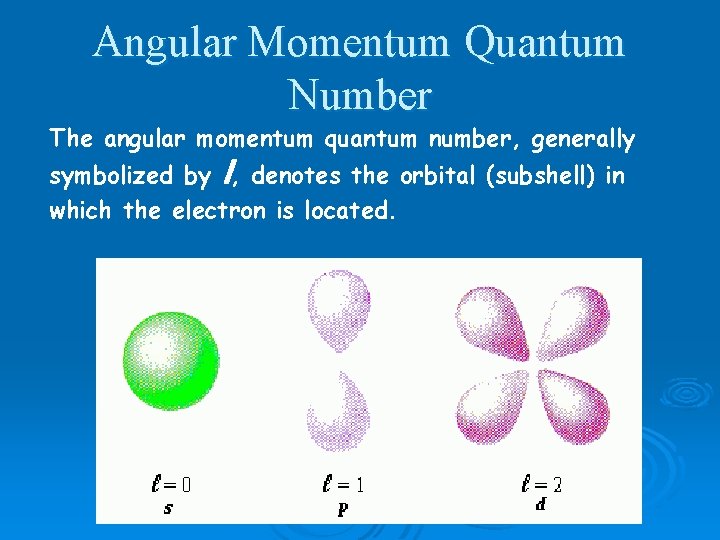
Angular Momentum Quantum Number The angular momentum quantum number, generally symbolized by l, denotes the orbital (subshell) in which the electron is located.

Magnetic Quantum Number The magnetic quantum number, generally symbolized by m, denotes the orientation of the electron’s orbital with respect to the three axes in space.

Assigning the Numbers v The three quantum numbers (n, l, and m) are integers. v The principal quantum number (n) cannot be zero. v n must be 1, 2, 3, etc. v The angular momentum quantum number (l) can be any integer between 0 and n - 1. v For n = 3, l can be either 0, 1, or 2. v The magnetic quantum number (m) can be any integer between -l and +l. v For l = 2, m can be either -2, -1, 0, +1, or +2.

Spin Quantum Number Spin quantum number denotes the behavior (direction of spin) of an electron within a magnetic field. Possibilities for electron spin:

Uncertainty Principle Ø In 1927 by Werner Heisenberg (German theoretical physicist) Ø electrons can only be detected by their interaction with light Ø any attempt to locate a specific electron knocks the electron off course

Uncertainty Principle Ø Heisenberg Uncertainty Principle- it is impossible to know both the position and velocity of an electron Ø We can find the most likely position of an electron

Orbitals Ø each sublevel is broken into orbitals Ø each orbital can hold a maximum of 2 electrons Ø orbital- a 3 D region around the nucleus that has a high probability of holding electrons

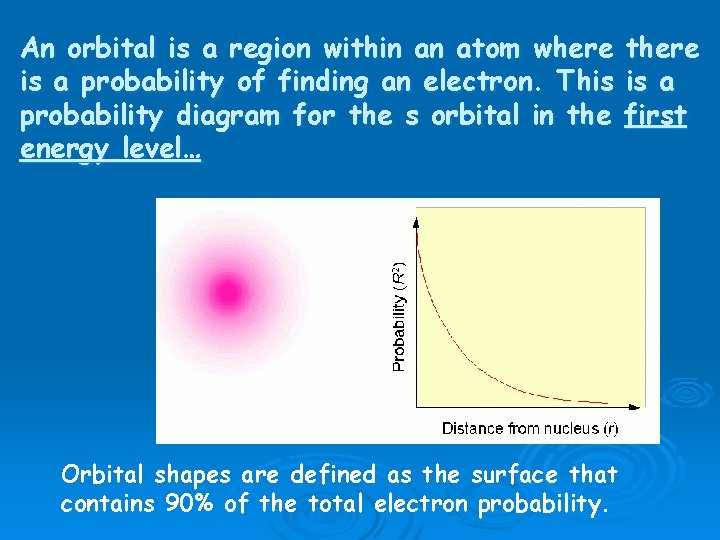
An orbital is a region within an atom where is a probability of finding an electron. This probability diagram for the s orbital in the energy level… Orbital shapes are defined as the surface that contains 90% of the total electron probability. there is a first

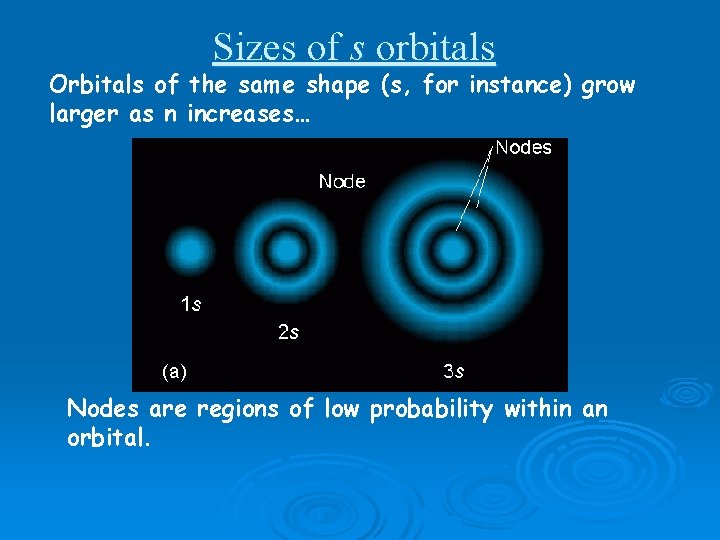
Sizes of s orbitals Orbitals of the same shape (s, for instance) grow larger as n increases… Nodes are regions of low probability within an orbital.

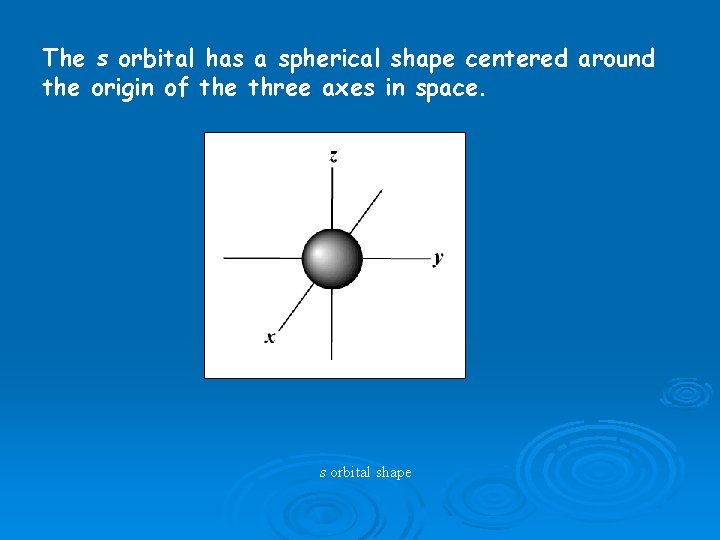
The s orbital has a spherical shape centered around the origin of the three axes in space. s orbital shape

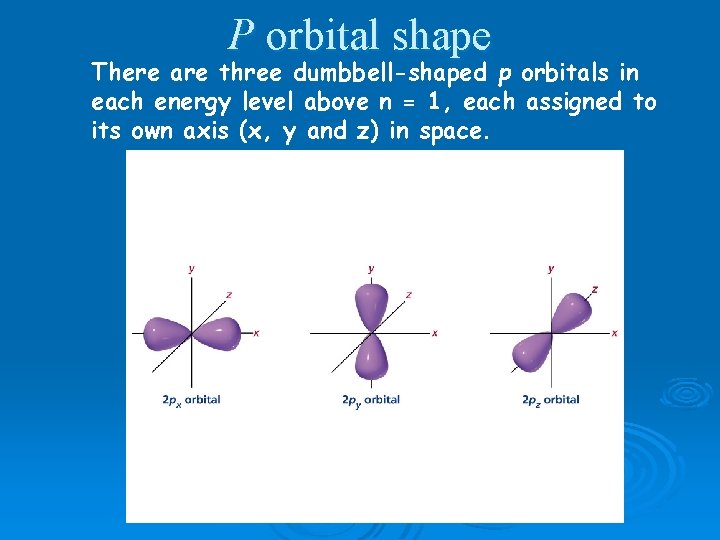
P orbital shape There are three dumbbell-shaped p orbitals in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.

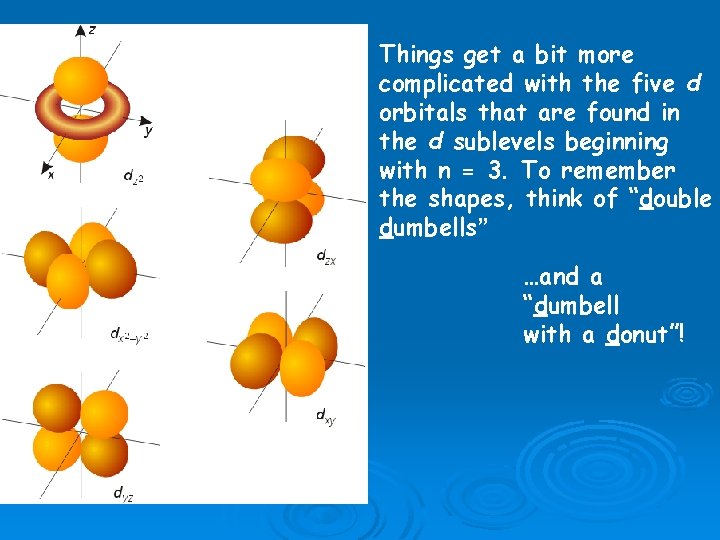
Things get a bit more complicated with the five d orbitals that are found in the d sublevels beginning with n = 3. To remember the shapes, think of “double dumbells” …and a “dumbell with a donut”!

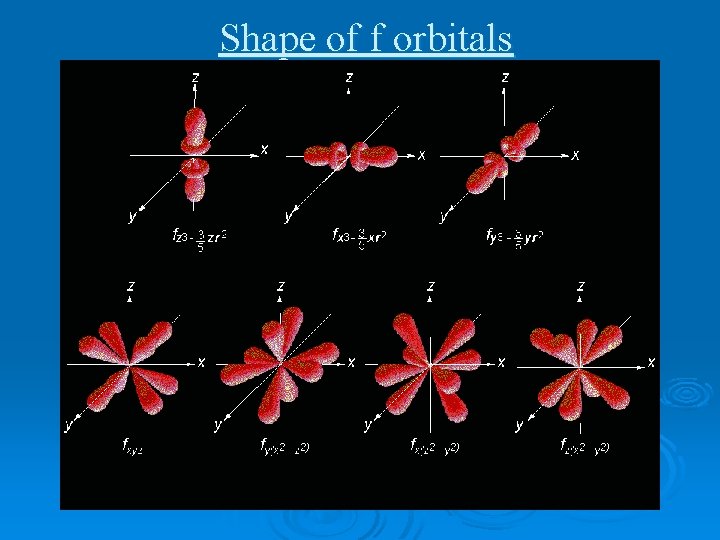
Shape of f orbitals

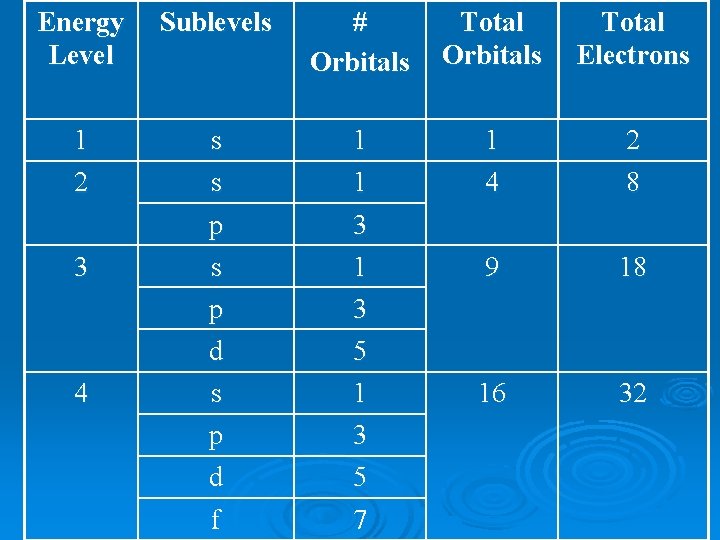
Number of Orbitals sublevel s p d f max # e- # orbitals 2 1 6 3 10 5 14 7

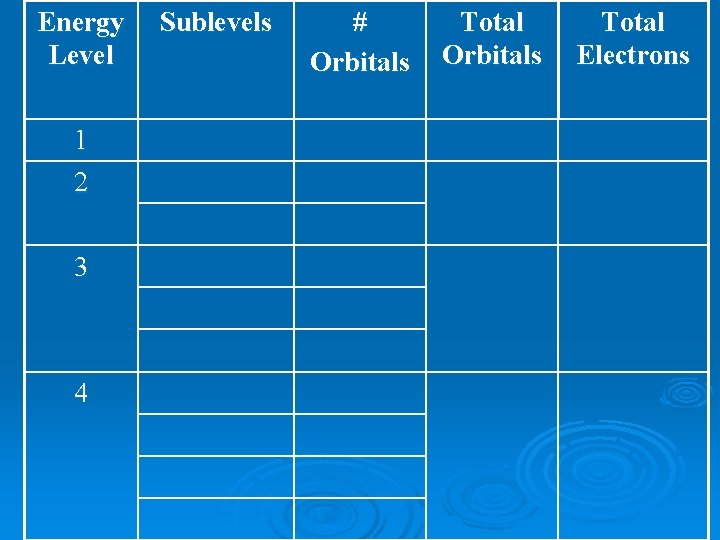
Energy Level 1 2 3 4 Number of Types of Sublevels 1 s 2 s, p 3 s, p, d 4 s, p, d, f

Energy Level 1 2 3 4 Sublevels # Orbitals Total Electrons

Energy Level Sublevels # Orbitals Total Electrons 1 2 s s 1 1 1 4 2 8 3 p s 3 1 9 18 p d 3 5 s p 1 3 16 32 d f 5 7 4

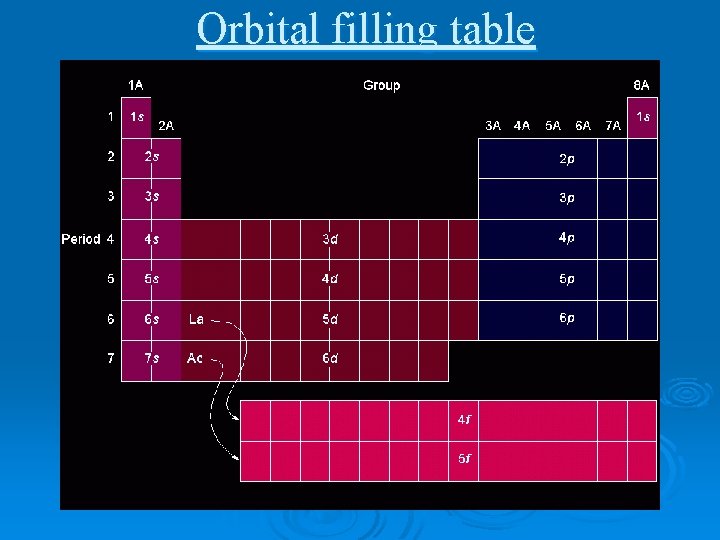
Orbital filling table

Electron configuration of the elements of the first three series

Irregular confirmations of Cr and Cu Chromium steals a 4 s electron to half fill its 3 d sublevel Copper steals a 4 s electron to FILL its 3 d sublevel

Electron Configurations Ø the arrangement of electrons in an atom Ø each type of atom has a unique electron configuration Ø electrons tend to assume positions that create the lowest possible energy for atom Ø ground state electron configuration- lowest energy arrangement of electrons

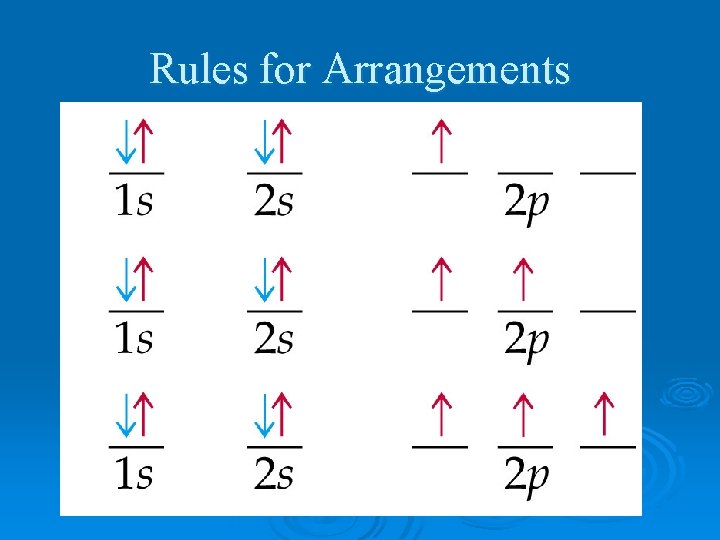
Rules for Arrangements Ø Aufbau Principle- an electron occupies the lowest-energy orbital that can receive it Ø Beginning in the 3 rd energy level, the energies of the sublevels in different energy levels begin to overlap

Rules for Arrangements Ø Pauli Exclusion Principle- no two electrons in the same atom can have the same set of 4 quantum numbers Ø Hund’s Rule- orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second Ø all unpaired electrons must have the same spin

Rules for Arrangements

Writing Configurations Ø Orbital Notation: l l l an orbital is written as a line each orbital has a name written below it electrons are drawn as arrows (up and down) Ø Electron Configuration Notation l number of electrons in sublevel is added as a superscript

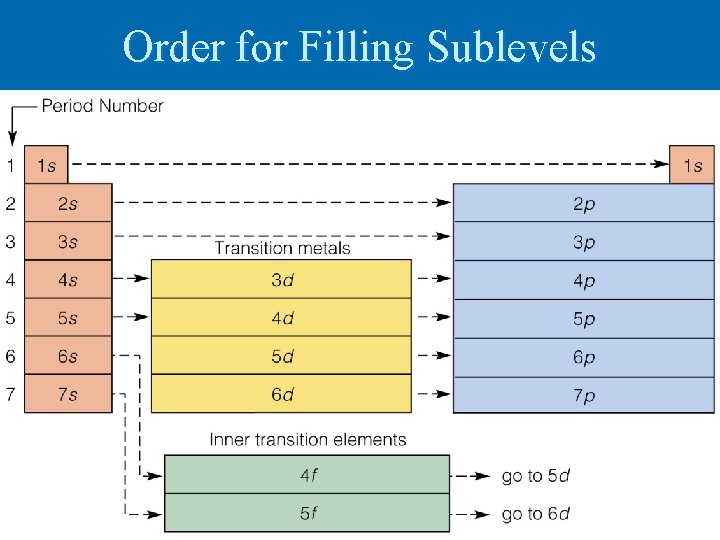
Order for Filling Sublevels

Writing Configurations Start by finding the number of electrons in the atom Ø Identify the sublevel that the last electron added is in by looking at the location in periodic table Ø Draw out lines for each orbital beginning with 1 s and ending with the sublevel identified Ø Add arrows individually to the orbitals until all electrons have been drawn Ø

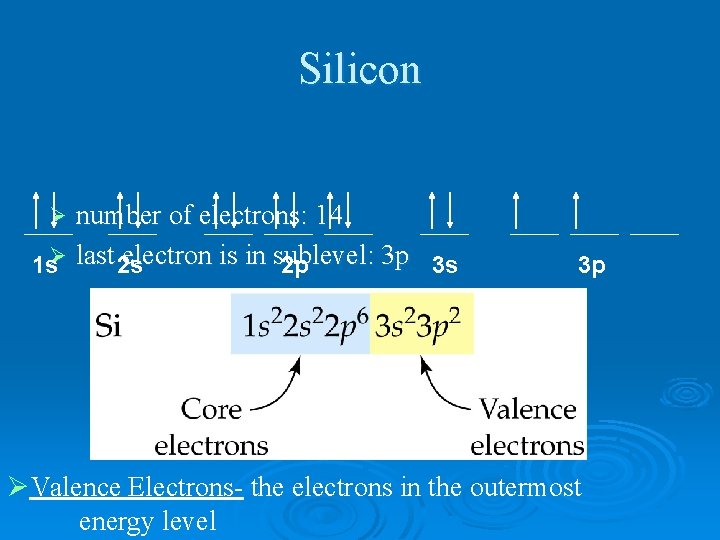
Silicon number of electrons: 14 electron is in sublevel: 3 p 3 s 1 sØ last 2 s 2 p Ø 3 p ØValence Electrons- the electrons in the outermost energy level

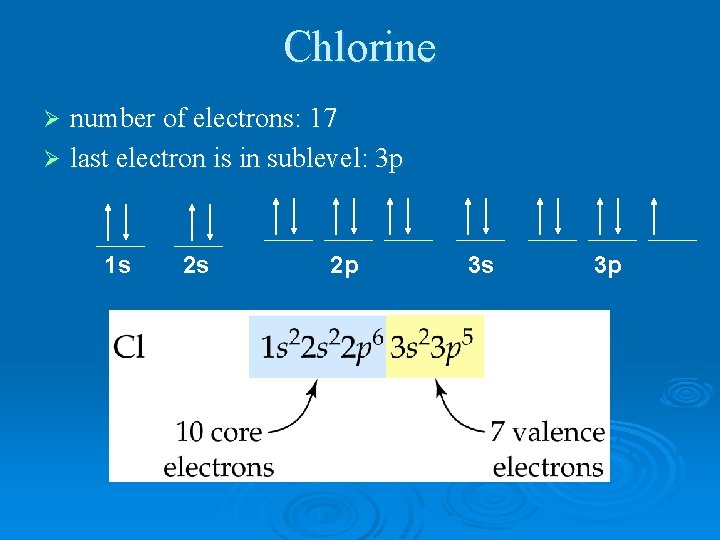
Chlorine number of electrons: 17 Ø last electron is in sublevel: 3 p Ø 1 s 2 s 2 p 3 s 3 p

Sodium number of electrons: 11 Ø last electron is in sublevel: 3 s Ø 1 s 2 s 1 s 2 2 p 6 3 s 1 2 p 3 s

Calcium number of electrons: 20 Ø last electron is in sublevel: 4 s Ø 1 s 2 s 3 p 2 p 4 s 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 s

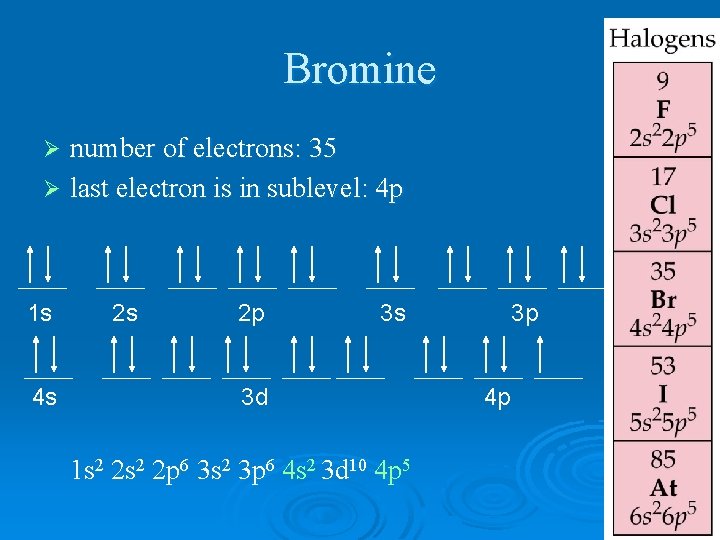
Bromine number of electrons: 35 Ø last electron is in sublevel: 4 p Ø 1 s 4 s 2 s 2 p 3 s 3 d 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 5 3 p 4 p

Argon number of electrons: 18 Ø last electron is in sublevel: 3 p Ø 1 s 2 s 2 p 1 s 2 2 p 6 3 s 2 3 p 6 3 s 3 p

Noble Gas Notation short hand for larger atoms Ø configuration for the last noble gas is abbreviated by the noble gas’s symbol in brackets Ø